Physicochemical Properties
| Molecular Formula | C33H45F3N2O3 |
| Molecular Weight | 574.7172 |
| Exact Mass | 574.338 |
| CAS # | 1191265-33-4 |
| PubChem CID | 66775163 |
| Appearance | White to off-white solid powder |
| LogP | 7.5 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 41 |
| Complexity | 1270 |
| Defined Atom Stereocenter Count | 8 |
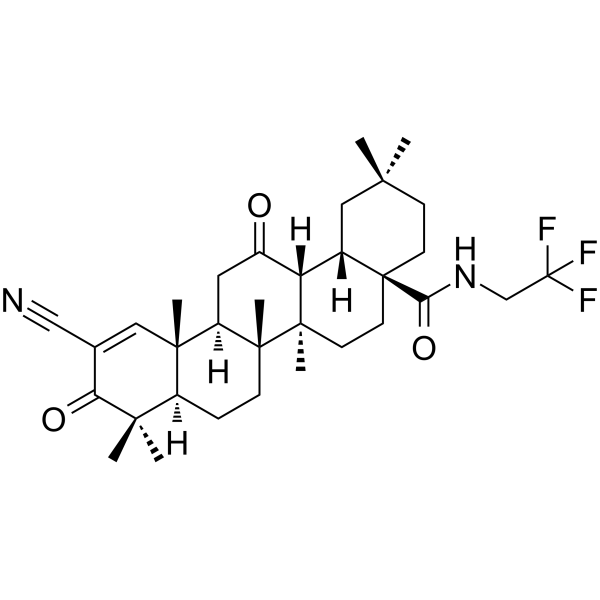
| SMILES | FC(C([H])([H])N([H])C([C@]12C([H])([H])C([H])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[C@@]1([H])[C@@]1([H])C(C([H])([H])[C@]3([H])[C@]4(C([H])=C(C#N)C(C(C([H])([H])[H])(C([H])([H])[H])[C@]4([H])C([H])([H])C([H])([H])[C@@]3(C([H])([H])[H])[C@]1(C([H])([H])[H])C([H])([H])C2([H])[H])=O)C([H])([H])[H])=O)=O)(F)F |
| InChi Key | RMYWYZJDSBWZHH-BFGQVZSYSA-N |
| InChi Code | InChI=1S/C33H45F3N2O3/c1-27(2)10-12-32(26(41)38-18-33(34,35)36)13-11-31(7)24(20(32)16-27)21(39)14-23-29(5)15-19(17-37)25(40)28(3,4)22(29)8-9-30(23,31)6/h15,20,22-24H,8-14,16,18H2,1-7H3,(H,38,41)/t20-,22-,23+,24-,29-,30+,31+,32-/m0/s1 |
| Chemical Name | (4aS,6aR,6aR,6bR,8aR,12aR,14aR,14bS)-11-cyano-2,2,6a,6b,9,9,12a-heptamethyl-10,14-dioxo-N-(2,2,2-trifluoroethyl)-1,3,4,5,6,6a,7,8,8a,13,14a,14b-dodecahydropicene-4a-carboxamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Potent activation of the cytoprotective transcription factor Nrf2 (Nuclear factor erythroid 2–related factor 2) through the Nrf2-Keap1 pathway. Inhibition of the pro-inflammatory transcription factor NF-κB (Nuclear factor kappa-light-chain-enhancer of activated B cells). |
| ln Vivo |
In a rat model of chronic kidney disease (CKD) induced by 5/6 nephrectomy, oral administration of CDDO-dhTFEA (2 mg/kg/day for 12 weeks) restored acetylcholine-induced vasorelaxation in aortic rings, which was markedly impaired in untreated CKD rats. The EC50 for acetylcholine was significantly increased (almost 3-fold) in untreated CKD rats and was restored to sham control levels by CDDO-dhTFEA treatment. [1] Treatment with CDDO-dhTFEA restored Nrf2 activity (nuclear translocation) in aortic tissue, which was significantly reduced in CKD rats. [1] The expression of Nrf2 target genes, including heme oxygenase-1 (Ho-1) and superoxide dismutase 2 (Sod2), which were decreased in CKD aorta, were fully or partially restored by CDDO-dhTFEA administration. [1] The expression of Keap1 (the repressor of Nrf2), which was increased in CKD aorta, was decreased by CDDO-dhTFEA treatment to control levels. [1] The increased protein abundance of nitrotyrosine (a marker of oxidative/nitrosative stress) and the upregulated expression of NAD(P)H oxidase subunits (p47phox, gp91phox, Rac1), 12-lipoxygenase (12-LO), and monocyte chemotactic protein-1 (MCP-1) in the aorta of CKD rats were fully or partially reversed by CDDO-dhTFEA. [1] The increased protein expression of the angiotensin II type 1 receptor (AT1) in the aorta of CKD rats was partially restored toward control levels by CDDO-dhTFEA treatment. [1] |
| Animal Protocol |
Male Sprague-Dawley rats were randomly assigned to undergo 5/6 nephrectomy (CKD model) or sham operation (control). [1] Starting immediately prior to surgery, subgroups of CKD rats were treated orally once daily for 12 weeks with either CDDO-dhTFEA (2 mg/kg) or its vehicle (sesame oil). [1] At the end of the treatment period, rats were euthanized. The thoracic aorta was excised for functional and molecular analyses. [1] For functional assessment, aortic rings were mounted in organ baths for isometric tension recording. After pre-contraction with phenylephrine, cumulative concentration-response curves to acetylcholine were obtained to evaluate endothelium-dependent relaxation. [1] For molecular analyses, aortic tissues were processed for protein extraction. Cytoplasmic and nuclear protein fractions were separated. [1] Western blot analyses were performed on these fractions using specific antibodies against target proteins (e.g., Nrf2, Ho-1, Sod2, Keap1, nitrotyrosine, NAD(P)H oxidase subunits, 12-LO, MCP-1, AT1). Histone H1 and GAPDH were used as loading controls for nuclear and cytoplasmic fractions, respectively. [1] |
| References |
[1]. The synthetic triterpenoid RTA dh404 (CDDO-dhTFEA) restores endothelial function impaired by reduced Nrf2 activity in chronic kidney disease. Redox Biol. 2013 Oct 31;1:527-31. [2]. The synthetic triterpenoid RTA dh404 (CDDO-dhTFEA) restores Nrf2 activity and attenuates oxidative stress, inflammation, and fibrosis in rats with chronic kidney disease. Xenobiotica. 2014 Jun;44(6):570-8. |
| Additional Infomation |
CDDO-dhTFEA is a synthetic oleanane triterpenoid compound derived from oleanolic acid. [1] Its chemical name is CDDO-9,11-dihydro-trifluoroethyl amide. [1] The study demonstrates that endothelial dysfunction in CKD is associated with impaired Nrf2 activity in arterial tissue. [1] The beneficial effects of CDDO-dhTFEA in restoring endothelial function in the CKD rat model are attributed to its dual action of activating the Nrf2-mediated antioxidative pathway and inhibiting the NF-κB-mediated pro-inflammatory pathway, thereby breaking the cycle of oxidative stress and inflammation. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~174.00 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.35 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 2: ≥ 1.72 mg/mL (2.99 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 17.2 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7400 mL | 8.6999 mL | 17.3998 mL | |
| 5 mM | 0.3480 mL | 1.7400 mL | 3.4800 mL | |
| 10 mM | 0.1740 mL | 0.8700 mL | 1.7400 mL |