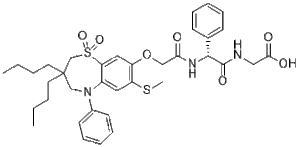
Elobixibat (also known as AZD-7806, AJG-533 and A 3309) is a novel, potent and first-in-class ileal bile acid transporter (IBAT) inhibitor with IC50 values of 0.53 ± 0.17 nM, 0.13 ± 0.03 nM, and 5.8 ± 1.6 nM for human IBAT, mouse IBAT, and canine IBAT. It is potentially useful for treatment of chronic idiopathic constipation (CIC; syn functional constipation). CIC affects up to 25% of the general population; and up to a half are unsatisfied with current therapies. There is an unmet need for safe and effective drugs to treat CIC. Elobixibat provides a novel approach to treat chronic constipation via IBAT inhibition with enhanced delivery of bile acids to the colon. Pharmacodynamic studies show that it accelerates colonic transit, increases stool frequency, loosens stool consistency and relieves constipation-related symptoms in CIC patients. These beneficial effects are maintained for a minimum of 8 consecutive weeks of treatment. With minimal absorption and low systemic bioavailability, elobixibat is generally well tolerated and may offer the added benefit of improving serum lipid profiles through bile acid depletion.
Physicochemical Properties
| Molecular Formula | C36H45N3O7S2 |
| Molecular Weight | 695.8884 |
| Exact Mass | 695.27 |
| Elemental Analysis | C, 62.14; H, 6.52; N, 6.04; O, 16.09; S, 9.21 |
| CAS # | 439087-18-0 |
| Related CAS # | Elobixibat hydrate;1633824-78-8; 439087-68-0 (S-isomer) |
| PubChem CID | 9939892 |
| Appearance | Typically exists as White to off-white solid at room temperature |
| LogP | 8.964 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 16 |
| Heavy Atom Count | 48 |
| Complexity | 1140 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | S1(C2=C([H])C(=C(C([H])=C2N(C2C([H])=C([H])C([H])=C([H])C=2[H])C([H])([H])C(C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H])(C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H])C1([H])[H])SC([H])([H])[H])OC([H])([H])C(N([H])[C@@]([H])(C(N([H])C([H])([H])C(=O)O[H])=O)C1C([H])=C([H])C([H])=C([H])C=1[H])=O)(=O)=O |
| InChi Key | XFLQIRAKKLNXRQ-UUWRZZSWSA-N |
| InChi Code | InChI=1S/C36H45N3O7S2/c1-4-6-18-36(19-7-5-2)24-39(27-16-12-9-13-17-27)28-20-30(47-3)29(21-31(28)48(44,45)25-36)46-23-32(40)38-34(26-14-10-8-11-15-26)35(43)37-22-33(41)42/h8-17,20-21,34H,4-7,18-19,22-25H2,1-3H3,(H,37,43)(H,38,40)(H,41,42)/t34-/m1/s1 |
| Chemical Name | (R)-(2-(2-((3,3-dibutyl-7-(methylthio)-1,1-dioxido-5-phenyl-2,3,4,5-tetrahydrobenzo[b][1,4]thiazepin-8-yl)oxy)acetamido)-2-phenylacetyl)glycine |
| Synonyms | AZD-7806; AZD 7806; AZD7806; A 3309; 439087-18-0; AZD7806; AZD-7806; A3309; 865UEK4EJC; A-3309; 2-[[(2R)-2-[[2-[(3,3-dibutyl-7-methylsulfanyl-1,1-dioxo-5-phenyl-2,4-dihydro-1lambda6,5-benzothiazepin-8-yl)oxy]acetyl]amino]-2-phenylacetyl]amino]acetic acid; A3309; A-3309; AJG-533 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Ileal bile acid transporter/IBAT (IC50 = 0.53 nM); IL-6; TNF-α/β Elobixibat reduces BA reabsorption in the terminal ileum, resulting in increased BA excretion in stool and higher BA concentration in the colon, which enhances the secretion of water and electrolytes into the colon, improves intestinal motility, and eases colonic transit.[2] Elobixibat (formerly A3309) is a first-in-class ileal bile acid transporter (IBAT) inhibitor for treatment of chronic idiopathic constipation (CIC; syn functional constipation). CIC affects up to 25% of the general population; and up to a half are unsatisfied with current therapies. There is an unmet need for safe and effective drugs to treat CIC.Elobixibat provides a novel approach to treat chronic constipation via IBAT inhibition with enhanced delivery of bile acids to the colon. [1] |
| ln Vitro |
Elobixibat reduces BA reabsorption in the terminal ileum, resulting in increased BA excretion in stool and higher BA concentration in the colon, which enhances the secretion of water and electrolytes into the colon, improves intestinal motility, and eases colonic transit.[2] Elobixibat (formerly A3309) is a first-in-class ileal bile acid transporter (IBAT) inhibitor for treatment of chronic idiopathic constipation (CIC; syn functional constipation). CIC affects up to 25% of the general population; and up to a half are unsatisfied with current therapies. There is an unmet need for safe and effective drugs to treat CIC.Elobixibat provides a novel approach to treat chronic constipation via IBAT inhibition with enhanced delivery of bile acids to the colon. [1] In a competitive inhibition assay using HEK-293 cells engineered to overexpress mammalian bile acid transporters, Elobixibat was shown to inhibit the intracellular accumulation of 30 µM radiolabeled glycocholic acid via the ileal bile acid transporter (IBAT) in a concentration-dependent manner. This confirmed its potent and selective inhibition of IBAT. [1] |
| ln Vivo |
Treatment with elobixibat reduced the serum BA and increased the fecal BA concentration, and ameliorated the liver inflammation and fibrosis. It also reduced the expression of proinflammatory cytokines in the liver and MLNs, and transforming growth factor-β expression in the liver. Finally, elobixibat normalized intestinal tight junction protein level and the composition of the intestinal microbiota.
Conclusion: elobixibat ameliorates NASH-related histopathology, reduces cytokine expression, and normalizes the intestinal microbial composition in MCD-fed mice, which suggests that it may represent a promising candidate for the therapy of NASH.[3] Both groups showed liver fat accumulation and fibrosis, with no significant differences between the two groups. However, mice with elobixibat showed fewer liver tumors. The total serum bile acid levels, including free, tauro-conjugated, glyco-conjugated, and tauro-α/β-muricholic acids in the liver, were noticeably reduced following elobixibattreatment. The proportion of gram-positive bacteria in feces was significantly lower in the group treated with elobixibat (5.4%) than in the group without elobixibat (33.7%). Conclusion: elobixibat suppressed tumor growth by inhibiting bile acid reabsorption, and decreasing total bile acid and primary bile acid levels in the serum and liver. Additionally, the presence of bile acids in the colon may have led to a significant reduction in the proportion of gram-positive bacteria, potentially resulting in decreased secondary bile acid synthesis.[2] Pharmacodynamic studies show that it accelerates colonic transit, increases stool frequency, loosens stool consistency and relieves constipation-related symptoms in CIC patients. These beneficial effects are maintained for a minimum of 8 consecutive weeks of treatment. With minimal absorption and low systemic bioavailability, elobixibat is generally well tolerated and may offer the added benefit of improving serum lipid profiles through bile acid depletion.[1] In a preclinical model of meat-induced constipation in healthy beagle dogs, oral administration of Elobixibat at doses of 1.5, 5, and 15 µmol/kg/day increased stool weight. The 5 and 15 µmol/kg/day doses resulted in an almost 3.5-fold increase in stool weight, which was statistically significant compared to vehicle. [1] In dogs, oral administration of Elobixibat at 5, 25, and 200 µmol/kg/day for 28 consecutive days increased hepatic bile acid synthesis in a dose-dependent manner, as measured by the plasma levels of the intermediate 7α-hydroxy-4-cholesten-3-one (C4). The increase was detectable as early as 24 hours after administration and was maintained throughout the 28-day period. [1] In a Phase I human study with 30 CIC patients, Elobixibat at doses of 3 and 10 mg daily for 14 days significantly accelerated colonic transit time (CTT) compared to placebo, primarily in the left colon. Plasma C4 levels increased, and total cholesterol and LDL cholesterol decreased dose-dependently. A trend toward increased spontaneous bowel movements (SBMs) and looser stool consistency was observed, particularly at the 10 mg dose. [1] In a Phase IIa human study with 36 female patients with functional constipation, Elobixibat at 15 and 20 mg daily for 14 days significantly accelerated colonic transit at 48 hours (both doses) and at 8 and 24 hours (20 mg dose). Stool consistency became significantly looser, ease of passage improved, and straining decreased. Serum C4 levels increased dose-dependently (~2-fold at 15 mg, ~3-fold at 20 mg). [1] In a Phase IIb human trial with 190 CIC patients, Elobixibat at 10 and 15 mg daily for 8 weeks significantly increased the weekly frequency of spontaneous bowel movements (SBMs) from baseline compared to placebo. The increases were dose-dependent and sustained over the 8-week treatment period. Time to first SBM was significantly reduced. Stool consistency was loosened, straining decreased, and bloating severity improved (at 15 mg). Plasma C4 increased, and total cholesterol and LDL cholesterol decreased with the 10 and 15 mg doses. [1] |
| Animal Protocol |
Three-week-old male C57BL/6J mice were randomly divided into two groups (Fig. 1a): (1) CDHF diet + DEN (control group) and (2) CDHF diet + DEN + elobixibat (elobixibat group) groups. The mice received a single intraperitoneal injection of 25-mg/kg DEN at 3 weeks of age. Then, they were fed a standard diet until they reached 8 weeks of age. For the next 20 weeks, mice in the control group were fed a CDHF diet (60 kcal% fat), while those in the elobixibat group were fed a CDHF diet mixed with elobixibat. The animals were housed in a controlled environment (temperature 23 ± 1 °C, humidity 50 ± 10%, 12-h light/dark cycle) at the animal facility with unlimited access to food and water.[2] Dose setting for elobixibat[2] Elobixibat was calculated to be 0.27 mg/kg/day. This study used animals (mean body weight of 23 g) based on the previously published data. The 50% inhibitory concentration of human IBAT is 0.53 nmol/L, and that of mouse IBAT is 0.13 nmol/L. Therefore, the inhibitory activity is four times higher in mice than in humans. The concentration of elobixibat in mice at 70% effective dose is 2.7 mg/kg; while at 50% effective dose, it is 0.27 (70% × [0.023/60])0.33 = 2.23 mg/kg; this would be 110 mg/day for a 50-kg human, 11 times the amount normally used. Therefore, we set our effective capacity at 50%: 0.27 (50%) × [0.023/60])0.33 = 0.223 mg/kg. A CDHF + elobixibat diet containing 3 mg elobixibat per kg of CDHF diet was created and used based on the mean expected body weight and expected food intake.[2] Elobixibat (1.2 mg/kg/day) was administered for the final 4 weeks of this period (elobixibat group). At the end of the study period, the mice were euthanized by inhalation of carbon dioxide.[2] Doses of 0.2, 0.6, or 1.2 mg/kg of elobixibat were administered for 4 weeks, 5 days per week, by oral gavage. Both the control group and the MCD-NASH group were administered PBS on the same schedule by oral gavage. There were no clear effects of 0.2 mg/kg or 0.6 mg/kg (data not shown), but beneficial effects were observed at the 1.2 mg/kg/day dose. This concentration is 4–6 times the dosage for human. According to “Drug Interview Form of Elobixibat”, elobixibat showed strong inhibitory activity to human IBAT, which was about four times more than that of mouse IBAT. Therefore, a dose of 1.2 mg/kg/day in mice is considered equivalent to 0.3 mg/kg/day in human. We evaluated the effects of this dose on the severity of NASH, cytokine production, the intestinal microbiota, and the intestinal TJs in the mice. No diarrhea was observed during the rearing period, and there was no difference in body weight between the NASH and elobixibat groups at the end of the experiment.[2] In a preclinical constipation model, healthy beagle dogs were fed a beef diet for up to 14 days to induce constipation (defined as wet stool weight < 30 g/day). Elobixibat was administered orally at doses of 1.5, 5, and 15 µmol/kg/day. Stool weight was measured as the primary outcome to assess laxative effect. [1] In a separate 28-day pharmacodynamic study in dogs, Elobixibat was administered orally at doses of 5, 25, and 200 µmol/kg/day consecutively. Blood samples were taken to measure plasma levels of 7α-hydroxy-4-cholesten-3-one (C4), a biomarker for hepatic bile acid synthesis, at various time points including 24 hours and after 28 days of treatment. [1] |
| ADME/Pharmacokinetics |
In a Phase I trial in humans, plasma concentrations of Elobixibat were very low following oral administration. The drug was undetectable at doses of 0.1 and 0.3 mg/day, and only detectable in the picomolar range at doses between 1 and 10 mg/day. The highest plasma concentration observed was 0.76 nmol/L. [1] The time to maximum plasma concentration (Tₘₐₓ) occurred within 4 hours of dosing. [1] Plasma elimination occurred within 24 hours of dosing. [1] Elobixibat is minimally absorbed from the gastrointestinal tract after oral administration, resulting in very low systemic bioavailability. [1] The parent compound and no metabolites were detectable in plasma. [1] Elobixibat is highly protein-bound in plasma (>99%). [1] The plasma half-life (t½) is less than 4 hours. [1] Elobixibat inhibits cytochrome P450 enzymes CYP2C9 and CYP3A4 in vitro with IC₅₀ values of 10.3 µM and 6.0 µM, respectively. However, due to its minimal systemic absorption, the risk of clinically relevant drug-drug interactions via this mechanism after oral dosing is considered low. [1] |
| Toxicity/Toxicokinetics |
In Phase I and II clinical trials, no serious adverse events related to Elobixibat were reported. [1] The most common adverse events were gastrointestinal in nature. In a Phase IIa study, mild to moderate abdominal cramps or pain occurred more frequently in the 15 mg (4 of 12 patients) and 20 mg (6 of 12 patients) groups compared to placebo (0 of 13). This pain often preceded bowel movements and resolved afterwards. [1] Diarrhea occurred more frequently at higher doses (four patients in the 20 mg group vs. one each in the 15 mg and placebo groups in the Phase IIa study). All study withdrawals due to adverse events were related to diarrhea. [1] In an 8-week Phase IIb study, the treatment discontinuation rate was higher in the 15 mg group (23%) compared to placebo and lower dose groups (~13%). Adverse events were reported in 54% of patients overall, with 7% being severe. Serious adverse events occurred in three patients but were deemed unrelated to the study medication. [1] There is a theoretical concern that increased delivery of bile acids to the colon could potentially increase the risk of colorectal cancer, although long-term studies in patients with similar conditions (e.g., after partial ileal bypass) have not shown an increased incidence. [1] The depletion of bile acids by Elobixibat could theoretically alter the cholesterol-to-bile acid ratio in bile, potentially increasing the risk of cholesterol gallstone formation. [1] |
| References |
[1]. Elobixibat for the treatment of constipation. Expert Opin Investig Drugs. 2013 Feb; 22(2):277-84. [2].Impact of elobixibat on liver tumors, microbiome, and bile acid levels in a mouse model of nonalcoholic steatohepatitis. Hepatol Int. 2023 Dec;17(6):1378-1392. [3].Elobixibat, an ileal bile acid transporter inhibitor, ameliorates non-alcoholic steatohepatitis in mice. Hepatol Int. 2021 Apr;15(2):392-404. |
| Additional Infomation |
Elobixibat has been used in trials studying the treatment and basic science of Dyslipidemia, Constipation, Chronic Constipation, Functional Constipation, and Chronic Idiopathic Constipation. Drug Indication Treatment of chronic constipation Elobixibat is a first-in-class, orally administered ileal bile acid transporter (IBAT) inhibitor developed for the treatment of chronic idiopathic constipation (CIC). [1] Its mechanism of action involves local inhibition of bile acid reabsorption in the ileum, leading to increased delivery of bile acids to the colon. In the colon, bile acids stimulate motility and secretion, resulting in accelerated colonic transit, looser stools, and improved constipation symptoms. [1] By inhibiting ileal reabsorption, Elobixibat upregulates hepatic synthesis of new bile acids from cholesterol, which may lead to beneficial effects on serum lipid profiles, such as lowering LDL cholesterol. [1] Clinical trials have demonstrated its efficacy in increasing spontaneous bowel movements, loosening stool consistency, reducing straining, and accelerating colonic transit within the first week of treatment, with effects sustained for at least 8 weeks. [1] Its low systemic absorption contributes to a generally favorable safety profile with primarily gastrointestinal side effects (abdominal pain, diarrhea), which are often dose-related and considered an extension of its pharmacological effect. [1] As of the publication date (2013), Elobixibat had not been submitted for regulatory approval in any country and Phase III trials were anticipated. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~250 mg/mL (~359.25 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (2.99 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.08 mg/mL (2.99 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (2.99 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.4370 mL | 7.1850 mL | 14.3701 mL | |
| 5 mM | 0.2874 mL | 1.4370 mL | 2.8740 mL | |
| 10 mM | 0.1437 mL | 0.7185 mL | 1.4370 mL |