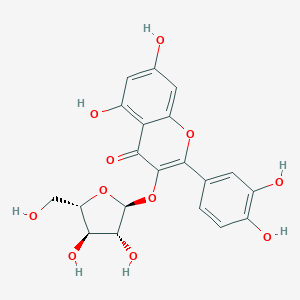
Avicularin is a naturally occurring flavonoid isolated from plants, with anti-inflammatory, anti-allergic, anti-oxidant, hepatoprotective, and anti-tumor activities. controls the activities of PPAR-γ, COX-2, and NF-κB (p65).
Physicochemical Properties
| Molecular Formula | C₂₀H₁₈O₁₁ |
| Molecular Weight | 434.35 |
| Exact Mass | 434.084 |
| Elemental Analysis | C, 55.31; H, 4.18; O, 40.52 |
| CAS # | 572-30-5 |
| Related CAS # | 572-30-5 |
| PubChem CID | 5490064 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.9±0.1 g/cm3 |
| Boiling Point | 855.4±65.0 °C at 760 mmHg |
| Melting Point | 240-243ºC |
| Flash Point | 305.8±27.8 °C |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.802 |
| LogP | 2.06 |
| Hydrogen Bond Donor Count | 7 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 31 |
| Complexity | 711 |
| Defined Atom Stereocenter Count | 4 |
| SMILES | O1[C@]([H])([C@@]([H])([C@]([H])([C@]1([H])C([H])([H])O[H])O[H])O[H])OC1C(C2=C(C([H])=C(C([H])=C2OC=1C1C([H])=C([H])C(=C(C=1[H])O[H])O[H])O[H])O[H])=O |
| InChi Key | BDCDNTVZSILEOY-UXYNSRGZSA-N |
| InChi Code | InChI=1S/C20H18O11/c21-6-13-15(26)17(28)20(30-13)31-19-16(27)14-11(25)4-8(22)5-12(14)29-18(19)7-1-2-9(23)10(24)3-7/h1-5,13,15,17,20-26,28H,6H2/t13-,15-,17+,20-/m0/s1 |
| Chemical Name | 3-[(2S,3R,4R,5S)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]oxy-2-(3,4-dihydroxyphenyl)-5,7-dihydroxychromen-4-one |
| Synonyms | Quercetin; 3-O-α-L-arabinofuranoside; Avicularin |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
GLUT4; PPARγ; COX-2 - ERK (No IC50/Ki/EC50 data available) [1] - NF‑κB, COX‑2, PPAR‑γ (No IC50/Ki/EC50 data available) [2] - C/EBPα, GLUT4 (No IC50/Ki/EC50 data available) [3] - TRAF6, MAPK (No IC50/Ki/EC50 data available) [4] - Amyloid Beta (Aβ)-related targets (No IC50/Ki/EC50 data available) [5] |
| ln Vitro |
In LPS-stimulated RAW 264.7 cells, avicuLarin (10-300 μM, 1 hour) suppresses the generation of NO and PGE2 [1]. In LPS-stimulated RAW 264.7 cells, AvicuLarin (10-300 μM, 1 h) suppresses ERK signaling. In Huh7 cells, AvicuLarin (25-100 μg/mL, 48 h) decreases apex, proliferation, and cell migration [2]. By reporting NF, AvicuLarin (25-100 μg/mL, 48 h) demonstrates anti-inflammatory efficacy [1]. AvicuLarin (50 μM, 6 days) decreases 3T3-L1 cells, PPARγ, and C/EBPα in addition to intracellular toxins. -κB (p65) causes an increase in PPAR-γ and COX-2 expression as well as cell disinfection [2]. 3T3-L1 cells' quantitative dose-mediated GLUT4-mediated inhibition is inhibited by avicuLarin (50 μM, 6 days) [3]. AvicuLarin (2.5-10 μM, 2 hours) increases the amounts of aP2 mRNA in both human formulations and solutions [3]. - In lipopolysaccharide (LPS)-stimulated RAW 264.7 macrophages, Avicularin inhibited the inflammatory response by suppressing ERK phosphorylation. It reduced nitric oxide (NO) production, decreased the levels of pro-inflammatory cytokines (TNF-α, IL-6), and downregulated the expression of inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2) [1] - In human hepatocellular carcinoma (HCC) cells, Avicularin exerted anti-tumor effects via regulating NF‑κB/COX‑2/PPAR‑γ activities. It inhibited cell proliferation, induced cell apoptosis, and suppressed cell migration and invasion [2] - In 3T3-L1 preadipocytes, Avicularin suppressed lipid accumulation by repressing C/EBPα-activated GLUT4-mediated glucose uptake. It downregulated the expression of adipogenic transcription factors (C/EBPα, PPARγ) and lipid-related proteins (aP2, FAS) [3] - In interleukin-1β (IL-1β)-treated chondrocytes, Avicularin inhibited cartilage extracellular matrix (ECM) degradation and inflammation by suppressing TRAF6/MAPK activation. It reduced the expression of matrix metalloproteinases (MMP-1, MMP-3, MMP-13) and ADAMTS-4, while upregulating the expression of type II collagen and aggrecan [4] - In amyloid Beta (Aβ)-induced PC12 cells (an Alzheimer's disease cell model), Avicularin attenuated Aβ-induced neurotoxicity. It increased cell viability, reduced reactive oxygen species (ROS) production, and inhibited apoptotic signaling pathway activation (decreased Bax/Bcl-2 ratio, suppressed caspase-3 activation) [5] |
| ln Vivo |
AvicuLarin, an intra-articular injection administered twice weekly for four weeks at a dose of 0.5-2 mg/kg, inhibits the progression of stent OA (osteoarthritis) caused by ACLT [4]. AvicuLarin (Laterolateralis, 50 and 100 mg/kg for 21 days) impairs memory in deposits of Alzheimer's disease caused by amyloid beta [5]. - In rats with Aβ-induced Alzheimer's disease, Avicularin attenuated memory impairment. It improved learning and memory abilities (evaluated by Morris water maze test), reduced Aβ deposition in the hippocampus, inhibited neuroinflammation (decreased TNF-α, IL-1β levels), and alleviated oxidative stress (increased SOD activity, decreased MDA level) in brain tissues [5] |
| Cell Assay |
Cell proliferation assay [2] Cell Types: Huh7 cells Tested Concentrations: 25, 50, 100 μg/mL Incubation Duration: 12, 24, 36 and 48 h Experimental Results: Inhibition of cell proliferation was dose-dependent. Western Blot Analysis [1] Cell Types: RAW 264.7 Cell Tested Concentrations: 10, 30, 100, 300 μM Incubation Duration: 1 h Experimental Results: Inhibited LPS-induced iNOS and COX-2 protein expression, released pro-inflammatory cytokines IL-1β, Cytoplasmic IκB degradation and ERK phosphorylation. RT-PCR[3] Cell Types: 3T3-L1 Cell Tested Concentrations: 50 μM Incubation Duration: 6 days Experimental Results: PPARγ, C/EBPα and aP2 mRNA levels were diminished by approximately 28.7%, 69.5 and 18.3% respectively. - For RAW 264.7 macrophage experiment: Cells were seeded in culture plates and stimulated with LPS (1 μg/mL) in the presence or absence of different concentrations of Avicularin. After 24 h of incubation, NO production was detected by Griess reagent, pro-inflammatory cytokines (TNF-α, IL-6) were measured by ELISA, and the expression of iNOS, COX-2, and phosphorylated ERK (p-ERK) was analyzed by western blot [1] - For human HCC cell experiment: HCC cells were treated with various concentrations of Avicularin for 24-72 h. Cell proliferation was detected by MTT assay, cell apoptosis was analyzed by flow cytometry (Annexin V-FITC/PI staining), cell migration and invasion were evaluated by transwell assay, and the expression of NF‑κB, COX‑2, and PPAR‑γ was detected by western blot and PCR [2] - For 3T3-L1 adipocyte experiment: 3T3-L1 preadipocytes were induced to differentiate into adipocytes with Avicularin treatment. Lipid accumulation was detected by Oil Red O staining, glucose uptake was measured using 2-NBDG (a fluorescent glucose analog), and the expression of C/EBPα, GLUT4, PPARγ, aP2, and FAS was analyzed by western blot and PCR [3] - For chondrocyte experiment: Chondrocytes were treated with IL-1β (10 ng/mL) and different concentrations of Avicularin. After incubation, the expression of MMPs, ADAMTS-4, type II collagen, aggrecan, TRAF6, and phosphorylated MAPK (p-p38, p-JNK, p-ERK) was detected by western blot and PCR, and the secretion of inflammatory cytokines (IL-6, TNF-α) was measured by ELISA [4] - For PC12 cell experiment: PC12 cells were treated with Aβ (25-35, 20 μM) and Avicularin for 24 h. Cell viability was detected by MTT assay, ROS production was measured by DCFH-DA staining, and the expression of Bax, Bcl-2, and cleaved caspase-3 was analyzed by western blot [5] |
| Animal Protocol |
Animal/Disease Models: ACLT (anterior cruciate ligament transection) induced rats [4] Doses: 0.5, 1, 2mg/kg Route of Administration: Inject into the right knee joint cavity, twice a week for 4 weeks. Experimental Results: Reduce tibial subchondral osteolysis, reduce bone loss, and increase tibial subchondral bone mass. Attenuated ECM degradation and loss of aggrecan and type II collagen in ACLT-induced rats. diminished MMP3 and MMP13 protein levels. Animal/Disease Models: Rats with beta-amyloid-induced Alzheimer's disease [5] Doses: 25, 50 and 100 mg/kg Route of Administration: Orally for 21 days Experimental Results: Enhanced cognitive activity , reverses the effects of amyloid-induced inflammatory responses and excessive oxidative stress. - For Aβ-induced Alzheimer's disease rat model: Rats were intracerebroventricularly injected with Aβ (25-35, 10 μg/rat) to establish the model. After model establishment, Avicularin was administered intragastrically at doses of 10, 20, and 40 mg/kg/day for 21 consecutive days. Morris water maze test was performed to evaluate memory function, and brain tissues were collected to detect Aβ deposition (immunohistochemistry), inflammatory cytokines (ELISA), and oxidative stress markers (SOD activity, MDA level) [5] |
| References |
[1]. Avicularin Inhibits Lipopolysaccharide-Induced Inflammatory Response by Suppressing ERK Phosphorylation in RAW 264.7 Macrophages. Biomol Ther (Seoul). 2012 Nov;20(6):532-7. [2]. Avicularin ameliorates human hepatocellular carcinoma via the regulation of NF‑κB/COX‑2/PPAR‑γ activities. Mol Med Rep. 2019 Jun;19(6):5417-5423. [3]. Avicularin, a plant flavonoid, suppresses lipid accumulation through repression of C/EBPα-activated GLUT4-mediated glucose uptake in 3T3-L1 cells. J Agric Food Chem. 2013 May 29;61(21):5139-47. [4]. Avicularin suppresses cartilage extracellular matrix degradation and inflammation via TRAF6/MAPK activation. Phytomedicine. 2021 Oct;91:153657. [5]. Avicularin Attenuates Memory Impairment in Rats with Amyloid Beta-Induced Alzheimer's Disease. Neurotox Res. 2022 Feb;40(1):140-153. |
| Additional Infomation |
Avicularin is a quercetin O-glycoside in which an alpha-L-arabinofuranosyl residue is attached at position 3 of quercetin via a glycosidic linkage. It is isolated particularly from Juglans regia and Foeniculum vulgare. It has a role as a hepatoprotective agent and a plant metabolite. It is a monosaccharide derivative, an alpha-L-arabinofuranoside, a tetrahydroxyflavone and a quercetin O-glycoside. Avicularin has been reported in Camellia sinensis, Rhododendron dauricum, and other organisms with data available. - Avicularin is a natural plant flavonoid with potential anti-inflammatory, anti-tumor, anti-adipogenic, chondroprotective, and neuroprotective activities [1]-[5] - The anti-inflammatory effect of Avicularin in LPS-induced macrophages is mediated by suppressing ERK phosphorylation [1] - The anti-tumor effect of Avicularin in human HCC cells is associated with the regulation of NF‑κB/COX‑2/PPAR‑γ signaling pathway [2] - The inhibitory effect of Avicularin on lipid accumulation in 3T3-L1 cells is related to the repression of C/EBPα-activated GLUT4-mediated glucose uptake [3] - The chondroprotective effect of Avicularin in IL-1β-induced chondrocytes is achieved by inhibiting TRAF6/MAPK activation [4] - The neuroprotective effect of Avicularin in Aβ-induced Alzheimer's disease model is attributed to reducing Aβ deposition, inhibiting neuroinflammation, and alleviating oxidative stress [5] |
Solubility Data
| Solubility (In Vitro) | DMSO: ~100 mg/mL (~230.2 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.79 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.79 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3023 mL | 11.5115 mL | 23.0229 mL | |
| 5 mM | 0.4605 mL | 2.3023 mL | 4.6046 mL | |
| 10 mM | 0.2302 mL | 1.1511 mL | 2.3023 mL |