MMP-9-IN-1 is a novel, potent and selective matrix metalloproteinase-9 (MMP-9) inhibitor that works by lowering cell migration and proliferation to stop tumor growth and metastasis. It was found using an in silico docking method, and biological and biochemical methods were used to assess it. MMP-9-IN-1 only attaches itself to the PEX domain of MMP-9; it does not attach to other MMPs. The disruption of MMP-9 homodimerization and subsequent blockade of a downstream signaling pathway necessary for MMP-9-mediated cell migration are the outcomes of this interaction between MMP-9-IN-1 and the PEX domain. In an MDA-MB-435 tumor xenograft model, MMP-9-IN-1 suppressed lung metastasis and slowed tumor growth.
Physicochemical Properties
| Molecular Formula | C16H17N3O3F2S |
| Molecular Weight | 369.386 |
| Exact Mass | 369.095 |
| Elemental Analysis | C, 52.03; H, 4.64; F, 10.29; N, 11.38; O, 12.99; S, 8.68 |
| CAS # | 502887-71-0 |
| Related CAS # | 502887-71-0 |
| PubChem CID | 135415473 |
| Appearance | White to off-white solid powder |
| LogP | 3.2 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 25 |
| Complexity | 547 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CCCC1=CC(=O)NC(=N1)SCC(=O)NC2=CC=C(C=C2)OC(F)F |
| InChi Key | OLTRRVUORWPRGF-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C16H17F2N3O3S/c1-2-3-11-8-13(22)21-16(20-11)25-9-14(23)19-10-4-6-12(7-5-10)24-15(17)18/h4-8,15H,2-3,9H2,1H3,(H,19,23)(H,20,21,22) |
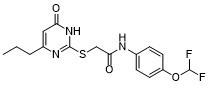
| Chemical Name | N-[4-(difluoromethoxy)phenyl]-2-[(6-oxo-4-propyl-1H-pyrimidin-2-yl)sulfanyl]acetamide |
| Synonyms | MMP9-IN-1; MMP-9-IN-1; MMP-9-IN1; OUN87710; OUN 87710; OUN-87710 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | MMP-9/matrix metalloproteinase-9 (Kd = 2.1 μM) | ||||||||||||||||||||||
| ln Vitro |
MMP-9-IN-1 (compound 2; 100 μM; 14 hours) does not cause notable cytotoxicity[1]. MMP-9-IN-1 (compound 2; 10 μM) significantly prevents MDA-MB-435 and HT-1080 cells from proliferating[1]. Inhibition of MMP-9-induced cell migration by the identified compounds [1] COS-1 cells expressing MMP-9 cDNA, or GFP cDNA as a control, were pre-incubated with or without the compounds (doses ranging from 100 nM to 100 μM) for 30 minutes, and examined by a Transwell chamber migration assay. Compounds 1, 2, 3 and 5 inhibited the migration of MMP-9-expressing COS-1 cells, whereas compound 4 showed no activity (Fig. 3A–E). Compounds 3 and 5, but not 1 and 2/MMP-9-IN-1, inhibited the migration of control cells (GFP-transfected) as well as MMP-9 transfected cells (Fig. 3C & E). To rule out the possibility that the reduction of cell migration by these compounds was due to cytotoxicity, a cell viability assay was performed. COS-1 cells were treated with the compounds for 24 hours followed by a cytotoxicity assay. Thapsigargin, an ER stress inducer that inhibits intracellular Ca2+-ATPases, was used as a positive control to trigger cell death. Treatment with compounds 1, 2 and 4 did not cause notable cytotoxicity at the maximum concentration used, whereas treatment with compounds 3 and 5 induced cell death (Fig. 3F). Compounds 3 and 5 were therefore excluded from further evaluation. The lethal dose (LD50) of compounds 1 and 2/MMP-9-IN-1 was determined in COS-1 cells. Cells were treated with increasing doses of the compounds for 24 hours followed by cell viability assay (Suppl. Fig. 1). The LD50 of compounds 1 and 2 were 360 ± 2 μM and 3.5 ± 0.3 mM, respectively, suggesting that their inhibition of MMP-9-induced cell migration was not due to cytotoxicity. To further determine the specificity and selectivity of compound 1 and 2/MMP-9-IN-1 for MMP-9-induced cell migration, we examined the effect of compounds 1 and 2 on cell migration induced by other MMPs in which the PEX domain has been reported to play a critical role in enhanced cell migration, e.g., MMP-2 and MT1-MMP (MMP-14). In contrast to MMP-9 expressing cells, neither compound inhibited the migration of MMP-2 or MT1-MMP ectopically expressing COS-1 cells (Fig. 4A). In addition, compound 2 did not interfere with MT1-MMP-mediated cancer cell invasion examined by a 3D invasion assay (Suppl. Fig. 2). Thus, small synthetic compounds that potentially bind to the PEX domain of MMP-9, inhibit MMP-9-induced cell migration with enhanced-specificity and -selectivity. Inhibition of migration in cancer cells that produce endogenous MMP-9 by compounds 1 and 2/MMP-9-IN-1 [1] We next investigated whether compounds 1 and 2 inhibit migration of cells producing a pathologically relevant level of endogenous MMP-9. Two human invasive cancer cell lines, HT-1080 and MDA-MB-435, expressing high endogenous levels of MMP-9 were employed. Treatment of the cells with compounds 1 and 2 significantly reduced cell migratory abilities. Furthermore, both compounds inhibited the migration of HT-1080 and MDA-MB-435 cells in a dose-dependent manner (Fig. 4B & C). Cell migration is a critical determinant of cancer cell invasiveness. Therefore, HT-1080 cells were assessed in the 3D type I collagen invasion assay. As anticipated, the cell invasive ability of HT-1080 cells was significantly inhibited in cells treated with compounds 1 and 2 (Fig. 4D & E). Inhibition of MDA-MB-435 cell invasion was also observed (data not shown). These data suggest that inhibition of MMP-9-mediated cell migration by the compounds results in suppressed cancer cell invasion. Compounds 1 and 2/MMP-9-IN-1 do not affect MMP-9 expression or proteolytic activity [1] Cell lysates from HT-1080 cells treated with and without compounds 1 and 2 were examined for MMP-9 expression levels by Western blot using an anti-MMP-9 antibody. Western blotting using an antibody to tubulin was employed as a control. No effect on MMP-9 expression by the compounds was observed (Fig. 5A). Activated MMP-9 was obtained by incubating purified proMMP-9 with p-aminophenyl mercuric acetate (APMA). Addition of compounds 1 and 2 to APMA-activated MMP-9 did not inhibit the catalytic activity of MMP-9 as measured by cleavage of the fluorescent Mca-P-L-G-L-Dpa-A-R-NH2 peptide (Fig. 5B). These data suggest that inhibition of MMP-9-induced cell migration by compounds 1 and 2 is not due to inhibition of MMP-9 expression or proteolytic activity. Binding of compound 2/MMP-9-IN-1 to the MMP-9 PEX domain [1] We titrated the binding of compound 2 to MMP-9 by monitoring MMP-9 tryptophan fluorescence. Saturation of purified proMMP-9 with compound 2 resulted in a 7 nm blue shift in the λmax of MMP-9 emission (Fig. 5C). No effect on the protein fluorescence occurred in the buffer only control. The Kd for MMP-9 binding to compound 2 is 2.1 ± 0.2 μM. To further characterize the binding between compound 2 and MMP-9, we employed a previously generated chimera of MMP-9 in which the PEX domain of MMP-9 was replaced with that of MMP-2 (MMP-9/MMP-2PEX). Upon addition of compound 2 to MMP-9/MMP-2PEX, no shift in fluorescence was detected (Fig. 5D). Likewise, compound 2 did not bind to purified recombinant soluble MT1-MMP. These data confirmed that compound 2 binds specifically to the PEX domain of MMP-9. The absorption of compound 1 at 280 nm precluded evaluation of its binding properties. To test whether compound 2 interferes with proMMP-9 homodimerization, co-immunoprecipitation of COS-1 cells transfected with both proMMP-9/Myc and proMMP-9/HA cDNAs in the presence or absence of compounds 2 and 4 was utilized. Treatment of the transfected cells with compound 2, but not inactive compound 4, resulted in blocked MMP-9 homodimer formation (Fig. 5E). This defect was not due to inhibition of expression of MMP-9 by compound 2 as evidenced by Western blotting of the cell lysate from HT-1080 cells (Fig. 5A). Similar results were obtained in reciprocal co-immunoprecipitation assays (Fig. 5E). This experiment confirms that compound 2 specifically inhibits MMP-9 homodimerization Homodimerized MMP-9 interacts with cell surface adhesion molecule, CD44, which leads to activation of EGFR and downstream MAPK (ERK1/2) pathway. To explore this network, the activity status of downstream effector ERK1/2 was examined. COS-1 cells ectopically expressing MMP-9 cDNA were serum starved in the presence or absence of compounds for 18 hours followed by Western blotting using anti-phospho-ERK1/2 and total ERK1/2 antibodies. As depicted in Fig. 5F, decreased activation of ERK1/2 was observed in compound 2 treated cells. Taken together, these data suggest that abrogation of MMP-9-mediated cell migration by compound 2 is due to disruption of MMP-9 homodimerization, which results in failure to cross-talk with CD44 and the EGFR-MAPK signaling pathway. Effect on MMP-9-mediated cell proliferation by compound 2/MMP-9-IN-1 [1] COS-1 cells transfected with MMP-9 or GFP cDNA (control) were monitored for cell proliferation in the absence or presence of compound 2/MMP-9-IN-1 or 4 with a CellTiter-Glo® Luminescent assay. In agreement with previous observations, the rate of cell proliferation increased significantly (P < 0.05) in COS-1 cells expressing MMP-9 as compared to GFP expressing COS-1 cells (Fig. 6A). MMP-9-induced cell proliferation was not affected by compound 4, consistent with its lack of effect on MMP-9-induced cell migration. In contrast, compound 2 significantly decreased MMP-9-induced cell proliferation (Fig. 6A), but did not affect the proliferation of COS-1 cells transfected with GFP cDNA (Fig. 6B). To determine if compound 2 also affects the proliferation of cancer cells producing endogenous MMP-9, HT-1080 and MDA-MB-435 cancer cells were treated with 10 μM compound 2. Significant inhibition of cell proliferation was observed for HT-1080 and MDA-MB-435 cells treated with compound 2, but not with compound 4 or with DMSO controls (Fig. 6C & D). |
||||||||||||||||||||||
| ln Vivo |
MMP-9-IN-1 (compound 2; 20 mg/kg; intraperitoneal and intratumoral injection alternately; 6 days/week; for 14 weeks) causes a significant tumor growth delay in NCR-Nu mice that have an MDA-MB-435/GFP tumor[1].
MMP-9-IN-1 inhibits cancer cell metastasis in vivo[1].
Decreased tumor growth and lung metastases in MMP-9-IN-1/compound 2-treated mice [1] MDA-MB-435 cells are highly metastatic in nude mice (30, 31) and produce high levels of MMP-9, thus serving as an appropriate experimental model to explore the in vivo inhibitory activity of compounds exhibiting anti-MMP-9 activity. To facilitate in vivo analysis of tissues and visualization of the lung metastases, MDA-MB-435 cells were stably transfected with GFP cDNA and implanted subcutaneously within the mammary fat pad of female immunodeficient mice. Treatment of mice with compound 2/MMP-9-IN-1 resulted in a profound delay in tumor growth, whereas treatment with the inactive control compound 4 or the vehicle alone failed to inhibit tumor growth (Fig. 7A & B). Tumor incidence was unaffected by compound 2. The lungs of tumor-bearing mice were removed and slices of the lungs (3 mm thickness) were examined under a fluorescent microscope (Fig. 7C). In the vehicle control and compound 4-treated groups, multiple large nodules were evident in MDA-MB-435/GFP tumor-bearing mice, whereas the extent of lung metastasis was dramatically reduced in mice treated with compound 2/MMP-9-IN-1 (Fig. 7C). Also, dimensions of tumor foci area in the lung and the percent of mice displaying lung metastases were significantly decreased in these mice (Fig. 7D & E). Thus, treatment with compound 2 impaired the in vivo effect of MMP-9 on both primary tumor growth and metastasis. No significant change in body weight nor other signs of toxicity during the 14-week period were observed in compound 2-treated mice. |
||||||||||||||||||||||
| Enzyme Assay |
Fluorogenic Assay of Enzyme Activity [1] Fluorogenic peptide substrate (50 μM) (18) was incubated with the compounds either in the presence or absence of latent MMP-9 and APMA-activated MMP-9 for 30 min at 25 °C before detection. Fluorescence emission at 393 nm with excitation at 328 nm was measured in a fluorescent plate reader. Fluorescence Spectroscopy [1] Binding of compound 2 to MMP-9 was assayed by observing the change of tryptophan emission upon binding. Purified recombinant MMP-9 (50 nM) or MMP-9/MMP-2PEX (50 nM) was diluted in buffer (50 mM Tris-HCl, 60 mM KCl and 0.05% Tween 20, pH 7.4) in the presence or absence of compound 2. As a control for protein stability and loss, an analogous buffer solution was added to the protein. The protein sample was excited at 280 nm and emission scans were collected from 290 to 400 nm, using slit widths of 0.3 nm on a QM-4/200SE spectrofluorimeter with double excitation and emission monochromators. Three emission scans were collected and averaged at each concentration. The Kd was determined using the Prism software package (GraphPad V5) to fit the data to equation (1). Fluorogenic Assay of Enzyme Activity [2] Mca-PLGL-Dpa-AR-NH2 fluorogenic peptide substrate (final concentration of 10 μM in DMSO) was incubated with the compounds and p-aminophenylmercuric acetate (APMA)-activated proMMP-9 for 30 min at 25 °C before detection. Fluorescence emission at 405 nm with excitation at 320 nm was measured in a fluorescent plate reader. |
||||||||||||||||||||||
| Cell Assay |
Cell Viability [1] Compound cytotoxicity was determined using the CellTiter-Glo™ Luminescent Cell Viability Assay .. 2.5 × 104 COS-1 cells were plated to a 96-well plate and incubated for 18 h with compounds 1–5. Luminescence was recorded using a SpectraMax Microplate Reader. LD50’s of the compounds were measured over a 100 pM to 10 mM concentration range. The LD50 was determined using the Prism software package (GraphPad V5) and fitting to equation (2). ΔL=(ΔLmax∗[ |
|