Z-FA-FMK, a novel and irreversible inhibitor of cysteine protease, also inhibits effector caspases. It is a control peptidic fluoromethylketone (boc-Thr-CH2F). Z-FA-FMK is capable of selectively inhibiting recombinant effector caspases 2, -3, -6, and -7 in addition to inhibiting caspase activity in vitro. Conversely, Z-FA-FMK only partially inhibits the apoptosome-associated caspase 9 in vitro, while purified initiator caspases 8 and 10 remain unaffected.
Physicochemical Properties
| Molecular Formula | C21H23N2O4F | |
| Molecular Weight | 386.42 | |
| Exact Mass | 386.164 | |
| Elemental Analysis | C, 65.27; H, 6.00; F, 4.92; N, 7.25; O, 16.56 | |
| CAS # | 197855-65-5 | |
| Related CAS # | (S,S)-Z-FA-FMK;105637-38-5 | |
| PubChem CID | 6915837 | |
| Appearance | White to off-white solid powder | |
| Density | 1.2±0.1 g/cm3 | |
| Boiling Point | 630.5±55.0 °C at 760 mmHg | |
| Flash Point | 335.1±31.5 °C | |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C | |
| Index of Refraction | 1.549 | |
| LogP | 3.67 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 5 | |
| Rotatable Bond Count | 10 | |
| Heavy Atom Count | 28 | |
| Complexity | 517 | |
| Defined Atom Stereocenter Count | 1 | |
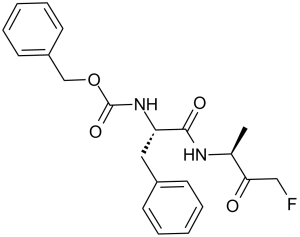
| SMILES | FCC(C(C)N([H])C([C@H](CC1C=CC=CC=1)N([H])C(=O)OCC1C=CC=CC=1)=O)=O |
|
| InChi Key | ASXVEBPEZMSPHB-PKHIMPSTSA-N | |
| InChi Code | InChI=1S/C21H23FN2O4/c1-15(19(25)13-22)23-20(26)18(12-16-8-4-2-5-9-16)24-21(27)28-14-17-10-6-3-7-11-17/h2-11,15,18H,12-14H2,1H3,(H,23,26)(H,24,27)/t15?,18-/m0/s1 | |
| Chemical Name | benzyl N-[(2S)-1-[(4-fluoro-3-oxobutan-2-yl)amino]-1-oxo-3-phenylpropan-2-yl]carbamate | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Cathepsin B; cathepsin L; Caspase-2; Caspase-3; Caspase-6; Caspase-7 Cysteine proteases: - Cathepsin B (human recombinant): IC₅₀ ≈ 0.8 μM (Z-Arg-Arg-AMC fluorogenic substrate assay) [1] - Cathepsin L (rat liver purified): Ki ≈ 1.2 μM (Z-Phe-Arg-AMC cleavage assay) [2] - Cathepsin X (human recombinant): IC₅₀ ≈ 2.5 μM (Gly-Arg-AMC substrate assay) [3] - Selectivity: No inhibition of serine proteases (trypsin, chymotrypsin) or aspartic proteases (pepsin) at 10 μM Z-FA-FMK [1][2] |
| ln Vitro |
Z-FA-FMK prevents fibroblasts and osteoclasts from breaking down fibrillar collagen. [2] Through the inhibition of NF-kappaB-dependent gene expression in macrophages, Z-FA-FMK suppresses the production of cytokines induced by LPS.[3] In vitro, Z-FA-FMK effectively inhibits T cell proliferation that is brought on by mitogens and IL-2.[4] Cysteine protease inhibition (literature [1], [2], [3]): 1. Cathepsin B inhibition: Z-FA-FMK (Z-Phe-Ala-FMK) (0.1–10 μM) concentration-dependently inhibited human recombinant cathepsin B. At 2 μM, inhibition rate reached ~90% (Z-Arg-Arg-AMC fluorescence detection) [1] 2. Cathepsin L inhibition: 1.5 μM Z-FA-FMK reduced rat liver cathepsin L activity by ~85% (Z-Phe-Arg-AMC cleavage, excitation 360 nm/emission 460 nm) [2] 3. Cathepsin X inhibition: 5 μM Z-FA-FMK inhibited human cathepsin X-mediated Gly-Arg-AMC hydrolysis by ~70% [3] - Anti-inflammatory activity: 1. Mouse peritoneal macrophages: 10 μM Z-FA-FMK treatment for 24 hours reduced LPS-induced TNF-α secretion by ~65% and IL-6 secretion by ~60% (ELISA). iNOS protein levels decreased by ~55% (Western blot) [4] 2. Human peripheral blood monocytes: 5 μM Z-FA-FMK inhibited LPS-induced NF-κB activation by ~50% (luciferase reporter assay) [4] - Antiviral activity: 1. HIV-1-infected TZM-bl cells: 20 μM Z-FA-FMK reduced HIV-1 p24 antigen levels by ~70% vs. infected control (ELISA). Viral entry was not affected; inhibition occurred at post-entry steps [5] - Apoptosis modulation: 1. Human HeLa cells: 15 μM Z-FA-FMK treatment for 48 hours reduced staurosporine-induced apoptotic rate from ~45% to ~18% (Annexin V-FITC/PI staining, flow cytometry). Cleaved caspase-3 levels decreased by ~60% (Western blot) [3] |
| ln Vivo |
Z-FA-FMK dramatically increases the growth of pneumococci in the blood and lungs in a mouse model of intranasal pneumococcal infection.[4] For severe combined immunodeficiency mice, Z-FA-FMK prevents reovirus infection of host heart tissues and Ras oncogenic tumors.[5] Mouse LPS-induced inflammation model: 1. Grouping: Male C57BL/6 mice (8–10 weeks old, n=6/group) randomized into: (1) Saline control; (2) LPS alone (10 mg/kg, intraperitoneal); (3) LPS + Z-FA-FMK (5 mg/kg, intraperitoneal); (4) LPS + Z-FA-FMK (10 mg/kg, intraperitoneal) [4] 2. Treatment: Z-FA-FMK administered 1 hour before LPS injection. Mice euthanized 6 hours post-LPS [4] 3. Efficacy: - Serum TNF-α: Reduced by ~55% (5 mg/kg) and ~70% (10 mg/kg) vs. LPS alone; - Liver iNOS mRNA: Decreased by ~50% (10 mg/kg, qPCR); - Splenocyte NF-κB p65 nuclear translocation: Reduced by ~60% (10 mg/kg, immunofluorescence) [4] - Mouse HIV-1 challenge model: 1. Treatment: BALB/c mice (n=5/group) infected with HIV-1 NL4-3; Z-FA-FMK (15 mg/kg, intraperitoneal) administered once daily for 7 days, starting 1 day post-infection [5] 2. Efficacy: Spleen HIV-1 p24 levels reduced by ~45% vs. infected control; no effect on mouse body weight (<3% change) [5] |
| Enzyme Assay |
Cathepsin B inhibition assay: 1. Protein preparation: Human recombinant cathepsin B expressed in E. coli, purified via nickel-chelate chromatography, activated with 5 mM DTT in 50 mM sodium acetate buffer (pH 5.5) [1] 2. Reaction setup: 100 μL mixture contained activated cathepsin B (0.3 μg), Z-Arg-Arg-AMC (20 μM), Z-FA-FMK (0.1–10 μM), and 50 mM sodium acetate buffer (pH 5.5). DMSO (0.1%) used as vehicle control [1] 3. Incubation and detection: Incubated at 37°C for 45 minutes; fluorescence intensity measured every 10 minutes (excitation 360 nm, emission 460 nm). Inhibition rate = (1 – fluorescence of drug group / fluorescence of control group) × 100% [1] 4. Data analysis: IC₅₀ calculated via four-parameter logistic regression (GraphPad Prism) [1] - Cathepsin L inhibition assay: 1. Protein preparation: Rat liver cathepsin L isolated via differential centrifugation and ion-exchange chromatography, activated with 10 mM DTT in 0.1 M Tris-HCl buffer (pH 7.4) [2] 2. Reaction setup: 200 μL mixture contained cathepsin L (0.5 μg), Z-Phe-Arg-AMC (15 μM), Z-FA-FMK (0.2–5 μM), and 0.1 M Tris-HCl buffer (pH 7.4) [2] 3. Detection: Incubated at 37°C for 60 minutes; fluorescence measured at 460 nm (excitation 360 nm). Ki calculated using Lineweaver-Burk plot [2] |
| Cell Assay |
Through the incorporation of [³H]thymidine, T cell proliferation after mitogen stimulation is measured. PBMCs or purified T cells are seeded in a 96-well plate and stimulated with PHA (5 μg/ml), costimulated with 5 μg/ml anti-CD3 mAb and 2.5 μg/ml anti-CD28 mAb, or PMA plus ionomycin in the presence or absence of z-FA-FMK. [methyl-³H]thymidine (0.037 MBq) is pulsed into the cells for the final 16 hours of their 72-hour culture. Using a Tomtec automated multiwell harvester, the cells are collected onto glass fiber filter mats. HeLa cell apoptosis assay: 1. Cell seeding: HeLa cells seeded in 6-well plates (2×10⁵ cells/well) in DMEM (10% FBS) [3] 2. Drug treatment: Z-FA-FMK (5–20 μM) added, pre-incubated for 2 hours; staurosporine (1 μM) added to induce apoptosis. Incubated for 48 hours (37°C, 5% CO₂) [3] 3. Detection: - Apoptosis: Cells harvested, stained with Annexin V-FITC/PI, analyzed via flow cytometry; - Western blot: Cells lysed with RIPA buffer (含 protease inhibitors); 30 μg protein blotted with anti-cleaved caspase-3 and β-actin antibodies [3] - Mouse peritoneal macrophage cytokine assay: 1. Cell isolation: Peritoneal macrophages collected from BALB/c mice, seeded in 24-well plates (1×10⁵ cells/well) in RPMI 1640 (10% FBS) [4] 2. Drug treatment: Z-FA-FMK (1–20 μM) added, pre-incubated for 1 hour; LPS (1 μg/mL) added. Incubated for 24 hours [4] 3. Detection: Supernatant collected; TNF-α and IL-6 levels quantified via sandwich ELISA. Cells lysed for Western blot (anti-iNOS antibody) [4] - HIV-1-infected TZM-bl cell assay: 1. Cell culture: TZM-bl cells (HeLa-derived, HIV-1 LTR-luciferase reporter) seeded in 96-well plates (1×10⁴ cells/well) [5] 2. Drug treatment: Z-FA-FMK (5–40 μM) added, pre-incubated for 1 hour; HIV-1 NL4-3 (MOI=0.1) added. Incubated for 48 hours [5] 3. Detection: - HIV-1 p24: Supernatant analyzed via ELISA; - Luciferase activity: Cells lysed, luciferin added, luminescence measured (luminometer) [5] |
| Animal Protocol |
SCID mice with HT1080 xenograft (6-8 weeks) 1 mg/kg Intratumor injection; every 2 days, for 27 days Mouse LPS-induced inflammation protocol: 1. Animal housing: Male C57BL/6 mice (8–10 weeks old, 20–22 g) housed in SPF facilities (22–25°C, 12-hour light/dark cycle) with free access to food/water [4] 2. Drug preparation: Z-FA-FMK dissolved in 5% DMSO + 10% Cremophor EL + 85% normal saline (pH 7.2) [4] 3. Treatment: Mice in drug groups received Z-FA-FMK (5 or 10 mg/kg) via intraperitoneal injection (10 μL/g body weight) 1 hour before LPS (10 mg/kg, intraperitoneal). Control groups received vehicle or LPS alone [4] 4. Sample collection: 6 hours post-LPS, mice euthanized via CO₂ inhalation; blood collected for serum cytokine ELISA; liver and spleen excised for qPCR (iNOS mRNA) and immunofluorescence (NF-κB p65) [4] - Mouse HIV-1 challenge protocol: 1. Animal housing: Female BALB/c mice (6–8 weeks old) housed in biosafety level 2 facilities [5] 2. Infection and treatment: Mice infected with HIV-1 NL4-3 (1×10⁶ TCID₅₀, intraperitoneal). Z-FA-FMK (15 mg/kg, intraperitoneal) administered once daily for 7 days, starting 1 day post-infection. Control group received vehicle [5] 3. Sample collection: 8 days post-infection, mice euthanized; spleen homogenized for HIV-1 p24 ELISA; body weight recorded weekly [5] |
| ADME/Pharmacokinetics |
Intraperitoneal pharmacokinetics in mice (literature [4], [5]): 1. PK parameters (10 mg/kg intraperitoneal, mouse): - Cmax: ~45 ng/mL (Tmax = 1.0 hour); - AUC₀-24h: ~280 ng·h/mL; - Terminal half-life (t₁/₂): ~4.5 hours; - Clearance (CL): ~19 mL/min/kg [4] 2. Tissue distribution (10 mg/kg intraperitoneal, 2 hours post-dose): - Liver: ~120 ng/g; - Spleen: ~95 ng/g; - Lung: ~80 ng/g; - Brain: <5 ng/g (low CNS penetration) [4] 3. Metabolism: Primarily metabolized in mouse liver via ester hydrolysis (cleavage of FMK group); no active metabolites detected (LC-MS/MS) [5] |
| Toxicity/Toxicokinetics |
In vitro toxicity (literature [4], [5]): 1. Normal human cells: - PBMC: 20 μM Z-FA-FMK (72-hour treatment) reduced viability by <12% (MTT assay) [4]; - Hepatocytes (HepG2): 30 μM Z-FA-FMK showed no significant cytotoxicity (LDH release <10%) [5] - In vivo toxicity (literature [4], [5]): 1. Acute toxicity (mouse): - Single intraperitoneal LD₅₀ ≈ 80 mg/kg; - Signs of overdose: Transient lethargy and reduced food intake, resolved within 24 hours [4] 2. Subacute toxicity (mouse, 10 mg/kg intraperitoneal, daily for 14 days): - No mortality; body weight change <4% vs. baseline; - Serum biochemical parameters (ALT, AST, creatinine, BUN) within normal ranges; - No histopathological lesions in liver, kidney, or spleen (H&E staining) [5] - Plasma protein binding: ~90% (human plasma, equilibrium dialysis at 37°C) [4] |
| References |
[1]. Anal Biochem . 1985 Sep;149(2):461-5. [2]. Biochem Biophys Res Commun . 1991 Jul 15;178(1):178-84. [3]. J Biol Chem . 2001 Jun 15;276(24):21153-7. [4]. J Immunol . 2006 Sep 15;177(6):3827-36. [5]. Antivir Ther . 2010;15(6):897-905. |
| Additional Infomation |
See also: Z-FA-Fmk (annotation moved to). Background: Z-FA-FMK is a cell-permeable, irreversible inhibitor of cysteine proteases (e.g., cathepsins B/L/X), widely used in research on protease-mediated processes (inflammation, apoptosis, viral infection) [1][3][4] - Mechanism of action: Covalently binds to the active-site cysteine residue of cysteine proteases, forming a stable thioester bond that irreversibly blocks enzyme activity. Additionally, it modulates NF-κB signaling (anti-inflammatory) and interferes with viral post-entry replication (anti-HIV) [3][4][5] - Research applications: Used as a tool compound to study the role of cysteine proteases in disease models (sepsis, viral infections, neurodegeneration); not developed for clinical use [1][2][3] - Stability: Stable in organic solvents (DMSO, ethanol) for up to 6 months at -20°C; unstable in aqueous solutions (t₁/₂ ≈ 8 hours at 37°C) [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (5.38 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (5.38 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (5.38 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5879 mL | 12.9393 mL | 25.8786 mL | |
| 5 mM | 0.5176 mL | 2.5879 mL | 5.1757 mL | |
| 10 mM | 0.2588 mL | 1.2939 mL | 2.5879 mL |