Physicochemical Properties
| Molecular Formula | C17H14N2O2 |
| Molecular Weight | 278.311 |
| Exact Mass | 278.106 |
| Elemental Analysis | C, 73.37; H, 5.07; N, 10.07; O, 11.50 |
| CAS # | 117545-11-6 |
| PubChem CID | 60674 |
| Appearance | Solid powder |
| Density | 1.29g/cm3 |
| Boiling Point | 498.4ºC at 760mmHg |
| Flash Point | 255.2ºC |
| Vapour Pressure | 4.56E-10mmHg at 25°C |
| Index of Refraction | 1.651 |
| LogP | 2.78 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 21 |
| Complexity | 591 |
| Defined Atom Stereocenter Count | 0 |
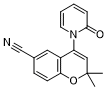
| SMILES | CC1(C)C=C(N2C=CC=CC2=O)C2C=C(C#N)C=CC=2O1 |
| InChi Key | JTVSKASWNROQQF-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C17H14N2O2/c1-17(2)10-14(19-8-4-3-5-16(19)20)13-9-12(11-18)6-7-15(13)21-17/h3-10H,1-2H3 |
| Chemical Name | 2,2-dimethyl-4-(2-oxopyridin-1(2H)-yl)-2H-chromene-6-carbonitrile |
| Synonyms | Bimakalim; EMD 52692; EMD-52692; EMD52692; SR 44866; SR-44866; SR44866; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| References | 1: Alexandre J, Puddu PE, Simard C, Hof T, Sallé L, Guinamard R, Manrique A, Rouet R, Beygui F, Milliez P. Proarrhythmic effects of aldosterone during myocardial ischemia-reperfusion: implication of the sarcolemmal-KATP channels. J Cardiovasc Pharmacol. 2014 Aug;64(2):134-41. doi: 10.1097/FJC.0000000000000097. PMID: 24662493. 2: Aidonidis I, Poyatzi A, Stamatiou G, Lymberi M, Stamatoyannis N, Molyvdas PA. Dose-related shortening of ventricular tachycardia cycle length after administration of the KATP channel opener bimakalim in a 4-day-old chronic infarct anesthetized pig model. J Cardiovasc Pharmacol Ther. 2009 Sep;14(3):222-30. doi: 10.1177/1074248409338929. Epub 2009 Jul 8. PMID: 19587223. 3: Stephan D, Salamon E, Weber H, Russ U, Lemoine H, Quast U. KATP channel openers of the benzopyran type reach their binding site via the cytosol. Br J Pharmacol. 2006 Sep;149(2):199-205. doi: 10.1038/sj.bjp.0706858. Epub 2006 Aug 14. PMID: 16921394; PMCID: PMC2013803. 4: Merkus D, Sorop O, Houweling B, Hoogteijling BA, Duncker DJ. KCa+ channels contribute to exercise-induced coronary vasodilation in swine. Am J Physiol Heart Circ Physiol. 2006 Nov;291(5):H2090-7. doi: 10.1152/ajpheart.00315.2006. Epub 2006 May 12. PMID: 16699076. 5: Gumina RJ, El Schultz J, Moore J, Beier N, Schelling P, Gross GJ. Cardioprotective-mimetics reduce myocardial infarct size in animals resistant to ischemic preconditioning. Cardiovasc Drugs Ther. 2005 Oct;19(5):315-22. doi: 10.1007/s10557-005-3693-8. PMID: 16382293. 6: Bayés M, Rabasseda X, Prous JR. Gateways to clinical trials. Methods Find Exp Clin Pharmacol. 2005 Mar;27(2):145-59. PMID: 15834466. 7: Burian M, Piske M, Petkovic D, Mitrovic V. Lack of anti-ischemic efficacy of the potassium channel opener bimakalim in patients with stable angina pectoris. Cardiovasc Drugs Ther. 2004 Jan;18(1):37-46. doi: 10.1023/B:CARD.0000025754.08942.03. PMID: 15115902. 8: Camara AK, Chen Q, Rhodes SS, Riess ML, Stowe DF. Negative inotropic drugs alter indexes of cytosolic [Ca(2+)]-left ventricular pressure relationships after ischemia. Am J Physiol Heart Circ Physiol. 2004 Aug;287(2):H667-80. doi: 10.1152/ajpheart.01142.2003. Epub 2004 Apr 1. PMID: 15059780. 9: Gauthier KM, Jagadeesh SG, Falck JR, Campbell WB. 14,15-epoxyeicosa-5(Z)-enoic-mSI: a 14,15- and 5,6-EET antagonist in bovine coronary arteries. Hypertension. 2003 Oct;42(4):555-61. doi: 10.1161/01.HYP.0000091265.94045.C7. Epub 2003 Sep 2. PMID: 12953017. 10: Fujimoto K, Bosnjak ZJ, Kwok WM. Isoflurane-induced facilitation of the cardiac sarcolemmal K(ATP) channel. Anesthesiology. 2002 Jul;97(1):57-65. doi: 10.1097/00000542-200207000-00009. PMID: 12131104. 11: Gauthier KM, Deeter C, Krishna UM, Reddy YK, Bondlela M, Falck JR, Campbell WB. 14,15-Epoxyeicosa-5(Z)-enoic acid: a selective epoxyeicosatrienoic acid antagonist that inhibits endothelium-dependent hyperpolarization and relaxation in coronary arteries. Circ Res. 2002 May 17;90(9):1028-36. doi: 10.1161/01.res.0000018162.87285.f8. PMID: 12016270. 12: Salamon E, Mannhold R, Weber H, Lemoine H, Frank W. 6-Sulfonylchromenes as highly potent K(ATP)-channel openers. J Med Chem. 2002 Feb 28;45(5):1086-97. doi: 10.1021/jm010999g. PMID: 11855989. 13: Das B, Sarkar C, Karanth KS. Effects of administration of nicorandil or bimakalim prior to and during ischemia or reperfusion on survival rate, ischemia/reperfusion-induced arrhythmias and infarct size in anesthetized rabbits. Naunyn Schmiedebergs Arch Pharmacol. 2001 Nov;364(5):383-96. doi: 10.1007/s002100100457. PMID: 11692221. 14: Pountney DJ, Sun ZQ, Porter LM, Nitabach MN, Nakamura TY, Holmes D, Rosner E, Kaneko M, Manaris T, Holmes TC, Coetzee WA. Is the molecular composition of K(ATP) channels more complex than originally thought? J Mol Cell Cardiol. 2001 Aug;33(8):1541-6. doi: 10.1006/jmcc.2001.1407. PMID: 11448141. 15: Pratt PF, Li P, Hillard CJ, Kurian J, Campbell WB. Endothelium-independent, ouabain-sensitive relaxation of bovine coronary arteries by EETs. Am J Physiol Heart Circ Physiol. 2001 Mar;280(3):H1113-21. doi: 10.1152/ajpheart.2001.280.3.H1113. PMID: 11179054. 16: Eells JT, Henry MM, Gross GJ, Baker JE. Increased mitochondrial K(ATP) channel activity during chronic myocardial hypoxia: is cardioprotection mediated by improved bioenergetics? Circ Res. 2000 Nov 10;87(10):915-21. doi: 10.1161/01.res.87.10.915. PMID: 11073888. 17: Holmes DS, Sun ZQ, Porter LM, Bernstein NE, Chinitz LA, Artman M, Coetzee WA. Amiodarone inhibits cardiac ATP-sensitive potassium channels. J Cardiovasc Electrophysiol. 2000 Oct;11(10):1152-8. doi: 10.1111/j.1540-8167.2000.tb01762.x. PMID: 11059980. 18: Mitrovic V, Oehm E, Thormann J, Pitschner H, Hamm C. Potassium channel openers and blockers in coronary artery disease. Comparison to betablockers and calcium antagonists. Herz. 2000 Mar;25(2):130-42. doi: 10.1007/pl00001951. PMID: 10829253. 19: Buchheit KH, Hofmann A, Manley P, Pfannkuche HJ, Quast U. Atypical effect of minoxidil sulphate on guinea pig airways. Naunyn Schmiedebergs Arch Pharmacol. 2000 Apr;361(4):418-24. doi: 10.1007/s002100000218. PMID: 10763857. 20: Monti F, Iwashiro K, Picard S, Criniti A, La Francesca S, Ruvolo G, Papalia U, Campa PP, Marino B, Puddu PE. Adenosine triphosphate-dependent potassium channel modulation and cardioplegia-induced protection of human atrial muscle in an in vitro model of myocardial stunning. J Thorac Cardiovasc Surg. 2000 Apr;119(4 Pt 1):842-8. doi: 10.1016/S0022-5223(00)70022-5. PMID: 10733778. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.5931 mL | 17.9656 mL | 35.9312 mL | |
| 5 mM | 0.7186 mL | 3.5931 mL | 7.1862 mL | |
| 10 mM | 0.3593 mL | 1.7966 mL | 3.5931 mL |