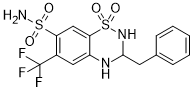
Bendroflumethiazide (Benuron; Bendrofluazide; Bentride) is a orally bioactive diuretic and an antihypertensive agent used to treat hypertension. It acts by inhibiting sodium reabsorption at the beginning of the distal convoluted tubule and may play an important role in the treatment of mild heart failure.
Physicochemical Properties
| Molecular Formula | C15H14F3N3O4S2 |
| Molecular Weight | 421.41 |
| Exact Mass | 421.037 |
| CAS # | 73-48-3 |
| Related CAS # | (Rac)-Bendroflumethiazide-d5;1330183-13-5 |
| PubChem CID | 2315 |
| Appearance |
Crystals from dioxane. WHITE TO CREAM-COLORED, FINELY DIVIDED, CRYSTALLINE POWDER |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 602.1±65.0 °C at 760 mmHg |
| Melting Point | 205-207ºC |
| Flash Point | 318.0±34.3 °C |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C |
| Index of Refraction | 1.584 |
| LogP | 2.07 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 27 |
| Complexity | 740 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | HDWIHXWEUNVBIY-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C15H14F3N3O4S2/c16-15(17,18)10-7-11-13(8-12(10)26(19,22)23)27(24,25)21-14(20-11)6-9-4-2-1-3-5-9/h1-5,7-8,14,20-21H,6H2,(H2,19,22,23) |
| Chemical Name | 3-Benzyl-1,1-dioxo-6-(trifluoromethyl)-3,4-dihydro-2H-1,2,4- benzothiadiazine-7-sulfonamide |
| Synonyms | Bentride Benuron FT 81Livesan NaigarilUrleaRelan Beta SaluralRauzideBenzy-RodiuranOrsile PlurylBendroflumethiazide Aprinox Be 724A Bendrofluazide Berkozide BristuronCentyl SinesalinNaturetin Neo-Rontyl NiagarilPluryle Poliuron Sodiuretic |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Absorbed relatively rapidly after oral administration MOST CMPD ARE RAPIDLY EXCRETED WITHIN 3-6 HR. BENDROFLUMETHIAZIDE...LONGER DURATION OF ACTION THAT IS CORRELATED WITH...SLOWER EXCRETION. THIAZIDES ARE ABSORBED FROM GI TRACT & OWE THEIR USEFULNESS LARGELY TO THEIR EFFECTIVENESS BY ORAL ROUTE. ABSORPTION IS RELATIVELY RAPID. MOST AGENTS SHOW DEMONSTRABLE DIURETIC EFFECT WITHIN HR AFTER ORAL ADMIN. /BENZOTHIADIAZIDES/ IN GENERAL, THIAZIDES WITH RELATIVELY LONG DURATIONS OF ACTION SHOW PROPORTIONATELY HIGH DEGREE OF BINDING TO PLASMA PROTEINS & ARE REABSORBED... BY RENAL TUBULES. ... DRUG PASSES READILY THROUGH PLACENTAL BARRIER TO FETUS. ALL THIAZIDES PROBABLY UNDERGO ACTIVE SECRETION IN PROXIMAL TUBULE. /THIAZIDE DIURETICS/ BENDROFLUMETHIAZIDE WAS ADMIN ORALLY TO 9 HEALTHY VOLUNTEERS. PEAK PLASMA LEVELS REACHED @ 1 + OR - 0.4 HR. CONCN DECLINED WITH MEAN T/2 OF 3 HR. APPARENT VOL OF DISTRIBUTION AVG 1.48 L/KG. MAJOR PART ELIMINATED VIA NONRENAL MECHANISMS. URINARY RECOVERY AVG 30%. Bendroflumethiazide appears to be well absorbed from the GI tract. The drug is excreted unchanged in urine, and excretion is essentially complete within 24 hours. Biological Half-Life 8.5 hours Halflife is 3-3.9 hrs. /From table/ |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation No information is available on the amount of bendroflumethiazide in breastmilk. Intense diuresis with large doses of bendroflumethiazide can decrease breastmilk production, especially during the neonatal period. Shorter-acting diuretics in low doses are preferred over bendroflumethiazide. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Bendroflumethiazide has been used to suppress lactation in oral doses of 5 mg twice daily for 5 days, and in doses of 10 mg in the morning and 5 mg in the afternoon. The added contribution of the diuretic to these measures, which are effective in suppressing lactation, has not been studied. There are no data on the effects of potent diuretics on established, ongoing lactation. Protein Binding 96% |
| References | :Treatment of arterial hypertensive disease with diuretics. I. Effects on blood pressure of bendroflumethiazide, potassium chloride, and spironolactone. Arch Intern Med. 1962;110:26-33. |
| Additional Infomation |
Bendroflumethiazide is a sulfonamide consisting of 7-sulfamoyl-3,4-dihydro-2H-1,2,4-benzothiadiazine 1,1-dioxide in which the hydrogen at position 6 is substituted by a trifluoromethyl group and that at position 3 is substituted by a benzyl group. It has a role as a diuretic and an antihypertensive agent. It is a benzothiadiazine and a sulfonamide. A thiazide diuretic with actions and uses similar to those of hydrochlorothiazide. It has been used in the treatment of familial hyperkalemia, hypertension, edema, and urinary tract disorders. (From Martindale, The Extra Pharmacopoeia, 30th ed, p810) Bendroflumethiazide is a Thiazide Diuretic. The physiologic effect of bendroflumethiazide is by means of Increased Diuresis. Bendroflumethiazide is a long-acting agent, also known as bendrofluazide, belonging to the class of thiazide diuretics with antihypertensive activity. A thiazide diuretic with actions and uses similar to those of HYDROCHLOROTHIAZIDE. It has been used in the treatment of familial hyperkalemia, hypertension, edema, and urinary tract disorders. (From Martindale, The Extra Pharmacopoeia, 30th ed, p810) See also: Bendroflumethiazide; nadolol (component of). Drug Indication For the treatment of high blood pressure and management of edema related to heart failure. Mechanism of Action As a diuretic, bendroflumethiazide inhibits active chloride reabsorption at the early distal tubule via the Na-Cl cotransporter, resulting in an increase in the excretion of sodium, chloride, and water. Thiazides like bendroflumethiazide also inhibit sodium ion transport across the renal tubular epithelium through binding to the thiazide sensitive sodium-chloride transporter. This results in an increase in potassium excretion via the sodium-potassium exchange mechanism. The antihypertensive mechanism of bendroflumethiazide is less well understood although it may be mediated through its action on carbonic anhydrases in the smooth muscle or through its action on the large-conductance calcium-activated potassium (KCa) channel, also found in the smooth muscle. ...BENZOTHIADIAZIDES HAVE DIRECT EFFECT ON RENAL TUBULAR TRANSPORT OF SODIUM & CHLORIDE...INDEPENDENT OF ANY EFFECT ON CARBONIC ANHYDRASE. /THIAZIDE DIURETICS/ NATURE OF CHEM INTERACTION BETWEEN THIAZIDES & SPECIFIC RENAL RECEPTORS RESPONSIBLE FOR CHLORURETIC EFFECT IS NOT KNOWN; NO CRITICAL ENZYMATIC REACTIONS HAVE BEEN IDENTIFIED. /THIAZIDE DIURETICS/ ...MAY DECR EXCRETION OF URIC ACID IN MAN, THUS INCR ITS CONCN IN PLASMA. HYPERURICEMIC EFFECT RESULTS PRIMARILY FROM INHIBITION OF TUBULAR SECRETION OF URATE. ... UNLIKE MOST OTHER NATRIURETIC AGENTS...DECR RENAL EXCRETION OF CALCIUM RELATIVE TO THAT OF SODIUM... /ENHANCE/ EXCRETION OF MAGNESIUM... /THIAZIDE DIURETICS/ THIAZIDES INHIBIT REABSORPTION OF SODIUM &...CHLORIDE IN DISTAL SEGMENT. ... AS CLASS...HAVE IMPORTANT ACTION ON EXCRETION OF POTASSIUM THAT RESULTS FROM INCR SECRETION OF CATION BY DISTAL TUBULE. ... GLOMERULAR FILTRATION RATE MAY BE REDUCED BY THIAZIDES, PARTICULARLY WITH IV ADMIN FOR EXPTL PURPOSES. /THIAZIDE DIURETICS/ For more Mechanism of Action (Complete) data for BENDROFLUMETHIAZIDE (11 total), please visit the HSDB record page. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~250 mg/mL (~593.25 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3730 mL | 11.8649 mL | 23.7299 mL | |
| 5 mM | 0.4746 mL | 2.3730 mL | 4.7460 mL | |
| 10 mM | 0.2373 mL | 1.1865 mL | 2.3730 mL |