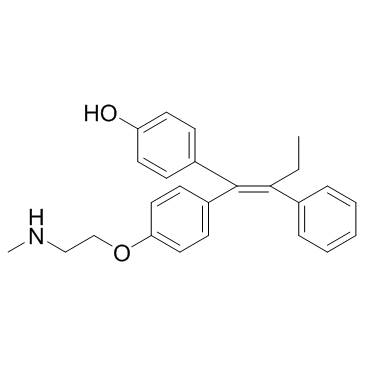
Endoxifen, an active metabolite of Tamoxifen, is a potent and selective antagonist of the estrogen receptor that has shown promise in treating patients who have not responded well to earlier hormonal treatments. In MCF-7 cells, doxifen exhibits anti-estrogenic properties and reduces E2-induced PR expression. Endoxifen also prevents the transcriptional activity of ER-alpha and stops the growth of breast cancer cells induced by estrogen. Endoxifen dramatically reduces the rate of cell division in MCF7, HS 578T, and BT-549 cells. In addition, compared to tamoxifen, edoxifen shows a four-fold greater inhibition of PKC activity.
Physicochemical Properties
| Molecular Formula | C₂₅H₂₇NO₂ |
| Molecular Weight | 373.48738 |
| Exact Mass | 373.204 |
| CAS # | 110025-28-0 |
| Related CAS # | Endoxifen (Z-isomer);112093-28-4;Endoxifen Z-isomer hydrochloride;1032008-74-4;Endoxifen hydrochloride;1197194-41-4;Endoxifen (E-isomer);114828-90-9;Endoxifen E-isomer hydrochloride;1197194-61-8;Endoxifen-d5;1185244-45-4 |
| PubChem CID | 10090750 |
| Appearance | White to light yellow solid |
| Density | 1.099g/cm3 |
| Boiling Point | 519.3ºC at 760mmHg |
| Flash Point | 267.9ºC |
| Vapour Pressure | 2.1E-11mmHg at 25°C |
| Index of Refraction | 1.598 |
| LogP | 5.75 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 28 |
| Complexity | 467 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CC/C(C1=CC=CC=C1)=C(C2=CC=C(O)C=C2)/C3=CC=C(OCCNC)C=C3 |
| InChi Key | MHJBZVSGOZTKRH-OCOZRVBESA-N |
| InChi Code | InChI=1S/C25H27NO2/c1-3-24(19-7-5-4-6-8-19)25(20-9-13-22(27)14-10-20)21-11-15-23(16-12-21)28-18-17-26-2/h4-16,26-27H,3,17-18H2,1-2H3/b25-24+ |
| Chemical Name | 4-[(E)-1-[4-[2-(methylamino)ethoxy]phenyl]-2-phenylbut-1-enyl]phenol |
| Synonyms | Endoxifen |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Aromatase; ERα |
| ln Vitro | Endoxifen, a hydroxylated Tamoxifen metabolite, is approximately 100-fold more effective than tamoxifen as an ER antagonist. It also implies that endoxifen, but not 4-hydroxytamoxifen, causes ER-alpha degradation in addition to its transcription-related effects on the ER[1]. Endoxifen is a strong antiestrogen that causes breast cancer cells' estrogen receptor α to degrade. Furthermore, research demonstrates that even in the presence of tamoxifen, N-desmethyl-tamoxifen, and 4-hydroxytamoxifen, endoxifen inhibits estrogen-induced breast cancer cell proliferation and blocks ERA transcriptional activity[2]. At 10 μM, doxifen significantly inhibits the growth of all breast cancer cell lines, with the exception of MDAMB-468, which experiences only moderate inhibition.At 10 μM concentration, cytotoxic effects on MCF7, HS 578T, and BT-549 cells are quite significant. While the inhibitory effects of Endoxifen at lower concentrations (0.01-1 μM) are not as strong as those of 10 μM, all tested cells die at concentrations of 100 μM[2]. |
| ln Vivo | Endoxifen taken orally is quickly absorbed and available throughout the body when tested in female rats. Rats treated with Endoxifen exhibit 1,500% higher Cmax and 787% higher exposure (AUC0–∞) levels of Endoxifen than Tamoxifen-treated rats. Oral Endoxifen at doses of 2, 4, and 8 mg/kg administered once daily for 28 days in a row is safe and causes the growth of human mammary tumor xenografts in female mice to be gradually inhibited[2]. |
| Animal Protocol | Mice: A subcutaneous (s.c.) implant of a 30-to 40-mg fragment of MCF-7 human mammary tumor from an in vivo passage is made in six-week-old female athymic NCr-nu/nu mice near the right flank. Day 0 is the date of the implantation of the tumor fragments. Each animal has a 0.72-mg 17 β-estradiol 60-day release pellet s.c. implanted in the back of their neck one day before the tumor fragment is inserted, in order to support the growth of the estrogen-dependent MCF-7 tumor. Day 13 following tumor fragment implantation, the day treatment was started, saw the growth of individual tumors to sizes ranging from 75 to 196 mm3. One control group (12 mice/group) and four treatment groups (6 mice/group) are created from a total of thirty-six tumor-bearing mice through randomization. On day 13 after the tumor was implanted, the patient was given oral gavage once a day for 28 days, along with three different doses of Endoxifen (2, 4, and 8 mg/kg) or Tamoxifen (10 mg/kg) twice a day, three hours apart. For every treatment group, the dosage volume of 0.2 mL/10 g body weight remains unchanged. Beginning on the first day of treatment, the s.c. tumors are measured and the animals are weighed twice a week. On Day 58, the research comes to an end. The overall delay in the growth of the median tumor is determined by taking the median time to reach two tumor mass doublings. Furthermore, an additional assessment of the antitumor efficacy is conducted on Days 41, 1 day after the last treatment, and 58, the day the study is terminated, by comparing the median tumor weight in the treatment groups to the median tumor weight in the control group (T/C 9 100%)[2]. |
| ADME/Pharmacokinetics |
Metabolism / Metabolites Endoxifen has known human metabolites that include Endoxifen O-glucuronide and Endoxifen O-sulfate. Endoxifen is a known human metabolite of 4-Hydroxytamoxifen and N-Desmethyltamoxifen. |
| References |
[1]. Tamoxifen, endoxifen, and CYP2D6: the rules for evaluating a predictive factor. Oncology (Williston Park). 2009 Dec;23(14):1233-4, 1236. [2]. The tamoxifen metabolite, endoxifen, is a potent antiestrogen that targets estrogen receptor alpha for degradation in breast cancer cells. Cancer Res. 2009 Mar 1;69(5):1722-7. |
| Additional Infomation | 4-Hydroxy-N-desmethyltamoxifen is a stilbenoid. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6774 mL | 13.3872 mL | 26.7745 mL | |
| 5 mM | 0.5355 mL | 2.6774 mL | 5.3549 mL | |
| 10 mM | 0.2677 mL | 1.3387 mL | 2.6774 mL |