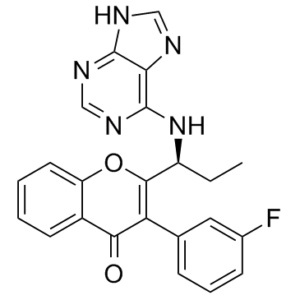
Tenalisib (also known as RP6530) is a new, potent and selective dual PI3Kδ/γ inhibitor with anticancer activity. With IC50 values of 24.5 nM and 33.2 nM, respectively, it inhibits both PI3Kδ and PI3Kγ. With selectivity over the α and β isoforms of more than 300-fold and 100-fold, respectively, it exhibits selective for PI3Kδ/γ. Both HEL-RS and HEL-RR cells are moderately inhibited in terms of proliferation by tenalisib (33-46% inhibition at 10 μM). The combination of 10 μM tenalisib and ruxolitinib inhibits proliferation almost completely (>90% for HEL-RS and >70% for HEL-RR). The EC50 of ruxolitinib (5.8 M) in HEL-RR cells is significantly decreased when 5 M tenalisib is added 4 hours beforehand.
Physicochemical Properties
| Molecular Formula | C23H18FN5O2 | |
| Molecular Weight | 415.42 | |
| Exact Mass | 415.144 | |
| Elemental Analysis | C, 66.50; H, 4.37; F, 4.57; N, 16.86; O, 7.70 | |
| CAS # | 1639417-53-0 | |
| Related CAS # | Tenalisib R Enantiomer;1639417-54-1; 1639417-53-0; 1693773-94-2 | |
| PubChem CID | 86291103 | |
| Appearance | white solid powder | |
| Density | 1.4±0.1 g/cm3 | |
| Index of Refraction | 1.715 | |
| LogP | 5.11 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 7 | |
| Rotatable Bond Count | 5 | |
| Heavy Atom Count | 31 | |
| Complexity | 704 | |
| Defined Atom Stereocenter Count | 1 | |
| SMILES | FC1=C([H])C([H])=C([H])C(=C1[H])C1C(C2=C([H])C([H])=C([H])C([H])=C2OC=1[C@]([H])(C([H])([H])C([H])([H])[H])N([H])C1C2=C(N=C([H])N=1)N=C([H])N2[H])=O |
|
| InChi Key | HDXDQPRPFRKGKZ-INIZCTEOSA-N | |
| InChi Code | InChI=1S/C23H18FN5O2/c1-2-16(29-23-19-22(26-11-25-19)27-12-28-23)21-18(13-6-5-7-14(24)10-13)20(30)15-8-3-4-9-17(15)31-21/h3-12,16H,2H2,1H3,(H2,25,26,27,28,29)/t16-/m0/s1 | |
| Chemical Name |
|
|
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | PI3Kδ (IC50 = 25 nM); PI3Kγ (IC50 = 33 nM) | ||
| ln Vitro | Tenalisib shows selectivity over PI3K α (>300-fold) and β (>100-fold) isoforms. In both HEL-RS and HEL-RR cells, tenalisib mildly inhibits proliferation (33-46% inhibition @ 10 μM). Inhibition of proliferation is nearly 100% (>90% for HEL-RS and >70% for HEL-RR) when 10 μM tenalisib is added to ruxolitinib. The EC50 of ruxolitinib (5.8 μM ) in HEL-RR cells is significantly decreased by the addition of 5 μM tenalisib 4 hours before the addition of ruxolitinib. When 10 μM tenalisib and ruxolitinib are combined for 72 hours, more cells undergo apoptosis (55% in HEL-RS and 37% in HEL-RR) than when either agent is used alone (16-27% in HEL-RS and 17-21% in HEL-RR)[1]. | ||
| ln Vivo | Tenalisib has been well tolerated in subjects with heavily pre-treated relapsed/refractory hematologic malignancies. With no DLTs, reported toxicities are controllable. At less than 200 mg BID, single agent activity can be seen in patients who are challenging to treat[2]. | ||
| Enzyme Assay | Myelofibrosis (MF) represents a life-threatening neoplasm that manifests particularly in the elderly population and is characterized by bone marrow fibrosis and extramedullary hematopoeisis. While ruxolitinib, a JAK1/2 inhibitor, has recently been approved by the USFDA for its disease modifying potential in MF patients, it is still not considered as a curative option. Targeting another kinase such as PI3K, downstream of JAK, could therefore be a more efficient way of treating myelofibrotic neoplasms. RP6530 is a novel, potent, and selective PI3K δ/γ inhibitor that demonstrated high potency against PI3Kδ (IC50 = 25 nM) and γ (IC50 = 33 nM) enzymes with selectivity over α (>300-fold) and β (>100-fold) isoforms. The objective of this study was to evaluate the effect of a combination of ruxolitinib and RP6530 in the JAK2-V617F mutant Human Erythroleukemia (HEL) cell line[1]. | ||
| Cell Assay | Potential of the combination (1 μM RP6530 + BORT between 0.1 nM and 1 μM) is studied in MM cell lines, namely, MM-1S and MM-1R. After incubating with the compound for 72 hours at 37°C, proliferation is assessed using a MTT assay. The combination's apoptotic potential is calculated using AnnexinV/PI staining. | ||
| Animal Protocol |
|
||
| References |
[1]. RP6530, a dual PI3K δ/γ inhibitor, potentiates ruxolitinib activity in the JAK2-V617F mutant erythroleukemia cell lines. In: Proceedings of the 106th Annual Meeting of the American Association for Cancer Research; Cancer Res 2015;75(15 Suppl):Abstract nr 2704. doi:10.1158/1538-7445.AM2015-2704. [2]. A Dose Escalation Study of RP6530, a Novel Dual PI3K Delta/Gamma Inhibitor, in Patients with Relapsed/Refractory Hematologic Malignancies. Blood 2015 126:1495; |
||
| Additional Infomation |
Tenalisib is under investigation in clinical trial NCT03711604 (Compassionate Use Study of Tenalisib (RP6530)). Tenalisib is an orally active, highly selective, small molecule inhibitor of the delta and gamma isoforms of phosphoinositide-3 kinase (PI3K) with potential immunomodulating and antineoplastic activities. Upon administration, tenalisib inhibits the PI3K delta and gamma isoforms and prevents the activation of the PI3K/AKT-mediated signaling pathway. This may lead to a reduction in cellular proliferation in PI3K delta/gamma-expressing tumor cells. In addition, this agent modulates inflammatory responses through various mechanisms, including the inhibition of both the release of reactive oxygen species (ROS) from neutrophils and tumor necrosis factor (TNF)-alpha activity. Unlike other isoforms of PI3K, the delta and gamma isoforms are overexpressed primarily in hematologic malignancies and in inflammatory and autoimmune diseases. By selectively targeting these isoforms, PI3K signaling in normal, non-neoplastic cells is minimally impacted or not affected at all, which minimizes the side effect profile for this agent. |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.02 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.02 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (6.02 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4072 mL | 12.0360 mL | 24.0720 mL | |
| 5 mM | 0.4814 mL | 2.4072 mL | 4.8144 mL | |
| 10 mM | 0.2407 mL | 1.2036 mL | 2.4072 mL |