Physicochemical Properties
| Molecular Formula | C34H36N6O14S2 |
| Molecular Weight | 816.81 |
| Exact Mass | 816.173 |
| Elemental Analysis | C, 50.00; H, 4.44; N, 10.29; O, 27.42; S, 7.85 |
| CAS # | 145124-30-7 |
| Related CAS # | 145124-30-7 (dimesylate);144849-63-8; |
| PubChem CID | 60916 |
| Appearance | Solid powder |
| Boiling Point | 849.7ºC at 760mmHg |
| Flash Point | 467.7ºC |
| Vapour Pressure | 3.74E-29mmHg at 25°C |
| LogP | 6.034 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 16 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 56 |
| Complexity | 1250 |
| Defined Atom Stereocenter Count | 2 |
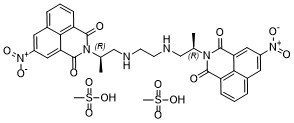
| SMILES | CS(=O)(O)=O.CS(=O)(O)=O.C(NC[C@H](N1C(=O)C2=CC=CC3=CC(=CC(=C23)C1=O)[N+]([O-])=O)C)CNC[C@H](N1C(=O)C2=CC=CC3=CC(=CC(=C23)C1=O)[N+]([O-])=O)C |
| InChi Key | KPQJSSLKKBKWEW-RKDOVGOJSA-N |
| InChi Code | InChI=1S/C32H28N6O8.2CH4O3S/c1-17(35-29(39)23-7-3-5-19-11-21(37(43)44)13-25(27(19)23)31(35)41)15-33-9-10-34-16-18(2)36-30(40)24-8-4-6-20-12-22(38(45)46)14-26(28(20)24)32(36)42;2*1-5(2,3)4/h3-8,11-14,17-18,33-34H,9-10,15-16H2,1-2H3;2*1H3,(H,2,3,4)/t17-,18-;;/m1../s1 |
| Chemical Name | 1H-Benz[de]isoquinoline-1,3(2H)-dione, 2,2′-[1,2-ethanediylbis[imino[(1R)-1-methyl-2,1-ethanediyl]]]bis[5-nitro-, methanesulfonate (1:2) |
| Synonyms | Bisnafide mesylate; DMP840; DMP-840; DMP 840 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| References | 1: Antonini I, Santoni G, Lucciarini R, Amantini C, Sparapani S, Magnano A. Synthesis and biological evaluation of new asymmetrical bisintercalators as potential antitumor drugs. J Med Chem. 2006 Nov 30;49(24):7198-207. doi: 10.1021/jm0606793. PMID: 17125272. 2: Adams MA, Iannuzzi P, Jia Z. MdaB from Escherichia coli: cloning, purification, crystallization and preliminary X-ray analysis. Acta Crystallogr Sect F Struct Biol Cryst Commun. 2005 Feb 1;61(Pt 2):235-8. doi: 10.1107/S1744309105001545. Epub 2005 Feb 1. PMID: 16511004; PMCID: PMC1952255. 3: Carrasco C, Joubert A, Tardy C, Maestre N, Cacho M, Braña MF, Bailly C. DNA sequence recognition by bispyrazinonaphthalimides antitumor agents. Biochemistry. 2003 Oct 14;42(40):11751-61. doi: 10.1021/bi034637h. PMID: 14529286. 4: Bailly C, Carrasco C, Joubert A, Bal C, Wattez N, Hildebrand MP, Lansiaux A, Colson P, Houssier C, Cacho M, Ramos A, Braña MF. Chromophore-modified bisnaphthalimides: DNA recognition, topoisomerase inhibition, and cytotoxic properties of two mono- and bisfuronaphthalimides. Biochemistry. 2003 Apr 15;42(14):4136-50. doi: 10.1021/bi027415c. PMID: 12680768. 5: Rubino JT, Chan LL, Walker JT, Segretario J, Everlof JG, Hussain MA. Photoinduced particulate matter in a parenteral formulation for bisnafide, an experimental antitumor agent. Pharm Dev Technol. 1999 Aug;4(3):439-47. doi: 10.1081/pdt-100101380. PMID: 10434290. 6: Ghaneolhosseini H, Sjöberg S. Synthesis of a boronated naphthalimide for potential use in boron neutron capture therapy (BNCT). Acta Chem Scand (Cph). 1999 Apr;53(4):298-300. doi: 10.3891/acta.chem.scand.53-0298. PMID: 10214688. 7: Segretario J, Cook SC, Umbles CL, Walker JT, Woodeshick RW, Rubino JT, Shea JA. Validation of cleaning procedures for highly potent drugs. II. Bisnafide. Pharm Dev Technol. 1998 Nov;3(4):471-6. doi: 10.3109/10837459809028628. PMID: 9834949. 8: Thompson J, Pratt CB, Stewart CF, Avery L, Bowman L, Zamboni WC, Pappo A. Phase I study of DMP 840 in pediatric patients with refractory solid tumors. Invest New Drugs. 1998;16(1):45-9. doi: 10.1023/a:1006014510078. PMID: 9740543. 9: Lai CM, Garner DM, Gray JE, Brogdon BL, Peterman VC, Pieniaszek HJ Jr. Determination of bisnafide, a novel bis-naphthalimide anticancer agent, in human plasma by high-performance liquid chromatography with UV detection. J Pharm Biomed Anal. 1998 Jul;17(3):427-34. doi: 10.1016/s0731-7085(97)00232-x. PMID: 9656154. 10: O'Reilly S, Baker SD, Sartorius S, Rowinsky EK, Finizio M, Lubiniecki GM, Grochow LB, Gray JE, Pieniaszek HJ Jr, Donehower RC. A phase I and pharmacologic study of DMP 840 administered by 24-hour infusion. Ann Oncol. 1998 Jan;9(1):101-4. doi: 10.1023/a:1008260515869. PMID: 9541690. 11: Nitiss JL, Zhou J, Rose A, Hsiung Y, Gale KC, Osheroff N. The bis(naphthalimide) DMP-840 causes cytotoxicity by its action against eukaryotic topoisomerase II. Biochemistry. 1998 Mar 3;37(9):3078-85. doi: 10.1021/bi9723257. PMID: 9485461. 12: Rubino JT, Nellore R, Parmar B. Dynamic compatibility testing of DMP 840, an experimental antitumor agent. PDA J Pharm Sci Technol. 1997 May-Jun;51(3):130-6. PMID: 9203827. 13: Raghavan KS, Nemeth GA, Gray DB, Hussain MA. Solubility enhancement of a bisnaphthalimide tumoricidal agent, DMP 840, through complexation. Pharm Dev Technol. 1996 Oct;1(3):231-8. doi: 10.3109/10837459609022591. PMID: 9552305. 14: Chatterjee PK, Sternberg NL. A general genetic approach in Escherichia coli for determining the mechanism(s) of action of tumoricidal agents: application to DMP 840, a tumoricidal agent. Proc Natl Acad Sci U S A. 1995 Sep 12;92(19):8950-4. doi: 10.1073/pnas.92.19.8950. PMID: 7568050; PMCID: PMC41085. 15: Pratt CB, Rao BN, Stewart CF, Jenkins JJ 3rd. Successful treatment of metastatic choriocarcinoma after DMP 840. Urol Oncol. 1995 Jul-Aug;1(4):166-7. doi: 10.1016/1078-1439(95)00042-9. PMID: 21224112. 16: Chatterjee PK, Sternberg NL. Using cell-fractionation and photochemical crosslinking methods to determine the cellular binding site(s) of the antitumor drug DMP 840. Photochem Photobiol. 1995 Apr;61(4):360-6. doi: 10.1111/j.1751-1097.1995.tb08623.x. PMID: 7740079. 17: LoRusso P, Demchik L, Dan M, Polin L, Gross JL, Corbett TH. Comparative efficacy of DMP 840 against mouse and human solid tumor models. Invest New Drugs. 1995;13(3):195-203. doi: 10.1007/BF00873800. PMID: 8729946. 18: Cobb PW, Degen DR, Clark GM, Chen SF, Kuhn JG, Gross JL, Kirshenbaum MR, Sun JH, Burris HA 3rd, Von Hoff DD. Activity of DMP 840, a new bis-naphthalimide, on primary human tumor colony-forming units. J Natl Cancer Inst. 1994 Oct 5;86(19):1462-5. doi: 10.1093/jnci/86.19.1462. PMID: 8089865. 19: Kirshenbaum MR, Chen SF, Behrens CH, Papp LM, Stafford MM, Sun JH, Behrens DL, Fredericks JR, Polkus ST, Sipple P, et al. (R,R)-2,2'-[1,2-ethanediylbis[imino(1-methyl-2,1-ethanediyl)]]- bis[5-nitro-1H-benz[de]isoquinoline-1,3-(2H)-dione] dimethanesulfonate (DMP 840), a novel bis-naphthalimide with potent nonselective tumoricidal activity in vitro. Cancer Res. 1994 Apr 15;54(8):2199-206. PMID: 8174127. 20: Houghton PJ, Cheshire PJ, Hallman JC 3rd, Gross JL, McRipley RJ, Sun JH, Behrens CH, Dexter DL, Houghton JA. Evaluation of a novel bis-naphthalimide anticancer agent, DMP 840, against human xenografts derived from adult, juvenile, and pediatric cancers. Cancer Chemother Pharmacol. 1994;33(4):265-72. doi: 10.1007/BF00685898. PMID: 8281618. |
| Additional Infomation |
Bisnafide Dimesylate is the dimesylate salt form of bisnafide, a bis-naphthalimide compound with anticancer activity. Bisnafide selectively intercalates guanine-cytosine (GC) rich regions of DNA, thereby interfering with DNA replication machinery and activity of topoisomerase II. As a result, this agent causes potent cytotoxicity. See also: Bisnafide (annotation moved to). |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.2243 mL | 6.1214 mL | 12.2427 mL | |
| 5 mM | 0.2449 mL | 1.2243 mL | 2.4485 mL | |
| 10 mM | 0.1224 mL | 0.6121 mL | 1.2243 mL |