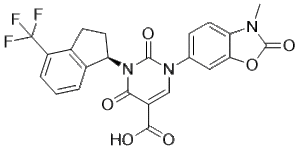
Fulacimstat (also known as BAY1142524) is a novel, potent and orally bioavailable chymase inhibitor that has IC50 values of 4 and 3 nM for the chymase enzymes in hamsters and humans, respectively. Fulacimstat is being developed as a first-in-class treatment for left-ventricular dysfunction following myocardial infarction. It has the potential to be used as an antifibrotic drug candidate.
Physicochemical Properties
| Molecular Formula | C23H16F3N3O6 |
| Molecular Weight | 487.38485622406 |
| Exact Mass | 487.099 |
| Elemental Analysis | C, 56.68; H, 3.31; F, 11.69; N, 8.62; O, 19.70 |
| CAS # | 1488354-15-9 |
| Related CAS # | 1488354-15-9 |
| PubChem CID | 91758792 |
| Appearance | Solid powder |
| Density | 1.6±0.1 g/cm3 |
| Boiling Point | 605.8±65.0 °C at 760 mmHg |
| Flash Point | 320.2±34.3 °C |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.650 |
| LogP | 1.97 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 35 |
| Complexity | 984 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | FC(C1=CC=CC2=C1CC[C@H]2N1C(C(C(=O)O)=CN(C2=CC=C3C(=C2)OC(N3C)=O)C1=O)=O)(F)F |
| InChi Key | JDARDSVOVYVQST-MRXNPFEDSA-N |
| InChi Code | InChI=1S/C23H16F3N3O6/c1-27-17-7-5-11(9-18(17)35-22(27)34)28-10-14(20(31)32)19(30)29(21(28)33)16-8-6-12-13(16)3-2-4-15(12)23(24,25)26/h2-5,7,9-10,16H,6,8H2,1H3,(H,31,32)/t16-/m1/s1 |
| Chemical Name | 1-(3-methyl-2-oxo-1,3-benzoxazol-6-yl)-2,4-dioxo-3-[(1R)-4-(trifluoromethyl)-2,3-dihydro-1H-inden-1-yl]pyrimidine-5-carboxylic acid |
| Synonyms | BAY1142524; BAY 1142524; BAY-1142524; Fulacimstat |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
human chymase enzyme (IC50 = 4 nM); hamster chymase enzyme (IC50 = 3 nM) Fulacimstat inhibits the chymase enzyme in humans and hamsters, with IC50 values of 4 nM and 3 nM, respectively[1][2]. |
| ln Vitro |
Fulacimstat inhibits the chymase enzyme in humans and hamsters, with IC50 values of 4 nM and 3 nM, respectively[1][2]. Fulacimstat (BAY 1142524) is an orally available chymase inhibitor that blocks the generation of profibrotic chymase-dependent factors such as angiotensin II, endothelin-1, and transforming growth factor-β1 in vitro with nanomolar half-maximal inhibitory concentration (IC₅₀). [2] |
| ln Vivo |
In hamsters, isoprenaline causes 24.4±1.8% cardiac fibrosis, which is dose-dependently decreased by enalapril (17.7±1.5% at 20 mg/kg) and Fulacimstat (16.4±1.2%, 12.4 ± 1.3%, and 10.9±1.4% at 1, 3 and 10 mg/kg, respectively). The hearts of hamsters exhibit reduced relaxation and contractility, along with an increase in end diastolic pressure, four weeks after MI. Without affecting blood pressure or heart rate, fulacimstat at 10 mg/kg significantly reduced the end diastolic pressure (13.2±1.4 mmHg) in comparison to placebo (19.3±2 mmHg). Additionally, Fulacimstat treatment improves the cardiac response to adrenergic stimulation and reduces the fibrotic area[1]. In hamsters, Fulacimstat (BAY 1142524) reduced isoprenaline-induced cardiac fibrosis and attenuated the deterioration of heart function after myocardial infarction (MI). In dogs with left-ventricular dysfunction (LVD) induced by intracoronary microembolization, Fulacimstat increased left-ventricular ejection fraction. These effects were observed at therapeutic exposures without affecting blood pressure or heart rate in the examined animal models. [2] |
| Animal Protocol |
Male Syrian hamstersC 10 mg/kg p.o. |
| ADME/Pharmacokinetics |
The pharmacokinetics of Fulacimstat (BAY 1142524) were evaluated in three phase I studies in healthy male volunteers. Absorption: Following oral administration, Fulacimstat was absorbed with a median time to peak plasma concentration (tₘₐₓ) of 0.5-0.75 hours for the liquid service formulation (LSF) and 1.0-2.98 hours for immediate-release (IR) tablets under fasting conditions. [2] Exposure and Dose Proportionality: Systemic exposure, measured as area under the plasma concentration-time curve (AUC) and maximum plasma concentration (Cₘₐₓ), appeared to increase in proportion to the dose after single (1-200 mg) and multiple (5-50 mg twice daily) dosing. The dose-normalized AUC (AUC/D) was independent of the dose level. [2] Elimination: Fulacimstat was eliminated from plasma with a terminal half-life (t½) ranging from 6.84 to 12.0 hours after administration of LSF or IR tablets. The interindividual range was 3.75-21.3 hours. [2] Food Effect: A high-fat, high-calorie meal had little effect on AUC for a 5 mg IR tablet (4.22% decrease) but reduced Cₘₐₓ by 23.5% and prolonged tₘₐₓ by approximately 2.01 hours. The 90% confidence interval for the AUC ratio (88.9%-103%) was within the 80%-125% bioequivalence range. [2] Accumulation: The accumulation of Fulacimstat was low after multiple dosing for 5 consecutive days. The accumulation ratios for Cₘₐₓ (RACₘₐₓ) and AUC over the dosing interval (RAAUC) were 1.13 and 1.22 for 100 mg once daily, and ranged from 1.14-1.61 and 1.26-1.65 for twice-daily regimens (5-50 mg BID), respectively. The pharmacokinetics were linear over time. [2] Bioavailability: The relative bioavailability of a 5 mg IR tablet compared to a 5 mg oral solution (LSF) under fasting conditions was approximately 106% for AUC (90%CI: 98.4-114) and 79.4% for Cₘₐₓ. [2] Excretion: Renal excretion of unchanged drug accounted for approximately 7.90% to 17.6% of the dose after single-dose administration and 19.5% to 23.7% after repeated twice-daily dosing, with high interindividual variability. The mean renal clearance was approximately 0.320 L/h. [2] |
| Toxicity/Toxicokinetics |
Fulacimstat (BAY 1142524) was safe and well-tolerated in healthy male volunteers at all examined doses (single doses up to 200 mg, multiple doses up to 100 mg once daily or 50 mg twice daily). The incidence and intensity of treatment-emergent adverse events (TEAEs) were similar between the Fulacimstat and placebo groups. Headache was the most common TEAE. There were no serious adverse events related to the drug. No dose relationship was observed for TEAEs. [2] Fulacimstat had no effects on systolic/diastolic blood pressure or heart rate compared with placebo after single or repeated dosing. [2] There was no evidence of drug-induced changes in laboratory parameters (hematology, serum chemistry, coagulation, urinalysis). Transient elevations in some laboratory values occurred but were not judged to be related to study drug intake. [2] |
| References |
[1]. Abstract 13624: A Novel Chymase Inhibitor BAY 1142524 Reduces Fibrosis and Improves Cardiac Function After Myocardial Infarction in Hamster. Circulation. 2018;136:A13624. [2]. Pharmacokinetics, Safety, and Tolerability of the Novel Chymase Inhibitor BAY 1142524 in Healthy Male Volunteers. Clin Pharmacol Drug Dev. 2018 Jun 7. |
| Additional Infomation |
Fulacimstat is under investigation in clinical trial NCT02452515 (A Single-blind Pilot Study to Investigate Safety and Tolerability of the Chymase Inhibitor BAY1142524 in Clinically Stable Patients With Left-ventricular Dysfunction). Fulacimstat (BAY 1142524) is a novel, orally available chymase inhibitor being developed as a first-in-class treatment for left-ventricular dysfunction after myocardial infarction. Its mechanism of action involves local inhibition of chymase, a serine protease secreted upon tissue injury, thereby blocking the generation of profibrotic factors like angiotensin II and attenuating adverse cardiac remodeling. [2] Based on human pharmacokinetic data and preclinical pharmacodynamic models, doses of 4 mg twice daily or 20 mg once daily are predicted to provide therapeutic plasma concentrations (unbound levels above the human chymase IC₅₀ of 4 nM) for nearly 24 hours. [2] The absence of blood pressure and heart rate effects supports the planned combination of Fulacimstat with hemodynamically active standard-of-care medications (e.g., ACE inhibitors, β-blockers) in post-MI patients. [2] The pharmacokinetic profile allows for once-daily dosing, which may improve patient adherence in chronic therapy. [2] |
Solubility Data
| Solubility (In Vitro) | DMSO: 50~97 mg/mL (102.6~199.0 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.13 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 0.5 mg/mL (1.03 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 0.5 mg/mL (1.03 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0518 mL | 10.2589 mL | 20.5179 mL | |
| 5 mM | 0.4104 mL | 2.0518 mL | 4.1036 mL | |
| 10 mM | 0.2052 mL | 1.0259 mL | 2.0518 mL |