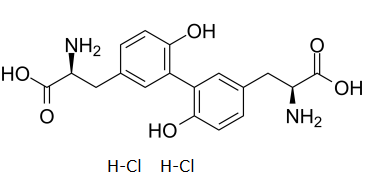
L,L-Dityrosine hydrochloride (o,o'-Dityrosine), a tyrosine dimer formed by 3,3'-biaryl bond formation, is used as a biomarker to detect oxidative protein damage and selective proteolysis. It is a component of acid hydrolysates of a number of biological materials, including the insect cuticular resilin.
Physicochemical Properties
| Molecular Formula | C18H20N2O6 |
| Molecular Weight | 360.361205101013 |
| Exact Mass | 217.050570 |
| CAS # | 221308-01-6 |
| Related CAS # | L,L-Dityrosine;63442-81-9 |
| Appearance | Light yellow to light brown solid |
| InChi Key | JJWFIVDAMOFNPS-QRPNPIFTSA-N |
| InChi Code | InChI=1S/C9H11NO3.ClH/c10-8(9(12)13)5-6-1-3-7(11)4-2-6;/h1-4,8,11H,5,10H2,(H,12,13);1H/t8-;/m0./s1 |
| Chemical Name | (2S)-2-amino-3-(4-hydroxyphenyl)propanoic acid;hydrochloride |
| Synonyms | o,o'-Dityrosine hydrochloride |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | The fluorescence of the rare amino acid LL-dityrosine, which is found in insoluble biological materials with structural features, was recently shown to decay non-exponentially (Kungl et al. (1992) J. Fluorescence 2, 63-74). Here we investigated the time-resolved fluorescence of a dityrosine-containing peptide (DCP) to study the influence of side chains on the fluorescence decay of the chromophore. The fluorescence decay of DCP was best fitted by three exponential terms including a sub-nanosecond rise term, the values of which are quite similar to the parameters obtained for the decay of free dityrosine. They were found to depend on the pH of the aqueous solution but not on the temperature. Analysis by an exponential series method revealed broad fluorescence lifetime distributions for DCP. Compared to the corresponding analysis of dityrosine transients, similar lifetime centers were found whereas the widths of the distributions were found broader for DCP. Molecular dyamics (MD) simulations of dityrosine at 300 K show that chi 1 and chi 2 side chain conformers (rotamers) of both tyrosine subunits interconvert on a picosecond timescale. The rates of interconversion were shown to depend critically upon the MD technique applied: in vacuo simulations yielded lower interconversion rates compared to stochastic dynamics (SD) and full MD (water explicitly included). However, MD simulations of the dityrosine-containing peptide revealed no interconversions of the chi 1 and chi 2 side chain rotamers of both tyrosine subunits within a 400 ps trajectory. Interconversions could be induced by raising the temperature of the system (DCP plus solvent) to 340 K. Side chain rotamers of dityrosine are not stable on a fluorescence time scale but are stable when a dityrosine-containing peptide is regarded. Nevertheless both molecules yield similar fluorescence decay patterns. We therefore conclude that the rotamer model proposed for the fluorescence decay of tyrosine and tryptophan cannot be applied to the fluorescence decay of dityrosine and peptides containing this chromophore. This should be of future interest when dityrosine is used as an intrinsic sensor to study complex dityrosine-containing macromolecules by fluorescence spectroscopy [1]. |
| ln Vivo | Mice treated with MPTP showed an increase in L,L-dityrosine hydrochloride (o,o'-dityrosine hydrochloride) in the ventral midbrain and striatum [2]. |
| Animal Protocol | Oxidative stress is implicated in the death of dopaminergic neurons in Parkinson's disease and in the 1-methyl-4-phenyl-1,2,3, 6-tetrahydropyridine (MPTP) model of Parkinson's disease. Oxidative species that might mediate this damage include hydroxyl radical, tyrosyl radical, or reactive nitrogen species such as peroxynitrite. In mice, we showed that MPTP markedly increased levels of o, o'-dityrosine and 3-nitrotyrosine in the striatum and midbrain but not in brain regions resistant to MPTP. These two stable compounds indicate that tyrosyl radical and reactive nitrogen species have attacked tyrosine residues. In contrast, MPTP failed to alter levels of ortho-tyrosine in any brain region we studied. This marker accumulates when hydroxyl radical oxidizes protein-bound phenylalanine residues. We also showed that treating whole-brain proteins with hydroxyl radical markedly increased levels of ortho-tyrosine in vitro. Under identical conditions, tyrosyl radical, produced by the heme protein myeloperoxidase, selectively increased levels of o,o'-dityrosine, whereas peroxynitrite increased levels of 3-nitrotyrosine and, to a lesser extent, of ortho-tyrosine. These in vivo and in vitro findings implicate reactive nitrogen species and tyrosyl radical in MPTP neurotoxicity but argue against a deleterious role for hydroxyl radical in this model. They also show that reactive nitrogen species and tyrosyl radical (and consequently protein oxidation) represent an early and previously unidentified biochemical event in MPTP-induced brain injury. This finding may be significant for understanding the pathogenesis of Parkinson's disease and developing neuroprotective therapies [2]. |
| References | [1]. Kungl AJ, et al. Molecular dynamics simulation of the rare amino acid LL-dityrosine and a dityrosine-containing peptide: comparison with time-resolved fluorescence. Biochim Biophys Acta. 1994 Dec 15;1201(3):345-52. |
Solubility Data
| Solubility (In Vitro) |
H2O : ~125 mg/mL (~288.50 mM) DMSO : ~62.5 mg/mL (~144.25 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7750 mL | 13.8750 mL | 27.7500 mL | |
| 5 mM | 0.5550 mL | 2.7750 mL | 5.5500 mL | |
| 10 mM | 0.2775 mL | 1.3875 mL | 2.7750 mL |