Physicochemical Properties
| Molecular Formula | C26H36O5 |
| Molecular Weight | 428.56104850769 |
| Exact Mass | 428.256 |
| CAS # | 41020-79-5 |
| PubChem CID | 10387872 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.187g/cm3 |
| Boiling Point | 579.346ºC at 760 mmHg |
| Flash Point | 248.372ºC |
| Index of Refraction | 1.554 |
| LogP | 4.771 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 31 |
| Complexity | 858 |
| Defined Atom Stereocenter Count | 7 |
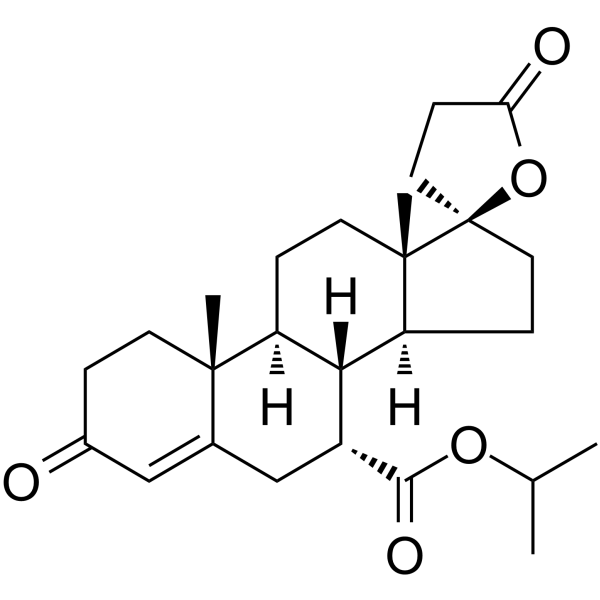
| SMILES | C[C@@]12[C@@]3(OC(CC3)=O)CC[C@@]1([H])[C@@]4([H])[C@]([C@@]5(C(C[C@H]4C(OC(C)C)=O)=CC(CC5)=O)C)([H])CC2 |
| InChi Key | WUVPAYPBMZMHJO-IMNLCBETSA-N |
| InChi Code | InChI=1S/C26H36O5/c1-15(2)30-23(29)18-14-16-13-17(27)5-9-24(16,3)19-6-10-25(4)20(22(18)19)7-11-26(25)12-8-21(28)31-26/h13,15,18-20,22H,5-12,14H2,1-4H3/t18-,19+,20+,22-,24+,25+,26-/m1/s1 |
| Chemical Name | propan-2-yl (7R,8R,9S,10R,13S,14S,17R)-10,13-dimethyl-3,5'-dioxospiro[2,6,7,8,9,11,12,14,15,16-decahydro-1H-cyclopenta[a]phenanthrene-17,2'-oxolane]-7-carboxylate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Dicirenone (code: SC-26304) competitively binds to renal mineralocorticoid receptors (aldosterone receptors, MR), with a Ki value of 0.2 μM for displacing [³H]aldosterone from the receptor[1] |
| ln Vitro |
- Competitive binding to renal aldosterone receptors: Dicirenone inhibited the binding of [³H]aldosterone to rat renal cortical mineralocorticoid receptors in a concentration-dependent manner. At 1 μM, it displaced 90% of [³H]aldosterone binding; at 0.1 μM, the displacement rate was 45%, confirming high affinity for the aldosterone receptor[1] |
| ln Vivo | Both [3H]dipyrenone and [3H]aldosterone have a similar degree of cytoplasmic binding, involving the same collection of sites. [3H]Dicirenone does not produce specific nuclei in three sets of conditions: (i) in intact rats; (ii) in kidney sections; and (iii) in reconstitution studies (mixing prelabeled cytoplasm with purified kidney nuclei or chromatin). In contrast, [3H]aldosterone consistently produces these complexes. The cytoplasmic [3H]aldosterone receptor complex is deposited at 8.5S and 4S in low salt concentrations and at 4.5S in high salt concentrations in a glycerol density gradient. In low salt concentrations, the cytoplasmic [3H]bipyrenone receptor complex is deposited at 3S, and in high salt concentrations, it is deposited at 4S. A receptor system allosteric model is used to discuss these findings. Urinary Na+:creatinine and K+:creatinine ratios were unaffected by the single dose of 3-600 mcg/100 g body weight of decipirenone (SC-26304). Urinary K+:Na+ ratio is the metric used to express these findings. The K+:Na+ ratio rises five times when aldosterone (0.3 μg/100 g body weight) is present. A dose of 180 μg/100 g body weight and 600 μg/100 g body weight of bisirenolone respectively entirely blocked and greatly reduced this rise. Rats administered with 0.036 μg [3H]aldosterone were also injected with the same dose of desirenolone in order to link inhibition with receptor occupancy. Aldosterone specific binding in the cytoplasmic and nuclear fractions was reduced to less than half of control levels at a dose of 180 μg body weight, and specific binding was eliminated at a dose of 600 μg/100 g body weight. Both the binding and physiological doses of aldosterone were significantly below saturating levels, with the physiological dose being around eight times higher than the binding dose [1]. |
| Enzyme Assay |
- Rat renal cortices were homogenized in ice-cold buffer and centrifuged to obtain a crude cytosol fraction (containing aldosterone receptors). The cytosol was incubated with fixed-concentration [³H]aldosterone and Dicirenone (0.01–10 μM) at 4°C for 18 hours. Receptor-ligand complexes were separated from free [³H]aldosterone via glass fiber filter filtration, followed by washing to remove unbound radioactivity. Radioactivity of the complex was measured with a liquid scintillation counter, and the Ki value was calculated from the competition curve using the Scatchard equation[1] |
| References |
[1]. Renal aldosterone receptors: studies with (3H)aldosterone and the anti-mineralocorticoid (3H)spirolactone (SC-26304). Proc Natl Acad Sci U S A. 1974 Apr;71(4):1431-5. |
| Additional Infomation |
Dicirenone is a steroid lactone. - Dicirenone is a synthetic anti-mineralocorticoid and spironolactone analog, with high selectivity for renal aldosterone receptors[1] - Its pharmacological effect relies on competitive antagonism of aldosterone-receptor binding, blocking aldosterone’s renal mineralocorticoid actions (e.g., sodium retention, potassium excretion)[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3334 mL | 11.6670 mL | 23.3340 mL | |
| 5 mM | 0.4667 mL | 2.3334 mL | 4.6668 mL | |
| 10 mM | 0.2333 mL | 1.1667 mL | 2.3334 mL |