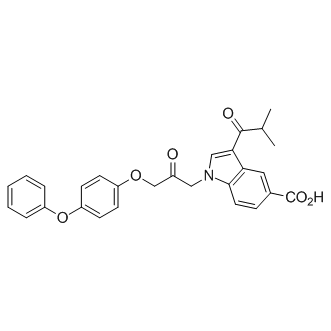
CAY10650 (CAY-10650) is a novel and highly potent inhibitor of cytosolic phospholipase A2α (cPLA2α) (IC50 = 12 nM) with anti-inflammatory activity. cPLA2α is involved in MIP-133 induced apoptosis of corneal epithelial cells, polymorphonuclear neutrophil infiltration, and production of CXCL2. Moreover, cPLA2α inhibitors can be used as a therapeutic target in Acanthamoeba keratitis. CAY10650 demonstrates strong anti-inflammatory effects when applied topically at a dose of 0.1 mg/ear in a mouse model of acute irritant contact dermatitis. Chinese hamsters (n = 6/group) were infected with parasite-laden contact lenses and treated with cPLA2α inhibitors (AACOCF3 and CAY10650) 50 μg/5 μl was injected with topical eye-drop under the contact lens of an infected cornea three times a day for 6 days and topically on days 7–14 postinfection.Treatment with the AACOCF3 and CAY10650 had a profound effect on the severity and chronicity of keratitis.
Physicochemical Properties
| Molecular Formula | C28H25NO6 | |
| Molecular Weight | 471.5 | |
| Exact Mass | 471.168 | |
| CAS # | 1233706-88-1 | |
| Related CAS # |
|
|
| PubChem CID | 45102612 | |
| Appearance | White to off-white solid powder | |
| Density | 1.2±0.1 g/cm3 | |
| Boiling Point | 685.4±55.0 °C at 760 mmHg | |
| Flash Point | 368.3±31.5 °C | |
| Vapour Pressure | 0.0±2.2 mmHg at 25°C | |
| Index of Refraction | 1.607 | |
| LogP | 4.63 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 6 | |
| Rotatable Bond Count | 10 | |
| Heavy Atom Count | 35 | |
| Complexity | 740 | |
| Defined Atom Stereocenter Count | 0 | |
| InChi Key | HTOJZPHWNDZOPQ-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C28H25NO6/c1-18(2)27(31)25-16-29(26-13-8-19(28(32)33)14-24(25)26)15-20(30)17-34-21-9-11-23(12-10-21)35-22-6-4-3-5-7-22/h3-14,16,18H,15,17H2,1-2H3,(H,32,33) | |
| Chemical Name | 3-(2-methylpropanoyl)-1-[2-oxo-3-(4-phenoxyphenoxy)propyl]indole-5-carboxylic acid | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
cytosolic phospholipase A₂-α (cPLA₂-α) [1] |
| ln Vitro |
Neutrophils treated with CAY10650 (12 nM; 30 min) exhibit inhibition of phosphorylated cPLA2-α (p-cPLA2-α) expression[1]. PGE2 release in neutrophils is inhibited by CAY10650 (12 nM; 2 h)[1]. 1. Human neutrophils were pretreated with CAY10650 at a concentration of 12 nM for 30 minutes, then stimulated with Cr-LAAO (50 μg/mL) for 1 hour under the conditions of 37 °C and 5% CO₂. The results showed that CAY10650 inhibited the phosphorylation of cPLA₂-α, reduced the formation of lipid droplets (lipid bodies), and significantly suppressed the secretion of PGE₂. In addition, Western blot analysis indicated that pretreatment with CAY10650 significantly decreased the phosphorylation level of cPLA₂-α, while no direct inhibitory effect on the expression of COX-1, COX-2 and other proteins was observed [1] |
| ln Vivo |
In vivo acanthamoeba keratitis is relieved by CAY10650 (50 μg/5 μL; inject with topical eye-drop; Chinese hamsters)[1]. 1. Chinese hamsters were infected with Acanthamoeba castellanii trophozoite-laden contact lenses to establish an Acanthamoeba keratitis model. CAY10650 was administered at a dose of 50 μg/5μl: subconjunctival injection was performed three times a day for the first 6 days post-infection, and topical administration was adopted from day 7 to day 20 post-infection. The results showed that compared with the untreated infected group, the CAY10650-treated group had significantly reduced clinical severity of Acanthamoeba keratitis. Histopathological examination of the cornea revealed only mild inflammation, minimal polymorphonuclear neutrophil (PMN) infiltration, and significant alleviation of lamellar connective tissue disruption, stromal swelling and neovascularization in the corneal stroma [2] |
| Enzyme Assay |
1. Cell lysates were prepared from 1 × 10⁷ human neutrophils, and pretreated with CAY10650 (12 nM) for 30 minutes, then stimulated with Cr-LAAO (50 μg/mL) for 1 hour at 37 °C and 5% CO₂. Subsequently, the phosphorylation level of cPLA₂-α was detected by Western blot, and the release of PGE₂ was measured by enzyme immunoassay (EIA). The experimental results showed that CAY10650 significantly inhibited the phosphorylation of cPLA₂-α, thereby reducing its enzymatic activity, decreasing the release of arachidonic acid, and ultimately inhibiting the synthesis of PGE₂ [1] |
| Cell Assay |
Western Blot Analysis[1] Cell Types: Neutrophils Tested Concentrations: 12 nM Incubation Duration: 30 minutes Experimental Results: Inhibited cPLA2-α and inhibited the expression of the p-cPLA2-α. Western Blot Analysis[1] Cell Types: Neutrophils Tested Concentrations: 12 nM Incubation Duration: 2 hrs (hours) Experimental Results: Inhibited the PGE2 secretion and inhibited PGE2 realease. 1. 2 × 10⁵ human neutrophils from 3 different donors were divided into control group (RPMI), positive control group (LPS 1 μg/mL) and experimental group (Cr-LAAO 50 μg/mL). Some samples in the experimental group were pretreated with CAY10650 (12 nM) for 30 minutes. The cells were cultured at 37 °C and 5% CO₂ for 1 hour, then lipid droplet staining and counting were performed (the number of lipid droplets in 50 cells was counted with or without fetal bovine serum (FBS)), and the supernatant was collected to detect the concentration of PGE₂ by EIA. The results showed that the number of lipid droplets in the CAY10650-pretreated group was significantly lower than that in the Cr-LAAO-stimulated group without pretreatment, and the concentration of PGE₂ was also significantly reduced. In addition, 1 × 10⁷ human neutrophils were used for Western blot experiment to detect the expression of cPLA₂-α, phosphorylated cPLA₂-α, COX-1/2 and other proteins, and the results showed that CAY10650 significantly decreased the level of phosphorylated cPLA₂-α [1] |
| Animal Protocol |
Animal/Disease Models: Chinese hamsters with infected with parasite-laden contact lenses[1] Doses: 50 μg/5 μL Route of Administration: Inject with topical eye-drop under the contact lens; three times a day for 6 days and topically on days 7 to 20 postinfection Experimental Results: decreased the severity of the keratitis and hasten the onset of resolution. Had little mild inflammation and very few PMNs infiltration in the corneal stroma. 1. Chinese hamsters (9 per group) were used to establish an Acanthamoeba keratitis model by infecting with Acanthamoeba castellanii trophozoite-laden contact lenses. CAY10650 was prepared at a dose of 50 μg/5μl; the administration route and frequency were as follows: subconjunctival injection under the contact lens of the infected cornea three times a day for the first 6 days post-infection, and topical administration from day 7 to day 20 post-infection. The control group of infected hamsters was given an equal amount of BSA (50 μg/5μl). The contact lenses were removed 7 days post-infection, and the clinical severity of the cornea was scored for the indicated time points. After 15 days of drug administration, the hamsters were anesthetized and sacrificed, and corneal tissues were collected for histological examination to observe inflammatory infiltration, stromal damage, neovascularization and other indicators [2] |
| References |
[1]. Cytosolic phospholipase A2-α participates in lipid body formation and PGE2 release in human neutrophils stimulated with an L-amino acid oxidase from Calloselasma rhodostoma venom. Sci Rep. 2020 Jul 3;10(1):10976. [2]. Role of phospholipase A₂ (PLA₂) inhibitors in attenuating apoptosis of the corneal epithelial cells and mitigation of Acanthamoeba keratitis. Exp Eye Res. 2013 Aug;113:182-91. |
| Additional Infomation |
1. CAY10650 is a specific inhibitor of cPLA₂-α. Its mechanism of action is to inhibit the phosphorylation and activation of cPLA₂-α, thereby blocking the cleavage of membrane phospholipids to produce arachidonic acid, reducing the synthesis of PGE₂, and simultaneously inhibiting the formation of lipid droplets (lipid droplets are rich in arachidonic acid and serve as a substrate pool for PGE₂ synthesis) [1] 2. As an inhibitor of cPLA₂-α, CAY10650 can alleviate apoptosis of corneal epithelial cells, polymorphonuclear neutrophil infiltration and production of CXCL2 in Acanthamoeba keratitis by inhibiting the cPLA₂-α-mediated pathway, thereby reducing the pathological damage of keratitis, suggesting that it can be used as a potential therapeutic target for Acanthamoeba keratitis [2] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.30 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.30 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.30 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1209 mL | 10.6045 mL | 21.2089 mL | |
| 5 mM | 0.4242 mL | 2.1209 mL | 4.2418 mL | |
| 10 mM | 0.2121 mL | 1.0604 mL | 2.1209 mL |