Aprotinin is a novel and potent small protein serine protease / bovine pancreatic trypsin inhibitor (BPTI) with antifibrinolytic activity. It has a Kis of 0.06 pM and 9 nM for trypsin and chymotrypsin inhibition, respectively. The use of aprotinin lowers transfusion and perioperative blood loss. Aprotinin use was associated with a higher percentage of patients with doubled serum creatinine levels, but did not significantly increase the risk of renal failure or the need for postoperative renal replacement. Renal failure was not mentioned in the death adjudication as a factor in the death linked to aprotinin use. According to Brown and colleagues' meta-analysis, high-dose aprotinin did not significantly increase the risk of renal failure.
Physicochemical Properties
| Molecular Formula | C284H432N84O79S7 | |
| Molecular Weight | 6511.51 | |
| Exact Mass | 6507 | |
| Elemental Analysis | C, 52.44; H, 6.59; N, 18.09; O, 19.43; S, 3.45 | |
| CAS # | 9087-70-1 | |
| Related CAS # |
|
|
| PubChem CID | 16130295 | |
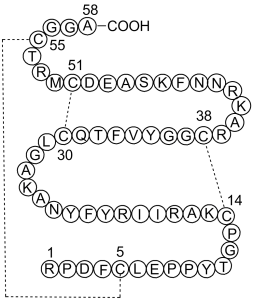
| Sequence | Arg-Pro-Asp-Phe-Cys-Leu-Glu-Pro-Pro-Tyr-Thr-Gly-Pro-Cys-Lys-Ala-Arg-Ile-Ile-Arg-Tyr-Phe-Tyr-Asn-Ala-Lys-Ala-Gly-Leu-Cys-Gln-Thr-Phe-Val-Tyr-Gly-Gly-Cys-Arg-Ala-Lys-Arg-Asn-Asn-Phe-Lys-Ser-Ala-Glu-Asp-Cys-Met-Arg-Thr-Cys-Gly-Gly-Ala(Disulfide bridge: Cys5-Cys55,Cys14-Cys38,Cys30-Cys51) | |
| SequenceShortening | RPDFCLEPPYTGPCKARIIRYFYNAKAGLCQTFVYGGCRAKRNNFKSAEDCMRTCGGA(Disulfide bridge: Cys5-Cys55,Cys14-Cys38,Cys30-Cys51) | |
| Appearance | Off-white to light brown solid powder | |
| Melting Point | >100 °C | |
| LogP | -25.4 | |
| Hydrogen Bond Donor Count | 93 | |
| Hydrogen Bond Acceptor Count | 97 | |
| Rotatable Bond Count | 111 | |
| Heavy Atom Count | 454 | |
| Complexity | 16700 | |
| Defined Atom Stereocenter Count | 57 | |
| SMILES | NC(=N)NCCC[C@@H](N)C(=O)N1[C@H](CCC1)C(=O)N[C@H](CC(O)=O)C(=O)N[C@@H](CC2=CC=CC=C2)C(=O)N[C@H]3C(=O)N[C@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N4[C@H](CCC4)C(=O)N5[C@@H](CCC5)C(=O)N[C@H](CC6=CC=C(O)C=C6)C(=O)N[C@@H]([C@H](C)O)C(=O)NCC(=O)N7[C@@H](CCC7)C(=O)N[C@H]8C(=O)N[C@@H](CCCCN)C(=O)N[C@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@H]([C@H](C)CC)C(=O)N[C@H](CCCNC(N)=N)C(=O)N[C@@H](CC9=CC=C(O)C=C9)C(=O)N[C@H](CC%10=CC=CC=C%10)C(=O)N[C@@H](CC%11=CC=C(O)C=C%11)C(=O)N[C@H](CC(N)=O)C(=O)N[C@@H](C)C(=O)N[C@H](CCCCN)C(=O)N[C@@H](C)C(=O)NCC(=O)N[C@@H](CC(C)C)C(=O)N[C@H]%12C(=O)N[C@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@H](CC%13=CC=CC=C%13)C(=O)N[C@H](C(C)C)C(=O)N[C@H](CC%14=CC=C(O)C=C%14)C(=O)NCC(=O)NCC(=O)N[C@H](CSSC8)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@H](C)C(=O)N[C@@H](CCCCN)C(=O)N[C@H](CCCNC(N)=N)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@H](CC(N)=O)C(=O)N[C@@H](CC%15=CC=CC=C%15)C(=O)N[C@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@H](C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CSSC%12)C(=O)N[C@@H](CCSC)C(=O)N[C@H](CCCNC(N)=N)C(=O)N[C@@H]([C@H](C)O)C(=O)N[C@@H](CSSC3)C(=O)NCC(=O)NCC(=O)N[C@H](C)C(O)=O |
|
| InChi Key | ZPNFWUPYTFPOJU-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C284H432N84O79S7/c1-21-144(9)222-271(439)337-174(68-46-105-309-282(300)301)239(407)340-187(120-160-77-85-164(374)86-78-160)251(419)341-185(116-156-55-29-24-30-56-156)250(418)342-188(121-161-79-87-165(375)88-80-161)252(420)346-191(123-208(291)378)246(414)322-149(14)230(398)326-168(62-35-39-98-285)234(402)319-146(11)227(395)314-132-215(385)324-181(113-141(3)4)247(415)354-199-137-452-453-138-200-263(431)336-179(97-112-448-20)242(410)331-176(70-48-107-311-284(304)305)244(412)363-226(154(19)372)274(442)358-197(233(401)316-129-212(382)312-130-213(383)318-151(16)278(446)447)135-449-451-139-201(355-253(421)186(117-157-57-31-25-32-58-157)344-256(424)195(127-220(393)394)350-267(435)204-72-50-109-366(204)275(443)167(289)61-43-102-306-279(294)295)265(433)339-182(114-142(5)6)248(416)338-180(93-96-218(389)390)276(444)368-111-52-74-206(368)277(445)367-110-51-73-205(367)268(436)349-189(122-162-81-89-166(376)90-82-162)259(427)362-224(152(17)370)269(437)317-133-216(386)365-108-49-71-203(365)266(434)357-202(264(432)333-169(63-36-40-99-286)235(403)320-148(13)229(397)328-175(69-47-106-310-283(302)303)243(411)360-223(145(10)22-2)272(440)361-222)140-454-450-136-198(325-214(384)131-313-211(381)128-315-232(400)183(119-159-75-83-163(373)84-76-159)351-270(438)221(143(7)8)359-258(426)190(118-158-59-33-26-34-60-158)352-273(441)225(153(18)371)364-245(413)177(335-262(199)430)91-94-207(290)377)261(429)334-172(66-44-103-307-280(296)297)236(404)321-147(12)228(396)327-170(64-37-41-100-287)237(405)330-173(67-45-104-308-281(298)299)238(406)345-192(124-209(292)379)255(423)347-193(125-210(293)380)254(422)343-184(115-155-53-27-23-28-54-155)249(417)332-171(65-38-42-101-288)240(408)353-196(134-369)260(428)323-150(15)231(399)329-178(92-95-217(387)388)241(409)348-194(126-219(391)392)257(425)356-200/h23-34,53-60,75-90,141-154,167-206,221-226,369-376H,21-22,35-52,61-74,91-140,285-289H2,1-20H3,(H2,290,377)(H2,291,378)(H2,292,379)(H2,293,380)(H,312,382)(H,313,381)(H,314,395)(H,315,400)(H,316,401)(H,317,437)(H,318,383)(H,319,402)(H,320,403)(H,321,404)(H,322,414)(H,323,428)(H,324,385)(H,325,384)(H,326,398)(H,327,396)(H,328,397)(H,329,399)(H,330,405)(H,331,410)(H,332,417)(H,333,432)(H,334,429)(H,335,430)(H,336,431)(H,337,439)(H,338,416)(H,339,433)(H,340,407)(H,341,419)(H,342,418)(H,343,422)(H,344,424)(H,345,406)(H,346,420)(H,347,423)(H,348,409)(H,349,436)(H,350,435)(H,351,438)(H,352,441)(H,353,408)(H,354,415)(H,355,421)(H,356,425)(H,357,434)(H,358,442)(H,359,426)(H,360,411)(H,361,440)(H,362,427)(H,363,412)(H,364,413)(H,387,388)(H,389,390)(H,391,392)(H,393,394)(H,446,447)(H4,294,295,306)(H4,296,297,307)(H4,298,299,308)(H4,300,301,309)(H4,302,303,310)(H4,304,305,311) | |
| Chemical Name | 4-[[1-[[29a,62a,69,84-tetrakis(4-aminobutyl)-35a,75,78-tris(2-amino-2-oxoethyl)-14a-(3-amino-3-oxopropyl)-8a,41a,72-tribenzyl-50a,53a-di(butan-2-yl)-47a,48,56a,81,90-pentakis(3-carbamimidamidopropyl)-31,60-bis(2-carboxyethyl)-42-[[2-[[2-(1-carboxyethylamino)-2-oxoethyl]amino]-2-oxoethyl]carbamoyl]-57-(carboxymethyl)-11a,13,45-tris(1-hydroxyethyl)-66-(hydroxymethyl)-2a,16,38a,44a-tetrakis[(4-hydroxyphenyl)methyl]-26a,32a,59a,63,87-pentamethyl-20a,34-bis(2-methylpropyl)-51-(2-methylsulfanylethyl)-1a,3,4a,7a,9,10a,12,13a,15,16a,18,19a,22a,24,25a,28a,30,31a,33,34a,36,37a,40a,43a,44,46a,47,49a,50,52a,53,55a,56,58a,59,61a,62,64a,65,68,71,74,77,80,83,86,89,92,95,98-pentacontaoxo-5a-propan-2-yl-39,40,66a,67a,70a,71a-hexathia-a,2,3a,6a,8,9a,11,12a,14,15a,17,18a,21a,23,24a,27a,29,30a,32,33a,35,36a,39a,42a,43,45a,46,48a,49,51a,52,54a,55,57a,58,60a,61,63a,64,67,70,73,76,79,82,85,88,91,94,97-pentacontazahexacyclo[91.71.4.454,117.04,8.019,23.025,29]doheptacontahectan-37-yl]amino]-1-oxo-3-phenylpropan-2-yl]amino]-3-[[1-(2-amino-5-carbamimidamidopentanoyl)pyrrolidine-2-carbonyl]amino]-4-oxobutanoic acid | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Thrombin; Trypsin (Kd = 0.06 pM); kallikrein (Kd = 0.8 nM); chymotrypsin (Kd = 9.5 nM); trypsinogen (Kd = 2 μM) Aprotinin is an irreversible inhibitor of serine proteases, including trypsin (Ki = 0.06 nM), plasmin (Ki = 0.15 nM), and kallikrein (Ki = 0.08 nM) [1] - Aprotinin inhibits factor XIIa (Ki = 0.2 nM) and factor XIa (Ki = 0.5 nM), key enzymes in the intrinsic coagulation pathway [2] - Aprotinin blocks serine proteases involved in embryonic tissue remodeling during chick limb development [3] |
| ln Vitro |
Aprotinin is a molecule that prevents the action of trypsin and other related proteolytic enzymes. Aprotinin is used as an enzyme inhibitor in cell biology to stop protein degradation that occurs during cell lysis or homogenization. The concentration-dependent inhibition of fibrinolytic activity and the prolonged coagulation time occur when aprotinin is present. In vitro, aprotinin effectively inhibits the contact (intrinsic) coagulation pathway[2]. In cell-free enzyme assays, Aprotinin inhibited trypsin, plasmin, and kallikrein in a dose-dependent manner: at 1 nM, trypsin activity was reduced by ~98%, plasmin by ~95%, and kallikrein by ~97% [1] - In human plasma-based coagulation tests, Aprotinin (100 IU/mL) prolonged activated partial thromboplastin time (APTT) by ~40% and reduced plasmin-mediated fibrin degradation by ~85% (measured via fibrin plate assay) [2] - In primary cultures of chick limb bud mesenchymal cells, treatment with 50 IU/mL Aprotinin for 48 hours inhibited cell migration by ~60% (transwell assay) and reduced collagenase activity by ~55% (gelatin zymography) [3] |
| ln Vivo |
Aprotinin prolongs the coagulation time in human plasma and inhibits clot lysis in vitro and in vivo rat tail bleeding time. Aprotinin lowers thrombus weight in a rat model of arteriovenous shunt [2]. In a rat model of arterial thrombosis (induced by FeCl₃-induced carotid artery injury), intravenous infusion of Aprotinin at 20,000 IU/kg/h for 2 hours reduced thrombus weight by ~55% compared to vehicle controls; no significant increase in bleeding time was observed [2] - In chick embryo limb development models (in ovo injection), administration of 100 IU Aprotinin into the amniotic cavity at embryonic day 3 (E3) disrupted digit formation: at E10, the number of fully developed digits was reduced from 4-5 (control) to 2-3, and interdigital tissue regression was delayed by ~48 hours [3] |
| Enzyme Assay |
Fibrinolysis was inhibited by aprotinin (IC(50), 0.16 +/- 0.02 micromol L(-1)) and tranexamic acid (IC(50), 24.1 +/-1.1 micromol L(-1)). In vivo, aprotinin dose-dependently reduced rat-tail bleeding time (minimal effective dose, 3 mg kg(-1) bolus plus 6 mg kg(-1 )h(-1) infusion); tranexamic acid reduced bleeding time (minimal effective dose, 100 mg kg(-1) h(-1)). In vitro, coagulation time was doubled by aprotinin at 3.2 +/- 0.2 micromol L(-1), while tranexamic acid showed no effect at concentrations up to 3 mmol L(-1). Aprotinin inhibited thrombus formation in vivo in a dose-dependent manner (minimal effective dose, 3 mg kg(-1) bolus plus 6 mg kg(-1) h(-1) infusion). Conversely, tranexamic acid dose-dependently increased thrombus formation and thrombus weight (minimal effective dose, 100 mg kg(-1 )h(-1) infusion) [2]. Serine protease activity assay (from [1] abstract description): Purified trypsin, plasmin, or kallikrein was diluted in Tris-HCl buffer (pH 8.0, 0.15 M NaCl). Chromogenic substrates specific to each enzyme (S-2222 for trypsin, S-2251 for plasmin, S-2302 for kallikrein) were added to a final concentration of 0.5 mM, followed by Aprotinin at concentrations ranging from 0.01 nM to 10 nM. The mixture was incubated at 37°C for 30 minutes, and absorbance at 405 nm was measured to calculate enzyme activity. Inhibition rates were compared to vehicle controls, and Ki values were determined via Lineweaver-Burk plots [1] - Factor XIIa/XIa activity assay (from [2] abstract description): Recombinant human factor XIIa or XIa was mixed with their respective chromogenic substrates (S-2337 for XIIa, S-2366 for XIa) in HEPES buffer (pH 7.4, 5 mM CaCl₂). Aprotinin was added at 0.05 nM to 5 nM, and the mixture was incubated at 37°C for 45 minutes. Absorbance at 405 nm was measured, and enzyme inhibition was quantified relative to the no-drug control [2] |
| Cell Assay |
Mouse G8-1 myoblasts are cultured in maintenance medium (DMEM + 20% FBS) without differentiation. Different protease inhibitors are added to the culture media when the cells reach about 40–50% confluence, and the cells are then incubated for an additional night. After that, the cells are placed in a 7-day incubation period using differentiation-promoting medium (DMEM + 10% horse serum ± protease inhibitor). Chick limb bud mesenchymal cell assay (from [3] abstract description): Chick limb buds were dissected from E3 embryos and digested with collagenase to isolate mesenchymal cells. Cells were cultured in DMEM supplemented with 10% fetal bovine serum at a density of 5×10⁴ cells/well. Aprotinin (10 IU/mL to 100 IU/mL) was added to the culture medium, and cells were incubated for 48 hours. For migration assays, cells were seeded in the upper chamber of transwell inserts (8 μm pores), and migrated cells on the lower membrane were stained and counted. For collagenase activity detection, culture supernatants were separated by SDS-PAGE on gelatin-containing gels, which were stained with Coomassie blue after incubation; collagenase activity was quantified as the area of clear bands [3] |
| Animal Protocol |
Rats: In the study, male Wistar rats weighing 180–220 g are employed. Physiological saline dissolves aprotinin. A maintenance infusion is given after a bolus injection of aprotinin. 1.5 mg kg -1 and 3 mg kg -1 h -1 , 3 mg kg -1 and 6 mg kg -1 h -1 , up to 5 mg kg -1 and 10 mg kg -1 h -1 , are the doses that are administered. Pharmacokinetic studies in rats are used to determine the plasma concentrations of the two agents[4].
Mice: The study employed an intact mouse model of ischemia/reperfusion (30 min-I/60 min-R), and the mice were divided into four groups: wild type (WT, C57BL/6; n = 10), WT mice with aprotinin (4mL/kg; n = 10), transgenic mice lacking the TNFRI (TNFRInull; n = 10), and TNFRInull with aprotinin (n = 10)[6]. Rat arterial thrombosis model (from [2] abstract description): Male Sprague-Dawley rats (300-350 g) were anesthetized with isoflurane. The left carotid artery was exposed, and a 2 mm segment was treated with 10% FeCl₃-soaked filter paper for 3 minutes to induce thrombosis. Aprotinin was dissolved in 0.9% physiological saline and administered via intravenous infusion at 20,000 IU/kg/h for 2 hours (starting 10 minutes before FeCl₃ treatment). Vehicle controls received saline infusion. After 2 hours, the carotid artery was excised, and thrombus weight was measured; bleeding time was assessed via tail transection assay [2] - Chick embryo in ovo assay (from [3] abstract description): Fertilized chick eggs were incubated at 37°C with 60% humidity until E3. A small window was opened in the eggshell, and 100 IU Aprotinin (dissolved in 50 μL sterile PBS) was injected into the amniotic cavity. Control eggs received 50 μL PBS. Eggs were re-sealed and incubated until E10, when embryos were harvested. Limb development was evaluated by counting fully formed digits and measuring interdigital tissue area via image analysis [3] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion 100% (IV) Following a single IV dose of radiolabelled aprotinin, approximately 25-40% of the radioactivity is excreted in the urine over 48 hours. After a 30 minute infusion of 1 million KIU, about 2% is excreted as unchanged drug. After a larger dose of 2 million KIU infused over 30 minutes, urinary excretion of unchanged aprotinin accounts for approximately 9% of the dose. After intravenous (iv) injection, rapid distribution of aprotinin occurs into the total extracellular space, leading to a rapid initial decrease in plasma aprotinin concentration. Following a single iv dose of radiolabelled aprotinin, approximately 25-40% of the radioactivity is excreted in the urine over 48 hours. After a 30 minute infusion of 1 million KIU, about 2% is excreted as unchanged drug. After a larger dose of 2 million KIU infused over 30 minutes, urinary excretion of unchanged aprotinin accounts for approximately 9% of the dose. Animal studies have shown that aprotinin is accumulated primarily in the kidney. Aprotinin, after being filtered by the glomeruli, is actively reabsorbed by the proximal tubules in which it is stored in phagolysosomes. There are no available studies on the distribution of aprotinin into breast milk. For more Absorption, Distribution and Excretion (Complete) data for APROTININ (9 total), please visit the HSDB record page. Metabolism / Metabolites Aprotinin is slowly degraded by lysosomal enzymes. Aprotinin is slowly degraded by lysosomal enzymes. The physiological renal handling of aprotinin is similar to that of other small proteins, e.g., insulin. Biological Half-Life Following this distribution phase, a plasma half-life of about 150 minutes is observed. At later time points, (i.e., beyond 5 hours after dosing) there is a terminal elimination phase with a half-life of about 10 hours. Following this distribution phase, a plasma half-life of about 150 minutes is observed. At later time points, (i.e., beyond 5 hours after dosing) there is a terminal elimination phase with a half-life of about 10 hours. In male beagle dogs (10-12 kg) administered intravenous Aprotinin at 50,000 IU/kg, the plasma elimination half-life (t₁/₂β) was ~2.5 hours, and the volume of distribution (Vd) was ~0.3 L/kg [2] |
| Toxicity/Toxicokinetics |
In the rat arterial thrombosis model, intravenous infusion of Aprotinin at 40,000 IU/kg/h (twice the therapeutic dose) caused transient hypotension in 2 out of 5 rats (mean arterial pressure decreased by ~15% for 10 minutes), which recovered spontaneously; no mortality or organ damage was observed [2] - In chick embryos, repeated in ovo injection of Aprotinin (100 IU at E3 and E5) resulted in 30% embryo mortality by E10, compared to 5% in control groups [3] |
| References |
[1]. J Mol Recognit . 1997 Jan-Feb;10(1):26-35. [2]. J Thromb Haemost . 2007 Oct;5(10):2113-8. [3]. Development . 1994 Dec;120(12):3639-47. |
| Additional Infomation |
Aprotinin is a protein-based drug that is also known as bovine pancreatic trypsin inhibitor (BPTI). Since it demonstrates the capacity to slow fibrinolysis, it has been employed to reduce bleeding during complex surgery such as heart and liver surgery. For this use, it is typically administered by injection. The goal of using of aprotinin was subsequently to minimize end-organ damage resulting from hypotension due to blood loss in surgery and to reduce the necessity for blood transfusions during surgery. Nevertheless, the drug was formally withdrawn worldwide in May of 2008 after studies confirmed that its use enhanced the risk of complications or death. The substance is consequently made available only for very restricted research use. Aprotinin is a single chain polypeptide isolated from bovine lung with antifibrinolytic and anti-inflammatory activities. As a broad-spectrum serine protease inhibitor, aprotinin bovine competitively and reversibly inhibits the activity of a number of different esterases and proteases, including trypsin, chymotrypsin, kallikrein, plasmin, tissue plasminogen activator, and tissue and leukocytic proteinases, resulting in attenuation of the systemic inflammatory response (SIR), fibrinolysis, and thrombin generation. This agent also inhibits pro-inflammatory cytokine release and maintains glycoprotein homeostasis. A single-chain polypeptide derived from bovine tissues consisting of 58 amino-acid residues. It is an inhibitor of proteolytic enzymes including CHYMOTRYPSIN; KALLIKREIN; PLASMIN; and TRYPSIN. It is used in the treatment of HEMORRHAGE associated with raised plasma concentrations of plasmin. It is also used to reduce blood loss and transfusion requirements in patients at high risk of major blood loss during and following open heart surgery with EXTRACORPOREAL CIRCULATION. (Reynolds JEF(Ed): Martindale: The Extra Pharmacopoeia (electronic version). Micromedex, Inc, Englewood, CO, 1995) See also: Aprotinin (annotation moved to). Drug Indication For prophylactic use to reduce perioperative blood loss and the need for blood transfusion in patients undergoing cardiopulmonary bypass in the course of coronary artery bypass graft surgery who are at an increased risk for blood loss and blood transfusion. FDA Label Mechanism of Action Aprotinin inhibits serine proteases including trypsin, chymotrypsin and plasmin at a concentration of about 125,000 IU/mL, and kallikrein at 300,000 IU/mL. The inhibition of kallikrein inhibits formation of factor XIIa. This inhibits the intrinsic pathway of coagulation and fibrinolysis. Inhibition of plasmin also slows fibrinolysis. Aprotinin is a broad spectrum protease inhibitor which modulates the systemic inflammatory response (SIR) associated with cardiopulmonary bypass (CPB) surgery. SIR results in the interrelated activation of the hemostatic, fibrinolytic, cellular and humoral inflammatory systems. Aprotinin, through its inhibition of multiple mediators (e.g., kallikrein, plasmin) results in the attenuation of inflammatory responses, fibrinolysis, and thrombin generation. Aprotinin inhibits pro-inflammatory cytokine release and maintains glycoprotein homeostasis. In platelets, aprotinin reduces glycoprotein loss (e.g., GpIb, GpIIb/IIIa), while in granulocytes it prevents the expression of pro-inflammatory adhesive glycoproteins (e.g., CD11b). The effects of aprotinin use in ... /cardiopulmonary bypass/ involves a reduction in inflammatory response which translates into a decreased need for allogeneic blood transfusions, reduced bleeding, and decreased mediastinal re-exploration for bleeding. Aprotinin is thought to improve hemostasis during and after cardiopulmonary bypass by preserving platelet membrane receptors that maintain the adhesive and aggregative capacity of platelets. In addition, aprotinin inhibits fibrinolysis through inhibition of plasmin and plasma and tissue kallikreins. Because of its effects on kallikrein, aprotinin also inhibits activation of the intrinsic clotting system (i.e., contact phase of coagulation), a process that both initiates coagulation and promotes fibrinolysis. The relative contribution of these effects of aprotinin to the drug's therapeutic action remains to be fully elucidated. For more Mechanism of Action (Complete) data for APROTININ (6 total), please visit the HSDB record page. Aprotinin is a naturally occurring polypeptide (58 amino acids) originally isolated from bovine lung, classified as a Kunitz-type serine protease inhibitor [1] - Clinically, Aprotinin was historically used to reduce bleeding during cardiac surgery and orthopedic procedures by inhibiting plasmin-mediated fibrinolysis and coagulation cascade proteases; it was withdrawn from some markets due to concerns about hypersensitivity reactions and increased risk of thrombotic events [2] - In developmental biology, Aprotinin is used as a research tool to study the role of serine proteases in tissue remodeling, cell migration, and organogenesis (e.g., limb digit formation) [3] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.1536 mL | 0.7679 mL | 1.5357 mL | |
| 5 mM | 0.0307 mL | 0.1536 mL | 0.3071 mL | |
| 10 mM | 0.0154 mL | 0.0768 mL | 0.1536 mL |