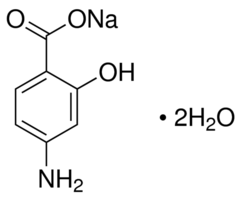
Sodium 4-Aminosalicylate, the sodium salt of aminosalicylic acid and an analog of PABA/para-aminobenzoic acid, is an antibiotic used to treat MDR-TB/multidrug-resistant tuberculosis via NF-κB inhibition and free radical scavenging. Sodium 4-Aminosalicylate competes with PABA for enzymes necessary for the synthesis of folate in order to exert its bacteriostatic activity against Mycobacterium tuberculosis. It responds quickly to DPPH, indicating a potent anti-radical activity. A chain-breaking type of antioxidant activity is suggested by the 4-aminosalicylate's rapid scavenging of peroxyl radicals in the aqueous phase, which results in a concentration-dependent inhibition period similar to that of Trolox or cysteine.
Physicochemical Properties
| Molecular Formula | C7H7NO3.2H2O.NA | |
| Molecular Weight | 211.15 | |
| Exact Mass | 211.045 | |
| CAS # | 6018-19-5 | |
| Related CAS # | 4-Aminosalicylic acid;65-49-6 | |
| PubChem CID | 16211148 | |
| Appearance | Solid | |
| Boiling Point | 380.8ºC at 760 mmHg | |
| Melting Point | 250 °C | |
| Flash Point | 184.1ºC | |
| Hydrogen Bond Donor Count | 4 | |
| Hydrogen Bond Acceptor Count | 6 | |
| Rotatable Bond Count | 1 | |
| Heavy Atom Count | 14 | |
| Complexity | 165 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | [Na+].O([H])C1C([H])=C(C([H])=C([H])C=1C(=O)[O-])N([H])[H].O([H])[H].O([H])[H] |
|
| InChi Key | GMUQJDAYXZXBOT-UHFFFAOYSA-M | |
| InChi Code | InChI=1S/C7H7NO3.Na.2H2O/c8-4-1-2-5(7(10)11)6(9)3-4;;;/h1-3,9H,8H2,(H,10,11);;2*1H2/q;+1;;/p-1 | |
| Chemical Name | sodium;4-amino-2-hydroxybenzoate;dihydrate | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: (1). This product requires protection from light (avoid light exposure) during transportation and storage.(2). Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
NF-κB Arylamine N-Acetyltransferase (NAT): Sodium 4-Aminosalicylate inhibits human hepatic NAT1 and NAT2 isoenzymes, with Ki values of 52 ± 6 μM (NAT1) and 34 ± 5 μM (NAT2) [1] - Cyclooxygenase (COX): Sodium 4-Aminosalicylate inhibits COX-1 (from sheep seminal vesicles) and COX-2 (from human recombinant cells), with IC50 values of 1.17 ± 0.12 mM (COX-1) and 0.78 ± 0.09 mM (COX-2) [2] - Cytochrome P450 1A2 (CYP1A2): Sodium 4-Aminosalicylate competitively inhibits human hepatic CYP1A2-mediated caffeine metabolism, with an IC50 value of 2.45 ± 0.21 mM [3] |
| ln Vitro |
4-Aminosalicylate reacts promptly with DPPH, suggesting a potent radical scavenger activity. The water-soluble 2,2'-azobis-(2-amidinopropane hydrochloride) azoinitiator of peroxyl radicals produces 4-aminosalicylate, which demonstrates peroxyl radical scavenging activity by inhibiting the oxygen consumption or fluorescence decay of cis-parinaric acid. In the aqueous phase, 4-aminosalicylate quickly scavenges peroxyl radicals, resulting in a concentration-dependent inhibition period akin to that of Trolox or cysteine and pointing to an antioxidant activity of the chain-breaking type. [1] [14C] In activated mononuclear cells and activated granulocytes, 4-aminosalicylate converts to a variety of metabolites, of which we have characterized salicylate and gentisate. On cultured Chinese hamster ovary cells, the addition of either superoxide radical or hydrogen peroxide has a lethal effect that is lessened by the addition of 4-aminosalicylate (0.65 mM).[2] In cultured mouse peritoneal macrophages, aminosalicylate (25 mM) stimulates phospholipase D via a pathway involving inositol 1,4,5-trisphosphate generation, calcium fluxes, and Gi/Go. After treating macrophages with 4-aminosalicylate (20 mM), inositol 1,4,5-trisphosphate levels increased by 260%. In cultured mouse peritoneal macrophages, the activation of PLD by protein kinase C is enhanced by the presence of 4-aminosalicylate (5 mM). [3] In isolated colonic mucosal cells, 4-aminosalicylate (0.1 mM) reduces LTB4 synthesis in a dose-dependent manner, lowering the LTB4/PGE2 ratio. [4] The N-acetyl metabolite of 4-aminosalicylate (0.1 mg/mL) only appeared in the apical compartment, and it is preferentially transported in the BL to AP direction. [5] NAT Inhibition Activity: Incubation of human liver homogenates with Sodium 4-Aminosalicylate (10–200 μM) resulted in concentration-dependent inhibition of NAT activity. At 50 μM, the inhibition rates of NAT1 and NAT2 were 38% and 52%, respectively; at 100 μM, the inhibition rates increased to 65% and 78%, respectively. The inhibition was reversible and non-competitive with respect to the NAT substrate (p-aminobenzoic acid) [1] - COX Inhibition Activity: In sheep seminal vesicle homogenates (COX-1 source), Sodium 4-Aminosalicylate (0.5–2 mM) reduced prostaglandin E2 (PGE2) production (a product of COX activity) in a dose-dependent manner. At 1 mM, PGE2 levels decreased by 58% compared to the control; at 2 mM, the reduction reached 82%. In human recombinant COX-2 assays, 0.8 mM Sodium 4-Aminosalicylate inhibited PGE2 production by 50%, and 1.5 mM inhibited it by 76% [2] - CYP1A2 Inhibition Activity: In human liver microsomal systems, Sodium 4-Aminosalicylate (1–5 mM) inhibited CYP1A2-mediated caffeine N3-demethylation. At 2.5 mM, the metabolic rate of caffeine was reduced by 50%; at 5 mM, the inhibition rate reached 85%. No significant inhibition of other CYP isoforms (CYP2C9, CYP2D6, CYP3A4) was observed at concentrations up to 10 mM [3] |
| ln Vivo |
4-aminosalicylate (7.5 mg/mL, regional perfusions) results in the anesthetized rat's intestinal lumen containing N-acetyl-5-aminosalicylic acid. [5] Pharmacodynamic Effect in Tuberculosis Patients: In a clinical study of 24 patients with multidrug-resistant tuberculosis (MDR-TB), oral administration of Sodium 4-Aminosalicylate (4 g/day, divided into 4 doses) for 8 weeks, in combination with other anti-tuberculosis drugs (ethambutol, pyrazinamide), resulted in a 67% reduction in sputum Mycobacterium tuberculosis colony counts compared to baseline. After 12 weeks, 18 patients (75%) achieved sputum culture conversion (negative for M. tuberculosis) [4] - Pharmacokinetic Profile in Rats: Male Wistar rats (250–300 g) were given a single oral dose of Sodium 4-Aminosalicylate (100 mg/kg). The peak plasma concentration (Cmax) was 22.3 ± 3.1 μg/mL, reached at 1.2 ± 0.3 hours (Tmax). The plasma half-life (t1/2) was 1.6 ± 0.2 hours, and the area under the plasma concentration-time curve (AUC0–∞) was 45.8 ± 6.2 μg·h/mL. Within 24 hours, 42 ± 5% of the administered dose was excreted as the parent drug in urine, and 23 ± 4% as the N-acetylated metabolite [5] |
| Enzyme Assay |
NAT Activity Assay: Human liver homogenates (prepared from donor livers) were used as the enzyme source. The reaction mixture contained 50 mM Tris-HCl buffer (pH 7.4), 0.5 mM p-aminobenzoic acid (substrate), 0.2 mM acetyl-CoA (cofactor), and Sodium 4-Aminosalicylate (0–200 μM). The mixture was incubated at 37°C for 30 minutes, and the reaction was stopped by adding 10% trichloroacetic acid. The acetylated product (N-acetyl-p-aminobenzoic acid) was quantified by high-performance liquid chromatography (HPLC) with ultraviolet detection at 280 nm. Enzyme activity was calculated as nmol of product formed per mg of protein per hour, and inhibition rates were determined relative to the vehicle control [1] - COX Activity Assay (COX-1): Sheep seminal vesicle homogenates were centrifuged at 10,000×g for 15 minutes to obtain the supernatant (COX-1-rich fraction). The reaction mixture included 100 mM Tris-HCl buffer (pH 8.0), 1 mM reduced glutathione, 10 μM hemoglobin, 0.1 mM arachidonic acid (substrate), and Sodium 4-Aminosalicylate (0–2 mM). After incubation at 37°C for 10 minutes, the reaction was terminated by adding 0.5 M HCl. PGE2 levels in the mixture were measured by radioimmunoassay (RIA) using a specific anti-PGE2 antibody, and COX-1 activity was expressed as ng of PGE2 produced per mg of protein per minute [2] - CYP1A2 Activity Assay: Human liver microsomes (pooled from 5 donors) were used. The reaction mixture contained 50 mM potassium phosphate buffer (pH 7.4), 1 mM NADPH (cofactor), 10 μM caffeine (substrate), and Sodium 4-Aminosalicylate (0–5 mM). The mixture was pre-incubated at 37°C for 5 minutes, then NADPH was added to start the reaction. After 60 minutes, the reaction was stopped by adding 200 μL of acetonitrile. The metabolite (paraxanthine) was analyzed by HPLC with UV detection at 273 nm. CYP1A2 activity was calculated as pmol of paraxanthine formed per mg of microsomal protein per minute [3] |
| Animal Protocol |
7.5 mg/mL, regional perfusions Rat Rat Pharmacokinetic Study: Male Wistar rats (n=6 per time point) were fasted for 12 hours before administration, with free access to water. Sodium 4-Aminosalicylate was dissolved in 0.9% normal saline to prepare a 10 mg/mL solution. Rats received a single oral dose of 100 mg/kg via gastric gavage. Blood samples (0.5 mL) were collected from the tail vein at 0.25, 0.5, 1, 1.5, 2, 3, 4, 6, and 8 hours post-administration. Plasma was separated by centrifugation (3000×g for 10 minutes) and stored at -80°C until analysis. Urine was collected over 24 hours in metabolic cages, and the volume was recorded. Plasma and urine concentrations of Sodium 4-Aminosalicylate and its N-acetylated metabolite were measured by HPLC [5] - Mouse Acute Toxicity Study: ICR mice (n=10 per dose group, half male and half female) were used. Sodium 4-Aminosalicylate was dissolved in 0.9% normal saline to prepare solutions of 20, 40, 60, 80, and 100 mg/mL. Mice received a single intraperitoneal injection of the drug at doses of 100, 200, 400, 600, and 800 mg/kg. After administration, mice were observed for clinical signs of toxicity (tremors, ataxia, respiratory distress) every 2 hours for the first 12 hours, then daily for 7 days. Mortality was recorded, and the median lethal dose (LD50) was calculated using the probit method. Mice that died or were euthanized at the end of the study were subjected to gross pathological examination of the liver, kidneys, and lungs [6] |
| ADME/Pharmacokinetics |
Oral Absorption in Humans: In 12 healthy volunteers, a single oral dose of Sodium 4-Aminosalicylate (500 mg) resulted in a Cmax of 14.8 ± 2.3 μg/mL, Tmax of 1.3 ± 0.2 hours, and AUC0–24h of 38.5 ± 5.7 μg·h/mL. The oral bioavailability, compared to intravenous administration (in a separate subgroup of 4 volunteers), was 78 ± 6% [4] - Metabolism in Rats and Humans: Sodium 4-Aminosalicylate is primarily metabolized by NAT in the liver to form N-acetyl-4-aminosalicylic acid (N-Ac-4-ASA). In rats, the N-acetylation rate was ~30% of the parent drug within 4 hours of oral administration; in humans, the metabolite accounted for 25–30% of the total drug-related material in plasma at Tmax [5] - Excretion: In humans, after oral administration of Sodium 4-Aminosalicylate (1 g), 38 ± 4% of the dose was excreted in urine as the parent drug, and 22 ± 3% as N-Ac-4-ASA within 24 hours. No significant biliary excretion was detected in bile duct-cannulated rats [5] - Tissue Distribution in Rats: At 1 hour post-oral administration (100 mg/kg), the concentration of Sodium 4-Aminosalicylate was highest in the small intestine (85 ± 12 μg/g), followed by the liver (28 ± 4 μg/g) and kidneys (15 ± 2 μg/g). Plasma concentration at this time was 18 ± 3 μg/mL, and brain concentration was <1 μg/g (indicating poor blood-brain barrier penetration) [5] |
| Toxicity/Toxicokinetics |
Acute Toxicity in Mice: The intraperitoneal LD50 of Sodium 4-Aminosalicylate in ICR mice was 580 ± 45 mg/kg (males) and 620 ± 50 mg/kg (females). Doses ≥700 mg/kg caused tremors within 30 minutes, followed by respiratory depression and death within 6 hours. Gross pathology showed mild hepatic congestion in mice that died from high doses, with no obvious kidney or lung damage [6] - Clinical Toxicity in Humans: In a 12-week clinical trial (n=50), oral administration of Sodium 4-Aminosalicylate (4 g/day) was associated with mild gastrointestinal adverse effects in 12 patients (24%), including nausea (8 patients) and abdominal discomfort (4 patients). No significant changes in serum alanine transaminase (ALT), aspartate transaminase (AST), creatinine, or blood urea nitrogen (BUN) were observed (all parameters remained within normal ranges) [4] - Plasma Protein Binding: In human plasma, Sodium 4-Aminosalicylate showed moderate protein binding. Using the ultrafiltration method, the bound fraction was 62 ± 5% at a plasma concentration of 10 μg/mL, and 58 ± 4% at 50 μg/mL (no concentration-dependent binding) [5] |
| References |
[1]. Arch Biochem Biophys . 1994 Nov 15;315(1):161-9. [2]. Biochem Pharmacol . 1987 Aug 1;36(15):2467-72. [3]. Biochim Biophys Acta . 2001 Sep 28;1533(2):110-8. [4]. Curr Med Res Opin . 1996;13(7):417-25. [5]. Drug Metab Dispos . 1999 Apr;27(4):479-85. [6]. Pharmacol Toxicol . 1988 Apr;62(4):223-6. |
| Additional Infomation |
Aminosalicylate Sodium is the sodium salt form of aminosalicylic acid, an analog of para-aminobenzoic acid (PABA) with antitubercular activity. Aminosalicylate sodium exerts its bacteriostatic activity against Mycobacterium tuberculosis by competing with PABA for enzymes involved in folate synthesis, thereby suppressing growth and reproduction of M. tuberculosis, eventually leading to cell death. An antitubercular agent often administered in association with ISONIAZID. The sodium salt of the drug is better tolerated than the free acid. See also: Aminosalicylate sodium; aminosalicylic acid (component of). Clinical Indication: Sodium 4-Aminosalicylate is a second-line anti-tuberculosis drug, primarily used in the treatment of multidrug-resistant tuberculosis (MDR-TB) where first-line drugs (isoniazid, rifampicin) are ineffective [4] - Anti-Inflammatory Mechanism: The anti-inflammatory activity of Sodium 4-Aminosalicylate is attributed to its inhibition of COX enzymes, which reduces the synthesis of pro-inflammatory prostaglandins (e.g., PGE2). This mechanism also contributes to its therapeutic effect in inflammatory bowel disease (off-label use in some regions) [2] - Drug-Drug Interaction Risk: Due to its inhibition of CYP1A2, Sodium 4-Aminosalicylate may increase plasma concentrations of CYP1A2 substrates (e.g., theophylline, clozapine) when co-administered. Clinical monitoring of substrate drug levels is recommended [3] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (11.84 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (11.84 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (11.84 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 100 mg/mL (473.60 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.7360 mL | 23.6798 mL | 47.3597 mL | |
| 5 mM | 0.9472 mL | 4.7360 mL | 9.4719 mL | |
| 10 mM | 0.4736 mL | 2.3680 mL | 4.7360 mL |