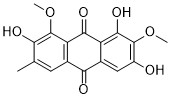
Aurantio-obtusin is a novel and potent anthraquinone isolated from Semen Cassiae. Aurantio-obtusin has a variety of biological effects, including those that are anti-oxidative, anti-coagulant, anti-inflammatory, and anti-hypertensive. Additionally, aurantio-obtusin, which relaxes systemic arteries in rats via an endothelial PI3K/AKT/eNOS-dependent signaling pathway, is a potential vasodilator. It is possible to treat diseases associated with allergies by using aurantio-obtusin, an inhibitor of allergic responses in IgE-mediated mast cells and anaphylactic models.
Physicochemical Properties
| Molecular Formula | C17H14O7 |
| Molecular Weight | 330.2889 |
| Exact Mass | 330.073 |
| Elemental Analysis | C, 61.82; H, 4.27; O, 33.91 |
| CAS # | 67979-25-3 |
| Related CAS # | 67979-25-3 |
| PubChem CID | 155011 |
| Appearance | Yellow to orange a crystalline solid |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 594.6±50.0 °C at 760 mmHg |
| Flash Point | 222.4±23.6 °C |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C |
| Index of Refraction | 1.679 |
| LogP | 3.81 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 24 |
| Complexity | 521 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O(C([H])([H])[H])C1=C(C(C([H])([H])[H])=C([H])C2C(C3=C([H])C(=C(C(=C3C(C=21)=O)O[H])OC([H])([H])[H])O[H])=O)O[H] |
| InChi Key | RNXZPKOEJUFJON-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C17H14O7/c1-6-4-7-11(17(24-3)12(6)19)14(21)10-8(13(7)20)5-9(18)16(23-2)15(10)22/h4-5,18-19,22H,1-3H3 |
| Chemical Name | 1,3,7-trihydroxy-2,8-dimethoxy-6-methylanthracene-9,10-dione |
| Synonyms | Aurantio obtusin; Aurantio-obtusin; Aurantioobtusin |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Aurantio-obtusin (6.25-100 μM; 24 h) can significantly reduce the production of NO and PGE2, and significantly inhibit IL-6, TNF-α and COX in RAW264.7 cells treated with LPS (0.2 μg/mL) -2 Aurantio-obtusin (6.25-100 μM; 12 h) inhibits NF-κB activation in RAW264.7 cells treated with LPS (0.2 μg/mL) by inhibiting i-κB and IKK phosphorylation. Aurantioobtusin (1-10000 nM) produces MA small resistance vasodilation in a concentration-dependent manner [1]. |
| ln Vivo | In HFSW-induced mice, aurantio-obtusin (5–15 mg/kg; face; single dose) decreases lipid droplet accumulation and widespread steatosis in a dosage-dependent manner [3]. Oryza obtusina (5). |
| Animal Protocol |
Animal/Disease Models: C57BL/6J mouse model [3] Doses: 5 mg/ -15 mg/kg; receptor; single dose) Inhibits factor synthesis in HFSW-induced cytokines and promotes FAO activation of AMPK signaling and autophagy [3 ]. kg, 10 mg/kg, 15 mg/kg Route of Administration: po (oral gavage) on HFSW diet for 4 weeks, followed by varying doses of Aurantio-obtusin for a further 4 weeks. Experimental Results: HSFW can reduce the levels of TG, TC in liver and TG, ALT and AST in serum. Reduces the number and size of fat droplets in liver cells. AMPK phosphorylation was Dramatically increased in HFSW-induced mice. |
| References |
[1]. Aurantio-obtusin relaxes systemic arteries through endothelial PI3K/AKT/eNOS-dependent signaling pathway in rats. Journal of pharmacological sciences vol. 128,3 (2015): 108-15. [2]. Anti-Inflammatory Effects of Aurantio-Obtusin from Seed of Cassia obtusifolia L. through Modulation of the NF-κB Pathway. Molecules (Basel, Switzerland) vol. 23,12 3093. 27 Nov. 2018. [3]. Aurantio-Obtusin Attenuates Non-Alcoholic Fatty Liver Disease Through AMPK-Mediated Autophagy and Fatty Acid Oxidation Pathways. Frontiers in pharmacology vol. 12 826628. 11 Jan. 2022. |
| Additional Infomation |
Aurantio-obtusin is a trihydroxyanthraquinone that is 1,3,7-trihydroxy-9,10-anthraquinone which is by methoxy groups at positions 2 and 8, and by a methyl group at position 6. Aurantio-obtusin has been reported in Senna obtusifolia and Senna tora with data available. |
Solubility Data
| Solubility (In Vitro) | DMSO: 25~66 mg/mL (75.7~199.8 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (6.30 mM) (saturation unknown) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 + to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0276 mL | 15.1382 mL | 30.2764 mL | |
| 5 mM | 0.6055 mL | 3.0276 mL | 6.0553 mL | |
| 10 mM | 0.3028 mL | 1.5138 mL | 3.0276 mL |