Physicochemical Properties
| Molecular Formula | C20H17F3N4NAO8P |
| Molecular Weight | 552.329726934433 |
| Exact Mass | 552.063 |
| CAS # | 1807365-35-0 |
| Related CAS # | Contezolid;1112968-42-9;Contezolid phosphoramidic acid;1807365-43-0;Contezolid acefosamil;1807497-11-5 |
| PubChem CID | 131750213 |
| Appearance | Off-white to light yellow solid powder |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 14 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 37 |
| Complexity | 967 |
| Defined Atom Stereocenter Count | 1 |
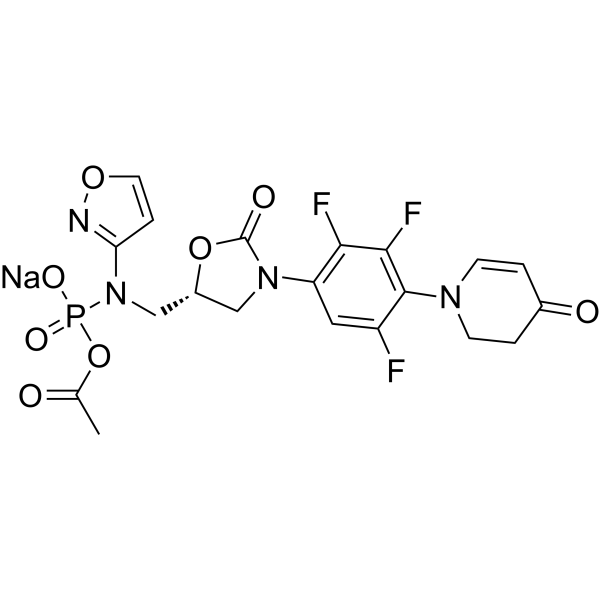
| SMILES | CC(=O)OP(=O)(N(C[C@H]1CN(C(=O)O1)C2=CC(=C(C(=C2F)F)N3CCC(=O)C=C3)F)C4=NOC=C4)[O-].[Na+] |
| InChi Key | JANNTEAGZXJITO-BTQNPOSSSA-M |
| InChi Code | InChI=1S/C20H18F3N4O8P.Na/c1-11(28)35-36(31,32)27(16-4-7-33-24-16)10-13-9-26(20(30)34-13)15-8-14(21)19(18(23)17(15)22)25-5-2-12(29)3-6-25;/h2,4-5,7-8,13H,3,6,9-10H2,1H3,(H,31,32);/q;+1/p-1/t13-;/m1./s1 |
| Chemical Name | sodium;acetyloxy-[1,2-oxazol-3-yl-[[(5R)-2-oxo-3-[2,3,5-trifluoro-4-(4-oxo-2,3-dihydropyridin-1-yl)phenyl]-1,3-oxazolidin-5-yl]methyl]amino]phosphinate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Contezolid (MRX-I) is extremely efficient against all clinical isolates of Gram-positive Staphylococcus, Streptococcus, and Enterococcus, including multidrug-resistant (MDR) pathogens including MRSA, PRSP, MRSA, and VRE[2]. |
| ln Vivo | Oral absorption of Contezolid (MRX-I) occurs rapidly in mice, rats, and dogs, with peak plasma concentrations seen 0.5−2.6 hours postdose. In mice, rats, and dogs, PK parameters were found as follows: dose-normalized Cmax/dose of 524, 1065, and 259 ng/mL/(mg/kg); dose-normalized AUC0−t/dose of 1654, 3703 and 1664 ng·h/mL/(mg/kg); T1/2 is 1, 1.5, and 3 hours; oral bioavailability is 69%, 109%, and 37% correspondingly [2]. Contezolid (MRX-I) showed no evident toxicity [2]. Contezolid (MRX-I, 100 mg/kg once daily) significantly reduced lung bacterial load compared with untreated early and late controls [3] |
| Animal Protocol |
Animal/Disease Models: balb/c (Bagg ALBino) mouse are intranasally infected with Mycobacterium tuberculosis Erdman[3]. Doses: 100, 50 (twice), 25 (twice) mg/kg. Route of Administration: intragastric (po) (po)administration, once or twice (two times) daily, five days a week for four weeks. Experimental Results: CFU recovered from the lungs was Dramatically lower (P < 0.05) compared with early and late control mice. MRX-I at 50 mg/kg and 25 mg/kg twice (two times) daily was Dramatically better than late-stage control mice (P < 0.05). MRX-I at 100 mg/kg one time/day was Dramatically better than 50 mg/kg and 25 mg/kg twice (two times) daily (P < 0.05). There was no statistical difference between 50 mg/kg MRX-I and 25 mg/kg twice (two times) daily (P > 0.05). Animal/Disease Models: Rat[2]. Doses: 20, 100 and 200/300 mg/kg/day. Doses: Take orally twice (two times) daily. Experimental Results: No deaths were observed. |
| References |
[1]. Evaluation of the Effect of Contezolid (MRX-I) on the Corrected QT Interval in a Randomized, Double-Blind, Placebo- and Positive-Controlled Crossover Study in Healthy Chinese Volunteers. Antimicrob Agents Chemother. 2020 May 21;64(6):e02158-19. [2]. New potent antibacterial oxazolidinone (MRX-I) with an improved class safety profile. J Med Chem. 2014 Jun 12;57(11):4487-97. [3]. In Vitro and In Vivo Activities of Contezolid (MRX-I) against Mycobacterium tuberculosis. Antimicrob Agents Chemother. 2018 Jul 27;62(8):e00493-18. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~180 mg/mL (~325.89 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 4.5 mg/mL (8.15 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 45.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 4.5 mg/mL (8.15 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 45.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 4.5 mg/mL (8.15 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 45.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8105 mL | 9.0526 mL | 18.1051 mL | |
| 5 mM | 0.3621 mL | 1.8105 mL | 3.6210 mL | |
| 10 mM | 0.1811 mL | 0.9053 mL | 1.8105 mL |