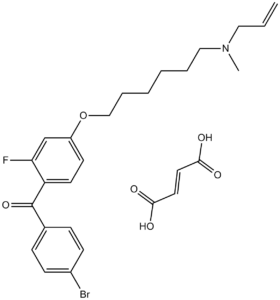
Ro 48-8071 fumarate is an orally bioavailable inhibitor of OSC (Oxidosqualene cyclase) with IC50 of ~6.5 nM. OSC represents a new and unique target for cholesterol-lowering drugs. In addition, Ro 48-8071 has shown anticancer activity, at 10 μM, it significantly reduces the viability of PC-3 prostate cancer cells, but not normal prostate cells. Ro 48-8071 (10-30 μM) induces apoptosis of both LNCaP and C4-2 cell lines in a dose-dependent manner. And castration-resistant PC-3 and DU145 cells also demonstrate significant levels of apoptosis following 24-hour treatment with Ro 48-8071.
Physicochemical Properties
| Molecular Formula | C27H31BRFNO6 | |
| Molecular Weight | 564.44 | |
| Exact Mass | 563.131 | |
| CAS # | 189197-69-1 | |
| Related CAS # | Ro 48-8071;161582-11-2 | |
| PubChem CID | 9959583 | |
| Appearance | White to light yellow solid powder | |
| Density | 1.2±0.1 g/cm3 | |
| Boiling Point | 522.8±50.0 °C at 760 mmHg | |
| Flash Point | 270.0±30.1 °C | |
| Vapour Pressure | 0.0±1.4 mmHg at 25°C | |
| Index of Refraction | 1.550 | |
| LogP | 6.24 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 8 | |
| Rotatable Bond Count | 14 | |
| Heavy Atom Count | 36 | |
| Complexity | 587 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | CN(CCCCCCOC1=CC(=C(C=C1)C(=O)C2=CC=C(C=C2)Br)F)CC=C.C(=C/C(=O)O)\C(=O)O |
|
| InChi Key | XCYAYLWZCRGKDS-WLHGVMLRSA-N | |
| InChi Code | InChI=1S/C23H27BrFNO2.C4H4O4/c1-3-14-26(2)15-6-4-5-7-16-28-20-12-13-21(22(25)17-20)23(27)18-8-10-19(24)11-9-18;5-3(6)1-2-4(7)8/h3,8-13,17H,1,4-7,14-16H2,2H3;1-2H,(H,5,6)(H,7,8)/b;2-1+ | |
| Chemical Name | (4-bromophenyl)-[2-fluoro-4-[6-[methyl(prop-2-enyl)amino]hexoxy]phenyl]methanone;(E)-but-2-enedioic acid | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Ro 48-8071 fumarate targets 2,3-oxidosqualene:lanosterol cyclase (OSC, E.C. 5.4.99.7, also named lanosterol synthase/intramolecular transferases) [1] Ro 48-8071 fumarate exerts anti-cancer effects via modulating estrogen receptor α (ERα) and estrogen receptor β (ERβ) expression in breast cancer cells, and androgen receptor (AR) expression in prostate cancer cells; [2] Ro 48-8071 fumarate modulates ERα/ERβ expression in hormone-dependent breast cancer cells, [3] Ro 48-8071 fumarate selectively inhibits intestinal OSC activity to suppress cholesterol synthesis [4] |
| ln Vitro |
With an IC50 value of roughly 1.5 nM, Ro 48 -8071 decreases cholesterol synthesis in HepG2 cells in a dose-dependent manner [1]. The viability of PC-3 prostate cancer cells is significantly reduced by Ro 48 -8071 (10 μM), but not of normal prostate cells. In LNCaP and C4-2 cell lines, Ro 48 -8071 (10-30 μM) causes apoptosis in a dose-dependent manner. Significant levels of apoptosis were also seen in castration-resistant PC-3 and DU145 cells 24 hours after they were treated with Ro 48–8071. The reduction of AR protein expression is dose-dependent and occurs with Ro 48 -8071 (10-25 μM). In castration-resistant PC-3 cells and hormone-dependent LNCaP, Ro 48 -8071 (0.1-1 μM) dose-dependently upregulates the expression of the ERβ protein [2]. Mammalian cells engineered to express human ERα or ERβ proteins in combination with the ER-responsive luciferase promoter, enables Ro 48-8071 to dose-dependently inhibit 17β-estradiol (E2)-induced ERα-responsive luciferase. Activity (IC50, approximately 10 μM), in a cell-nontoxic environment [3]. 1. Ro 48-8071 fumarate blocked human liver OSC activity and cholesterol synthesis in HepG2 cells in the nanomolar range; it triggered the production of monooxidosqualene, dioxidosqualene, and epoxycholesterol in HepG2 cells [1] 2. Ro 48-8071 fumarate reduced viability of hormone-dependent (LNCaP) and castration-resistant (PC-3, DU145) prostate cancer cell lines in a concentration-dependent manner (evaluated by SRB assay): LNCaP cells (7×10³/well) in 20% FBS RPMI-1640 were treated with RO in 10% FBS RPMI-1640 for 24/48 h; PC-3/DU145 cells (4×10³/well) in 10% FBS RPMI-1640 were treated with RO in 5% FBS RPMI-1640 for 24/48 h, with significant reduction in cell viability (P<0.05); RO had no effect on viability of normal human prostate RWPE-1 cells (5×10³/well) in complete growth medium treated with RO for 24 h (SRB assay, P>0.05) [2] 3. Low-dose (nM range) Ro 48-8071 fumarate treatment for 7 days reduced viability of LNCaP (8×10⁴/well, 10% FBS RPMI-1640) and PC-3 (4×10⁴/well, 5% FBS RPMI-1640) prostate cancer cells (SRB assay, P<0.05), with RO retreated every 48 h [2] 4. Ro 48-8071 fumarate induced apoptosis in hormone-dependent (LNCaP, C4-2) and castration-resistant (PC-3, DU145) prostate cancer cells: LNCaP (4×10⁵/well, 20% FBS RPMI-1640) and C4-2 (3×10⁵/well, 10% FBS RPMI-1640) cells were treated with RO (μM range) in 10%/5% FBS RPMI-1640 for 24 h; PC-3/DU145 (3×10⁵/well, 10% FBS RPMI-1640) cells were treated with RO (μM range) for 24 h; apoptotic/dead cells were quantified by Annexin V-FITC–based FACS analysis (P<0.05) [2] 5. Ro 48-8071 fumarate reduced AR protein expression in LNCaP prostate cancer cells (70% confluency, 20% FBS RPMI-1640) treated with 10/25 μM RO for 6 h or 0.1–1.0 μM RO for 7 days (Western blot); it increased ERβ protein expression in LNCaP (20% FBS RPMI-1640) and PC-3 (10% FBS RPMI-1640) cells treated with 10/25 μM RO for 6 h (Western blot) [2] 6. Combining Ro 48-8071 fumarate (15 μM, 2 h pretreatment + 22 h treatment) with ERβ agonist DPN (100 nM, 22 h treatment) potentiated the reduction of PC-3 cell viability (SRB assay, P<0.001); DPN alone (100 nM, 22 h) also reduced PC-3 viability (P=0.041) [2] 7. Ro 48-8071 fumarate potently reduced viability of ER-positive human breast cancer cells (BT-474, MCF-7, T47-D) in a concentration-dependent manner (pharmacological doses, 48 h treatment, SRB assay, P<0.05); low-dose (nM range) RO treatment for 7 days also reduced breast cancer cell viability (SRB assay, P<0.05); RO had no effect on viability of normal human mammary cells (AG11132A) treated with pharmacological doses for 24 h (SRB assay, P>0.05) [3] 8. Ro 48-8071 fumarate induced apoptosis and cell death in BT-474 and MCF-7 breast cancer cells: cells (1.5×10⁵/well, 10% FBS DMEM:F12) were treated with 5/10/20 μM RO for 24 h; apoptotic (Annexin V-positive) and dead (Annexin V-positive/PI-positive) cells were quantified by FACS analysis (10,000 cells/sample, P<0.05) [3] 9. Ro 48-8071 fumarate suppressed Estradiol (E2)-induced proliferation of breast cancer cells: cells were treated with 10 nM E2 ± 1/5/10 μM RO or 1 μM ICI 182,780 for 24 h in 5% charcoal stripped serum (SRB assay, P<0.05 vs E2 alone) [3] 10. Ro 48-8071 fumarate degraded ERα and induced ERβ expression in breast cancer cells: BT-474/MCF-7/T47-D cells (5% FBS DMEM:F12) were treated with 1/5/10/25 μM RO for 3/6/12 h (Western blot); ERβ inhibition/knockdown prevented RO-dependent loss of breast cancer cell viability (SRB assay, P<0.001) [3] 11. Combining Ro 48-8071 fumarate (10 μM, 48 h) with ERβ agonist DPN (1 μM) potentiated reduction of BT-474 cell viability (SRB assay, P<0.001); combining RO (10 μM, 24 h) with ERβ antagonist PHTPP (10/100 nM) reversed RO-induced loss of BT-474 viability (SRB assay, P<0.001) [3] |
| ln Vivo |
In hamsters, Ro 48 -8071 decreases LDL-C by approximately 60% at 150 μmol/kg per day and stops at 300 μmol/kg per day; HDL-C does not change at any dose. The amount of MOS in hamster liver increases at Ro 48 -8071 (≥00 μMol/kg per day). In hamsters, Ro 48 -8071 (300 μmol/kg daily) dramatically lowers VLDL secretion [1]. Without causing weight loss, Ro 48 -8071 (5 or 20 mg/kg) dramatically slowed the growth of tumors in mice. Furthermore, two of the twelve tumors observed in mice during the test period were totally eradicated by Ro 48-8071 at a dosage of 20 mg/kg [2]. In the entire small intestine of BALB/c mice, Ro 48-8071 (20 mg/day/kg body weight) rapidly and persistently inhibits (>50%) the synthesis of cholesterol. Additionally, the stomach and large intestine produce less cholesterol [4]. 1. Ro 48-8071 fumarate was safe at pharmacologically active doses in hamsters, squirrel monkeys, and Göttingen minipigs; it lowered LDL cholesterol by approximately 60% in hamsters, and at least 30% in squirrel monkeys and minipigs, with efficacy comparable to safe doses of simvastatin; hepatic monooxidosqualene increased dose-dependently after RO treatment (up to ~20 μg/g wet liver, <1% of hepatic cholesterol) and was inversely correlated with LDL levels [1] 2. Ro 48-8071 fumarate did not reduce coenzyme Q10 levels in liver and heart of hamsters, and did not trigger overexpression of hepatic HMG-CoA reductase, squalene synthase, or OSC; in contrast, simvastatin stimulated these enzymes and reduced coenzyme Q10 levels in liver and heart [1] 3. Ro 48-8071 fumarate effectively suppressed growth of aggressive castration-resistant PC-3 prostate cancer cell xenografts in male athymic nu/nu nude mice: PC-3 cells (5×10⁶ in 0.15 mL, matrigel:RPMI-1640=1:1 v/v) were injected subcutaneously into both flanks of 6-week-old mice; when tumors reached ~100 mm³, RO was administered by tail vein injection (5/20 mg/kg/d for 5 days as loading dose, then same dose every other day for 6 additional treatments, final injection 2 h before sacrifice); tumor volume was significantly reduced (P<0.05), with 2 out of 12 tumors in the 20 mg/kg group completely eradicated; no significant changes in animal weight were observed [2] 4. Ro 48-8071 fumarate prevented growth of BT-474 breast cancer xenografts in nude mice: 6-week-old nude mice received 1.7 mg/60-days estradiol slow-release pellets sc 48 h prior to injection of BT-474 cells (5×10⁶, Matrigel:DMEM/F12=4:1 v/v) into both flanks; when tumors reached ~100 mm³, RO was administered iv (5/10 mg/kg/d for 5 days, then every other day for 5 additional treatments, final injection 2 h before sacrifice); tumor growth was significantly inhibited (P<0.05), with no apparent toxicity (no weight loss); RO reduced ERα and increased ERβ staining in tumor tissues (immunohistochemistry, P<0.05) [3] 5. Ro 48-8071 fumarate (20 mg/day/kg bw, fed in chow diet for 7 days) induced rapid and sustained inhibition (>50%) of cholesterol synthesis in the whole small intestine of BALB/c mice; sterol synthesis was also reduced in the large intestine and stomach; hepatic cholesterol synthesis was markedly suppressed initially but rebounded to above baseline within 7 days; whole body cholesterol synthesis, fractional cholesterol absorption, and fecal neutral/acidic sterol excretion were not consistently changed [4] 6. Ro 48-8071 fumarate (20 mg/day/kg bw, 7 days) reduced sterol synthesis in liver, small intestine, large intestine, stomach of female BALB/c mice (measured by [³H] water incorporation into sterols); no significant changes in whole animal sterol synthesis (10 days treatment) were observed [4] 7. Ro 48-8071 fumarate (20 mg/day/kg bw, 10 days) reduced intestinal cholesterol synthesis in LDLR-deficient (ldlr−/−) mice and wild-type (ldlr+/+) controls (129/Sv background), with no significant genotype-dependent differences in intestinal/hepatic cholesterol concentration or sterol synthesis [4] 8. Ro 48-8071 fumarate (20 mg/day/kg bw, 18 days) had no significant effect on hepatic/plasma cholesterol concentrations in BALB/c mice fed a high cholesterol diet (1.0% w/w cholesterol); it did not alter mRNA levels of bile acid synthesis-related genes in liver [4] |
| Enzyme Assay |
1. OSC activity assay (human liver/ HepG2 cells): Cells/liver homogenates were incubated with OSC substrates (2,3-oxidosqualene) in the presence/absence of Ro 48-8071 fumarate (nanomolar range); cholesterol synthesis was measured by quantifying cholesterol production, and OSC activity was assessed by measuring lanosterol synthesis; the production of monooxidosqualene, dioxidosqualene, and epoxycholesterol was quantified to evaluate the downstream effects of OSC inhibition [1] 2. Cholesterol synthesis assay (HepG2 cells): HepG2 cells were treated with Ro 48-8071 fumarate (nanomolar range); intracellular cholesterol levels were quantified, and the production of oxidosqualene metabolites (monooxidosqualene, dioxidosqualene, epoxycholesterol) was measured via chromatographic methods to confirm OSC inhibition [1] 3. Sterol synthesis assay (mouse/hamster tissues): Tissue homogenates (small intestine, liver, large intestine, stomach) from animals treated with Ro 48-8071 fumarate were incubated with [³H] water; the incorporation of [³H] water into sterols was measured (nmol/h/g tissue) to assess de novo cholesterol synthesis and OSC activity; whole animal sterol synthesis was measured as umol of [³H] water incorporated into sterols/h/100 g bw [4] |
| Cell Assay |
1. Prostate cancer cell viability assay: LNCaP (7×10³/well) were seeded in 96-well plates with 20% FBS RPMI-1640 overnight; PC-3/DU145 (4×10³/well) were seeded with 10% FBS RPMI-1640 overnight; cells were washed with FBS-free medium, then treated with varying concentrations of Ro 48-8071 fumarate in 10% FBS RPMI-1640 (LNCaP) or 5% FBS RPMI-1640 (PC-3/DU145) for 24/48 h; cell viability was evaluated by SRB assay, with absorbance measured to quantify cell proliferation [2] 2. Normal prostate cell viability assay: RWPE-1 cells (5×10³/well) were seeded in 96-well plates with complete growth medium overnight; PC-3 cells (4×10⁴/well) were seeded with 10% FBS RPMI-1640 overnight; cells were washed with FBS-free medium, then treated with varying concentrations of Ro 48-8071 fumarate in complete growth medium (RWPE-1) or 5% FBS RPMI-1640 (PC-3) for 24 h; cell viability was assessed by SRB assay [2] 3. Long-term prostate cancer cell viability assay: LNCaP (8×10⁴/well) were seeded in 6-well plates with 20% FBS RPMI-1640 overnight; PC-3 (4×10⁴/well) were seeded with 10% FBS RPMI-1640 overnight; cells were washed with FBS-free medium, then treated with low-dose (nM) Ro 48-8071 fumarate in 10% FBS RPMI-1640 (LNCaP) or 5% FBS RPMI-1640 (PC-3) for 7 days (RO retreated every 48 h); cell viability was measured by SRB assay [2] 4. Prostate cancer cell apoptosis assay: LNCaP (4×10⁵/well) were seeded in 6-well plates with 20% FBS RPMI-1640 overnight; C4-2 (3×10⁵/well) were seeded with 10% FBS RPMI-1640 overnight; PC-3/DU145 (3×10⁵/well) were seeded with 10% FBS RPMI-1640 overnight; cells were washed with FBS-free medium, then treated with μM concentrations of Ro 48-8071 fumarate for 24 h; cells were harvested, stained with Annexin V-FITC and PI, and apoptotic/dead cells were quantified by FACS analysis (10,000 cells per sample) [2] 5. AR/ERβ expression assay (prostate cancer cells): LNCaP cells (70% confluency, 20% FBS RPMI-1640) were treated with 10/25 μM Ro 48-8071 fumarate for 6 h or 0.1–1.0 μM RO for 7 days; PC-3 cells (70% confluency, 10% FBS RPMI-1640) were treated with 10/25 μM RO for 6 h; whole-cell extracts were prepared, and AR/ERβ protein expression was analyzed by Western blotting with specific antibodies (β-actin as loading control) [2] 6. Breast cancer cell viability assay: BT-474/MCF-7/T47-D breast cancer cells were seeded in 96-well plates and treated with pharmacological/nM concentrations of Ro 48-8071 fumarate for 24/48/7 days (media change every 48 h for 7-day treatment); normal mammary cells (AG11132A) were treated with pharmacological doses of RO for 24 h; cell viability was evaluated by SRB assay, with absorbance measured to quantify cell proliferation [3] 7. Breast cancer cell apoptosis assay: BT-474/MCF-7 cells (1.5×10⁵/well) were seeded in 6-well plates with 10% FBS DMEM:F12 overnight; cells were washed with FBS-free medium, treated with 5/10/20 μM Ro 48-8071 fumarate for 24 h, harvested, stained with Annexin V-FITC and PI, and apoptotic/dead cells were quantified by FACS analysis (10,000 cells per sample) [3] 8. ERα/ERβ expression assay (breast cancer cells): BT-474/MCF-7/T47-D cells (70% confluency, 5% FBS DMEM:F12) were treated with 1/5/10/25 μM Ro 48-8071 fumarate for 3/6/12 h; whole-cell extracts were prepared, and ERα/ERβ protein expression was analyzed by Western blotting (β-actin as loading control); for ERβ knockdown assay, T47-D cells were transfected with 30/60 nM ERβ siRNA or scrambled siRNA for 72 h, then treated with 10 μM RO for 48 h, and cell viability was measured by SRB assay [3] 9. E2-induced breast cancer cell proliferation assay: Breast cancer cells were seeded in 96-well plates with 5% charcoal stripped serum, treated with 10 nM E2 ± 1/5/10 μM Ro 48-8071 fumarate or 1 μM ICI 182,780 for 24 h; cell viability was evaluated by SRB assay to assess the suppression of E2-induced proliferation [3] |
| Animal Protocol |
150, 300 μmol/kg BALB/c mice 1. Hamster/squirrel monkey/minipig cholesterol-lowering assay: Ro 48-8071 fumarate was administered to hamsters, squirrel monkeys, and Göttingen minipigs at pharmacologically active doses (dose not specified); simvastatin was administered as a control (doses >30 μmol/kg/day caused hepatotoxicity in hamsters); plasma LDL cholesterol levels were measured at regular intervals; hepatic monooxidosqualene levels and coenzyme Q10 levels (liver/heart) were quantified; hepatic gene expression (HMG-CoA reductase, squalene synthase, OSC) was analyzed [1] 2. Prostate cancer xenograft assay (nude mice): 6-week-old male athymic nu/nu nude mice were used; PC-3 cells (5×10⁶) were mixed with matrigel and RPMI-1640 (1:1 v/v) to a final volume of 0.15 mL, then injected subcutaneously into both flanks; when tumor volume reached ~100 mm³, Ro 48-8071 fumarate was administered by tail vein injection: 5/20 mg/kg/day for 5 days (loading dose), followed by the same dose every other day for 6 additional treatments, with a final injection 2 h before sacrifice; control mice received vehicle alone; tumor volume and animal weight were monitored throughout the experiment; tumors were harvested and photographed at the end [2] 3. Breast cancer xenograft assay (nude mice): 6-week-old nude mice received a 1.7 mg/60-days estradiol slow-release pellet via subcutaneous implantation 48 h before tumor cell injection; BT-474 cells (5×10⁶) were mixed with Matrigel and DMEM/F12 (4:1 v/v), then injected subcutaneously into both flanks; when tumor volume reached ~100 mm³, Ro 48-8071 fumarate was administered via tail vein injection: 5/10 mg/kg/day for 5 days, then every other day for 5 additional treatments, with a final injection 2 h before sacrifice; control mice received PBS alone; tumor volume and animal weight were monitored; tumors were harvested for immunohistochemistry (ERα/ERβ staining) [3] 4. Mouse intestinal cholesterol synthesis assay (BALB/c mice): Female/male BALB/c mice (7–16 weeks old) were fed a rodent chow diet containing Ro 48-8071 fumarate at doses of 5/15/20 mg/day/kg bw for 12 h to 18 days; simvastatin (20/200 mg/day/kg bw) was used as a control for 0.5–7 days; sterol synthesis rates in small intestine, liver, large intestine, stomach, and other organs were measured via [³H] water incorporation assay; intestinal histology (H&E staining) and Ki67 immunochemistry were performed; mRNA expression of intestinal cholesterol regulation genes (NPC1L1, CYP3A11, CES2A) was analyzed [4] 5. LDLR-deficient mouse assay (ldlr−/− mice): Female ldlr−/− mice and ldlr+/+ controls (21–25 weeks old, 129/Sv background) were fed a rodent chow diet containing Ro 48-8071 fumarate (20 mg/day/kg bw) for 10 days; intestinal/hepatic cholesterol concentration and sterol synthesis rates were measured; plasma cholesterol levels were quantified [4] 6. High cholesterol diet mouse assay: Female BALB/c mice (10–16 weeks old) were fed a rodent chow diet (0.02% w/w inherent cholesterol) supplemented with 1.0% w/w cholesterol, with/without Ro 48-8071 fumarate (20 mg/day/kg bw) for 18 days; hepatic/plasma cholesterol concentrations and mRNA levels of bile acid synthesis genes were measured; fecal neutral/acidic sterol excretion was quantified [4] 7. Ezetimibe comparison assay (BALB/c mice): Female BALB/c mice (7–10 weeks old) were fed a rodent chow diet containing Ro 48-8071 fumarate or ezetimibe (20 mg/day/kg bw) for 7–10 days; cholesterol absorption, fecal neutral/acidic sterol excretion, intestinal weight, unesterified cholesterol concentration, and intestinal gene expression (NPC1L1, CYP3A11, CES2A) were measured [4] |
| Toxicity/Toxicokinetics |
1. Ro 48-8071 fumarate was safe at pharmacologically active doses in hamsters, squirrel monkeys, and Göttingen minipigs; no hepatotoxicity was observed at effective doses, while simvastatin caused hepatotoxicity in hamsters at doses >30 μmol/kg/day [1] 2. Ro 48-8071 fumarate did not reduce coenzyme Q10 levels in liver and heart of hamsters, indicating no mitochondrial toxicity related to coenzyme Q10 depletion [1] 3. Ro 48-8071 fumarate showed no toxicity to athymic nu/nu nude mice bearing PC-3 prostate cancer xenografts: no significant changes in animal weight were observed during treatment (5/20 mg/kg via tail vein injection); 2 out of 12 tumors in the 20 mg/kg group were completely eradicated without apparent side effects [2] 4. Ro 48-8071 fumarate had no apparent toxicity to nude mice bearing BT-474 breast cancer xenografts: no significant changes in animal weight were observed during treatment (5/10 mg/kg via tail vein injection) [3] 5. Ro 48-8071 fumarate (20 mg/day/kg bw, 16 days) had no discernible effects on intestinal histology (H&E staining) and Ki67 expression (proliferation index) in BALB/c mice; mRNA expression of intestinal proliferatiopoptosis-related genes was not significantly altered [4] |
| References |
[1]. Ro 48-8.071, a new 2,3-oxidosqualene:lanosterol cyclase inhibitor lowering plasma cholesterol in hamsters, squirrel monkeys, and minipigs: comparison to simvastatin. J Lipid Res. 1997 Feb;38(2):373-90. [2]. Cholesterol biosynthesis inhibitor RO 48-8071 suppresses growth of hormone-dependent and castration-resistant prostate cancer cells. Onco Targets Ther. 2016 May 30;9:3223-32. [3]. Cholesterol biosynthesis inhibitors as potent novel anti-cancer agents: suppression of hormone-dependent breast cancer by the oxidosqualene cyclase inhibitor RO 48-8071. Breast Cancer Res Treat. 2014 Jul;146(1):51-62. [4]. Sustained and selective suppression of intestinal cholesterol synthesis by Ro 48-8071, an inhibitor of 2,3-oxidosqualene:lanosterol cyclase, in the BALB/c mouse. Biochem Pharmacol. 2014 Apr 1;88(3):351-63. |
| Additional Infomation |
Ro 48-8071 fumarate is a fumarate salt obtained by combining Ro 48-8071 with one molar equivalent of fumaric acid. An inhibitor of lanosterol synthase. It has a role as an EC 5.4.99.7 (lanosterol synthase) inhibitor and an antineoplastic agent. It contains a fumarate(1-) and a Ro 48-8071(1+). 1. 2,3-Oxidosqualene:lanosterol cyclase (OSC) is a unique target for cholesterol-lowering drugs; partial OSC inhibition reduces lanosterol/sterol synthesis and stimulates epoxysterol production, which represses HMG-CoA reductase expression, forming a synergistic negative regulatory loop [1] 2. Ro 48-8071 fumarate is a small-molecule OSC inhibitor with distinct pharmacological properties from statins (HMG-CoA reductase inhibitors): statins stimulate hepatic HMG-CoA reductase/squalene synthase/OSC expression and reduce coenzyme Q10 levels, while RO does not [1] 3. Cholesterol is an essential structural/functional component of cell membranes and a precursor of endogenous steroid hormones, making the cholesterol biosynthetic pathway an attractive target for endocrine-dependent cancers [2] 4. Ro 48-8071 fumarate is the first OSC inhibitor shown to suppress growth of hormone-dependent and castration-resistant prostate cancer cells; combining RO with ERβ agonists enhances its anti-prostate cancer efficacy [2] 5. Ro 48-8071 fumarate exerts anti-breast cancer effects partly via an off-target effect that increases the ERβ/ERα ratio in breast cancer cells; statins (Fluvastatin, Simvastatin) are less effective in reducing breast cancer cell viability and do not induce ERβ [3] 6. Ro 48-8071 fumarate selectively suppresses intestinal cholesterol synthesis in BALB/c mice, with hepatic cholesterol synthesis rebounding to baseline within 7 days; it does not consistently alter whole body cholesterol synthesis or fractional cholesterol absorption [4] 7. Ro 48-8071 fumarate upregulates mRNA levels of PXR target genes (CYP3A11, CES2A) in mouse small intestine, with no significant changes in mRNA expression of other intestinal cholesterol regulation genes (e.g., NPC1L1) [4] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.43 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.43 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (4.43 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 12.5 mg/mL (22.15 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication (<60°C). (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7717 mL | 8.8583 mL | 17.7167 mL | |
| 5 mM | 0.3543 mL | 1.7717 mL | 3.5433 mL | |
| 10 mM | 0.1772 mL | 0.8858 mL | 1.7717 mL |