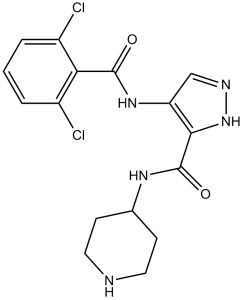
AT7519 (AT-7519; AT 7519) is an orally bioavailable and potent inhibitor of multiple CDKs (cyclin-dependent kinases) with potential antitumor activity. With IC50s of 10-210 nM, it inhibits CDK1, 2, 4, 6, and 9. It has minimal activity against CDK7 and is less effective against CDK3. In order to exert its effects, AT-7519 binds to CDKs specifically and inhibits them. This can lead to apoptosis induction, cell cycle arrest, and inhibition of tumor cell proliferation.
Physicochemical Properties
| Molecular Formula | C16H17CL2N5O2 | |
| Molecular Weight | 382.24 | |
| Exact Mass | 381.075 | |
| Elemental Analysis | C, 50.28; H, 4.48; Cl, 18.55; N, 18.32; O, 8.37 | |
| CAS # | 844442-38-2 | |
| Related CAS # | AT7519 TFA;1431697-85-6;AT7519 Hydrochloride;902135-91-5 | |
| PubChem CID | 11338033 | |
| Appearance | White to off-white solid powder | |
| Density | 1.5±0.1 g/cm3 | |
| Boiling Point | 586.0±50.0 °C at 760 mmHg | |
| Flash Point | 308.2±30.1 °C | |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C | |
| Index of Refraction | 1.654 | |
| LogP | 0.95 | |
| Hydrogen Bond Donor Count | 4 | |
| Hydrogen Bond Acceptor Count | 4 | |
| Rotatable Bond Count | 4 | |
| Heavy Atom Count | 25 | |
| Complexity | 479 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | O=C(C1C(Cl)=CC=CC=1Cl)NC1C(C(NC2CCNCC2)=O)=NNC=1 |
|
| InChi Key | OVPNQJVDAFNBDN-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C16H17Cl2N5O2/c17-10-2-1-3-11(18)13(10)15(24)22-12-8-20-23-14(12)16(25)21-9-4-6-19-7-5-9/h1-3,8-9,19H,4-7H2,(H,20,23)(H,21,25)(H,22,24) | |
| Chemical Name | 4-[(2,6-dichlorobenzoyl)amino]-N-piperidin-4-yl-1H-pyrazole-5-carboxamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | CDK9/Cyclin T (IC50 = 10 nM); CDK5/p35 (IC50 = 13 nM); cdk2/cyclin A (IC50 = 47 nM); Cdk4/cyclin D1 (IC50 = 100 nM); cdk6/cyclin D3 (IC50 = 170 nM); Cdk1/cyclin B (IC50 = 210 nM); CDK7/Cyclin H/MAT1 (IC50 = 2400 nM); GSK3β (IC50 = 89 nM) | |
| ln Vitro |
|
|
| ln Vivo |
|
|
| Enzyme Assay | Kinase assays using radiometric filter binding are conducted for CDK1, CDK2, and GSK3-β. The format of the assays is ELISA for CDKs 4 and 6, and DELFIA for CDK 5. The relevant CDK and 0.12 μg/mL Histone H1 are incubated for 2 or 4 hours, respectively, in 20 mM MOPS, pH 7.2, 25 mM β-glycerophosphate, 5 mM EDTA, 15 mM MgCl2, 1 mM sodium orthovanadate, 1 mM DTT, 0.1 mg/mL BSA, 45 μM ATP (0.78 Ci/mmol), and various concentrations of AT7519. In order to test GSK3-β, the appropriate enzyme and 5 μM glycogen synthase peptide 2 are added, and the mixture is incubated for three hours at 10 mM MOPS pH 7.0, 0.1 mg/mL BSA, 0.001% Brij-35, 0.5% glycerol, 0.2 mM EDTA, 10 mM MgCl2, 0.01% β-mercaptoethanol, 15 μM ATP (2.31 Ci/mmol), all of which are tested. Millipore MAPH filter plates are used to filter the assay reactions after an excess of orthophosphoric acid is added to stop the reaction. After that, the plates are cleaned, scintillant is added, and radioactivity is determined using a Packard TopCount scintillation counting device. For a duration of 30 minutes, CDK5, CDK5/p35, 1μM of a biotinylated Histone H1 peptide (Biotin-PKTPKKAKKL), pH 7.5, 25 mM Tris-HCl, 0.025% Brij-35, 0.1 mg/mL BSA, 1 mM DTT, 15 μM ATP, and various concentrations of AT7519 are incubated. Time-resolved fluorescence at λex=335nm, λem=620nm is used to stop the assay reactions using EDTA, transfer the mixture to Neutravidin-coated plates, and quantify the phosphorylated peptide using a rabbit phospho-cdk1 substrate polyclonal antibody and DELFIA europium-labelled anti-rabbit IgG secondary antibody. Plates are coated with GST-pRb769-921 and blocked with Superblock for the CDK 4 and 6 assays. In order to initiate the reaction, ATP is added to CDK4 or 6. The incubation conditions include 15 mM MgCl2, 50 mM HEPES, pH 7.4, 1 mM DTT, 1 mM EGTA, pH 8.0, 0.02% Triton X-100, 2.5% DMSO, and various concentrations of AT7519. Reactions are halted by adding 0.5 M EDTA pH 8.0 after 30 minutes. After that, plates are cleaned and incubated for one hour with a secondary antibody (alkaline phosphatase linked anti-rabbit) and another hour with the primary antibody (anti-p-Rb Serine 780) diluted in Superblock. Fluorescence is measured on a Spectramax Gemini plate reader at excitation of 450 nm and emission of 580 nm after plates are developed using the Attophos system. Using GraphPad Prism software, IC50 values are computed from replicate curves in every scenario. | |
| Cell Assay | The 3-(4,5-dimethylthiazol-2-yl)-2,5 diphenyl tetrasodium bromide (MTT) dye absorbance is used to measure the effects of AT7519 on the viability of MM cell lines, primary MM cells, and PBMNCs. Triiodothymidine uptake (3H-TdR) is used to quantify DNA synthesis. MM cells (2–3 × 104 cells/well) are cultured for 24 or 48 hours at 37°C in 96-well culture plates with media and varying concentrations of AT7519, recombinant IL-6 (10 ng/mL), or IGF-1 (50 ng/mL). 3H-TdR incorporation is then measured. | |
| Animal Protocol | In order to assess the in vivo anti-MM activity of AT7519, 5×106 MM.1S cells are subcutaneously injected into male SCID mice using 100 μL of serum-free RPMI 1640 medium. Mice are treated intraperitoneally (IP) with vehicle or AT7519 dissolved in 0.9% saline solution when tumors are detectable. Ten mice in the first group receive a daily dose of 15 mg/kg for two weeks, while the second group receives a daily dose of 15 mg/kg three times a week for four weeks in a row. The carrier is given to the control group separately at the same time. Tumor volume is calculated using the formula V= 0.5 a × b2, where a represents the tumor's long diameter and b its short diameter. Tumor size is measured every other day in two dimensions using calipers. When a tumor is ulcerated or grows to a size of 2 cm3, the animal is killed. From the first day of treatment until death, survival and tumor growth are assessed. | |
| References |
[1]. AT7519, A novel small molecule multi-cyclin-dependent kinase inhibitor, induces apoptosis in multiple myeloma via GSK-3beta activation and RNA polymerase II inhibition. Oncogene. 2010 Apr 22;29(16):2325-36. [2]. Biological characterization of AT7519, a small-molecule inhibitor of cyclin-dependent kinases, in human tumor cell lines. Biological characterization of AT7519, a small-molecule inhibitor of cyclin-dependent kinases, in human tumor cell lines. [3]. AT7519, a cyclin-dependent kinase inhibitor, exerts its effects by transcriptional inhibition in leukemia cell lines and patient samples. Mol Cancer Ther. 2010 Apr;9(4):920-8. |
|
| Additional Infomation |
4-(2,6-dichlorobenzamido)-N-(piperidin-4-yl)-pyrazole-3-carboxamide is a member of the class of pryrazoles that is 4-amino-1H-pyrazole-3-carboxylic acid in which the primary amino group has been acylated by a 2,6-dichlorobenzoyl group and in which the carboxylic acid has been converted into a carboxamide by formal condensation with the primary amino group of 4-aminopiperidine. It has a role as an EC 2.7.11.22 (cyclin-dependent kinase) inhibitor and an antineoplastic agent. It is a secondary carboxamide, a member of pyrazoles, a dichlorobenzene and a member of piperidines. AT7519 is a selective inhibitor of certain Cyclin Dependent Kinases (CDKs) leading to tumour regression. It is developed by Astex for the treatment of solid tumours and haematological malignancies. CDK Inhibitor AT7519 is an orally bioavailable small molecule with potential antineoplastic activity. AT7519M selectively binds to and inhibits cyclin dependent kinases (CDKs), which may result in cell cycle arrest, induction of apoptosis, and inhibition of tumor cell proliferation. CDKs are serine/threonine kinases involved in regulation of the cell cycle and may be overexpressed in some types of cancer cells. Drug Indication Investigated for use/treatment in leukemia (unspecified), lymphoma (unspecified), myelodysplastic syndrome, and solid tumors. Mechanism of Action AT7519 is a selective inhibitor of certain Cyclin Dependent Kinases (CDKs) leading to tumour regression. |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.54 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.54 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (6.54 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 30% Propylene glycol , 5% Tween 80 , 65% D5W: 30 mg/mL (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6162 mL | 13.0808 mL | 26.1616 mL | |
| 5 mM | 0.5232 mL | 2.6162 mL | 5.2323 mL | |
| 10 mM | 0.2616 mL | 1.3081 mL | 2.6162 mL |