Physicochemical Properties
| Molecular Formula | C32H22O10 |
| Molecular Weight | 566.51 |
| Exact Mass | 566.121 |
| Elemental Analysis | C, 67.84; H, 3.91; O, 28.24 |
| CAS # | 548-19-6 |
| Related CAS # | 548-19-6 |
| PubChem CID | 5318569 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 843.6±65.0 °C at 760 mmHg |
| Melting Point | 355ºC |
| Flash Point | 280.2±27.8 °C |
| Vapour Pressure | 0.0±3.2 mmHg at 25°C |
| Index of Refraction | 1.714 |
| Source | Ginkgo biloba |
| LogP | 5.24 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 42 |
| Complexity | 1080 |
| Defined Atom Stereocenter Count | 0 |
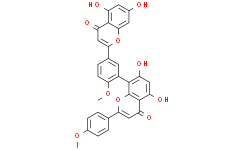
| SMILES | O1C(C2C([H])=C([H])C(=C([H])C=2[H])OC([H])([H])[H])=C([H])C(C2C(=C([H])C(=C(C1=2)C1=C(C([H])=C([H])C(C2=C([H])C(C3=C(C([H])=C(C([H])=C3O2)O[H])O[H])=O)=C1[H])OC([H])([H])[H])O[H])O[H])=O |
| InChi Key | HUOOMAOYXQFIDQ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C32H22O10/c1-39-18-6-3-15(4-7-18)26-14-24(38)31-22(36)12-21(35)29(32(31)42-26)19-9-16(5-8-25(19)40-2)27-13-23(37)30-20(34)10-17(33)11-28(30)41-27/h3-14,33-36H,1-2H3 |
| Chemical Name | 8-[5-(5,7-dihydroxy-4-oxochromen-2-yl)-2-methoxyphenyl]-5,7-dihydroxy-2-(4-methoxyphenyl)chromen-4-one |
| Synonyms | IGG; iso-ginkgetin; 4',4'''-Dimethylamentoflavone; 8-[5-(5,7-dihydroxy-4-oxochromen-2-yl)-2-methoxyphenyl]-5,7-dihydroxy-2-(4-methoxyphenyl)chromen-4-one; CHEBI:79087; 8-(5-(5,7-dihydroxy-4-oxo-4H-chromen-2-yl)-2-methoxyphenyl)-5,7-dihydroxy-2-(4-methoxyphenyl)-4H-chromen-4-one; 8-[5-(5,7-dihydroxy-4-oxo-4H-chromen-2-yl)-2-methoxyphenyl]-5,7-dihydroxy-2-(4-methoxyphenyl)-4H-chromen-4-one; Isoginkgetin |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | 20S proteasome; Pre-mRNA splicing; Natural biflavonoid; antioxidative; anti-inflammatory; anti-viral; anti-tumor |
| ln Vitro |
Isoginkgetin (0-20 μM, 24 h) inhibits cell invasion in a dose-dependent manner[1]. Isoginkgetin (10 μM, 0-24 h) activates autophagy, causes lysosomal swelling, repositioning, and acidification, and causes the accumulation of ubiquitinated protein cargo into perinuclear aggregates[2]. Isoginkgetin (10 μM, 0–6 h) induces ER stress, degrades the unfolded protein response, and causes an accumulation of ERAD substrates[2]. Isoginkgetin interferes with NF-κB signaling and directly inhibits the chymotrypsin, trypsin, and caspase-like activities of the 20S proteasome[2]. In MM cell lines, isoginkgetin (30 μM, 0-24 h) induces apoptosis[2]. |
| ln Vivo |
Isoginkgetin attenuated LPS-induced depression-like behavior in several tests such as “Open field” test, EPM test, and Tail suspension test.The post hoc test revealed that isoginkgetin significantly reversed LPS-induced hypoactivity in terms of total distance traveled (p < 0.01) and rearing numbers (p < 0.05)), whereas the reduction in the number of entries into the central zone was not significantly improved. The interaction between LPS and isoginkgetin was significantly different in terms of the number of entries into the open arms (F1, 33 = 5.836, p < 0.05). The post hoc test revealed that isoginkgetin significantly reversed LPS-induced decrease in the number of entries into the open arms (p < 0.05) and the time spent on open arms (p < 0.01). Meanwhile, there is no significant difference between isoginkgetin treatment alone and control group. The post hoc test further revealed that LPS significantly increased the immobility time (p < 0.001), whereas isoginkgetin treatment reversed this change (p < 0.001).The post hoc test revealed that isoginkgetin significantly reversed LPS-increased IL-1β level. Meanwhile, a slightly but significantly increased IL-10 level was found in LPS group, which was not attenuated by isoginkgetin treatment.[3] Isoginkgetin (4 mg/kg; i.p.; daily for 14 days) shows anti-inflammatory effects[3]. |
| Enzyme Assay |
Akt Kinase Assay [1] Cells were transfected with hemagglutinin-tagged Akt plasmids and lysed in a buffer solution containing 20 mmol/L Tris-HCl (pH 7.5), 12 mmol/L β-glycerophosphate, 150 mmol/L NaCl, 5 mmol/L EGTA, 10 mmol/L NaF, 1% Triton X-100, 0.5% deoxycholate, 3 mmol/L DTT, 1 mmol/L sodium orthovanadate, 1 mmol/L phenylmethylsulfonyl fluoride, and 1.5% aprotinin. Cell extracts were centrifuged, and 200 μg supernatants were immunoprecipitated with hemagglutinin antibody and protein A-agarose. The beads were washed three times with a solution containing 20 mmol/L Tris-HCl (pH 7.5), 150 mmol/L NaCl, 5 mmol/L EGTA, 2 mmol/L DTT, and 1 mmol/L phenylmethylsulfonyl fluoride. Akt activities were assayed in a reaction mixture consisting of 1 μg histone H2B (in vitro substrate for Akt), 2 μg PKI, 5 μmol/L ATP, and 5 μCi [γ-32P]ATP in a buffer [20 mmol/L HEPES (pH 7.2), 10 mmol/L MgCl2, 10 mmol/L MnCl2, 1 mmol/L DTT, and 0.2 mmol/L EGTA] at 30°C for 20 minutes. Protein kinase assays were terminated by the addition of SDS sample buffer, and the samples were subjected to SDS-PAGE. Phosphorylated proteins were visualized by autoradiography. MMP-9 Activity Assay [1] MMP-9 activity was measured using fluorogenic MMP-9 substrate [N-(2,4-dinitrophenyl)-Pro-Leu-Gly-Leu-Trp-Ala-D-Arg amide; Sigma]. Briefly, conditioned medium was mixed with substrate and incubated at 37°C. Fluorescence was measured by using a fluorometer, according to the instruction from the manufacturer. |
| Cell Assay |
Cell Death Assays [1] Cells were washed once with medium, once with PBS, once with Annexin V–FITC buffer [10 mmol/L HEPES-NaOH (pH 7.4), 140 mmol/L NaCl, 2.5 mmol/L CaCl2], and then incubated for 15 minutes at room temperature with 5 μg/mL Annexin V–FITC. After washing once with Annexin V buffer, the samples were resuspended in the same buffer. Immediately before analysis, 5 μg/mL propidium iodide (Biosource International) were added to distinguish apoptotic cells from necrotic cells, and the cells were analyzed using a flow cytometer. |
| Animal Protocol |
Adult male Kunming mice (age 8–10weeks, 30–50 g)[3] 4 mg/kg Daily for 14 days by intraperitoneally (i.p.) prior to LPS (0.83mg/kg) administration. Methods: Adult Kunming mice were intraperitoneally injected vehicle or isoginkgetin (4 mg/kg) daily for 14 days before saline or LPS (0.83 mg/kg) administration. Depression-like behavior, neurotransmitter levels, and markers of neuroinflammation were determined. Isoginkgetin effect on LPS-induced microglial activation was then assessed in BV2 cells. Finally, conditioned medium (CM) derived from isoginkgetin-treated BV2 cells was co-cultured with SH-SY5Y cells for 24 h. Cell viability and apoptosis were evaluated.[3] |
| References |
[1]. Isoginkgetin inhibits tumor cell invasion by regulating phosphatidylinositol 3-kinase/Akt-dependent matrix metalloproteinase-9 expression. Mol Cancer Ther. 2006 Nov;5(11):2666-75. [2]. Isoginkgetin, a Natural Biflavonoid Proteasome Inhibitor, Sensitizes Cancer Cells to Apoptosis via Disruption of Lysosomal Homeostasis and Impaired Protein Clearance. Mol Cell Biol. 2019 Apr 30;39(10):e00489-18. [3]. Isoginkgetin treatment attenuated lipopolysaccharide-induced monoamine neurotransmitter deficiency and depression-like behaviors through downregulating p38/NF-κB signaling pathway and suppressing microglia-induced apoptosis. J Psychopharmacol. 2021 Oct;35(10):1285-1299. |
| Additional Infomation |
Isoginkgetin is a biflavonoid resulting from the formal oxidative dimerisation between position 8 of one molecule of 5,7-dihydroxy-4'-methoxyflavone and the 3' position of another. Found in the leaves of Ginkgo biloba, it is a potent inhibitor of matrix metalloproteinase 9 (MMP-9). It has a role as an EC 3.4.24.35 (gelatinase B) inhibitor, an antineoplastic agent and a plant metabolite. It is a biflavonoid and an aromatic ether. It is functionally related to a 5,7-dihydroxy-4'-methoxyflavone. Isoginkgetin has been reported in Podocarpus neriifolius, Sequoiadendron giganteum, and other organisms with data available. |
Solubility Data
| Solubility (In Vitro) | DMSO: ~100 mg/mL (~176.5 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (3.67 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.08 mg/mL (3.67 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: 1.67 mg/mL (2.95 mM) in 50% PEG300 + 50% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7652 mL | 8.8260 mL | 17.6519 mL | |
| 5 mM | 0.3530 mL | 1.7652 mL | 3.5304 mL | |
| 10 mM | 0.1765 mL | 0.8826 mL | 1.7652 mL |