AMG 333 is a novel, potent and highly selective TRPM8 antagonist with an IC50 of 13 nM and 20 nM for hTRPM8 and rTRPM8, respectively. AMG-333can be potentially used for the treatment of migraine. In vitro toxicology profiling showed that AMG-333 was selective over several other TRP channels (IC50 TRPV1, V3, V4 > 20 μM; TRPA1 > 40 μM) and had no hits in off-target-activity panels (CEREP, 144 targets at 10 μM, POC > 45%; Ambit kinase, 100 at 1 μM, POC > 50%). AMG-333 was well-tolerated in 28 day rat and dog preclinical safety studies and advanced to Phase 1 human clinical trials.
Physicochemical Properties
| Molecular Formula | C20H12F5N3O4 |
| Molecular Weight | 453.319002151489 |
| Exact Mass | 453.074 |
| CAS # | 1416799-28-4 |
| PubChem CID | 71144018 |
| Appearance | White to off-white solid powder |
| LogP | 3.5 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 32 |
| Complexity | 668 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | FC1=CC=CN=C1C(C1C=CC(=C(C=1)F)OC(F)(F)F)NC(C1C=CC(C(=O)O)=CN=1)=O |
| InChi Key | QEBYISWYMFIXOZ-INIZCTEOSA-N |
| InChi Code | InChI=1S/C20H12F5N3O4/c21-12-2-1-7-26-17(12)16(10-4-6-15(13(22)8-10)32-20(23,24)25)28-18(29)14-5-3-11(9-27-14)19(30)31/h1-9,16H,(H,28,29)(H,30,31)/t16-/m0/s1 |
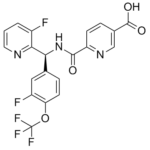
| Chemical Name | (S)-6-(((3-fluoro-4-(trifluoromethoxy)phenyl)(3-fluoropyridin-2-yl)methyl)carbamoyl)nicotinic acid |
| Synonyms | AMG-333; AMG 333; AMG333 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
AMG 333 targets transient receptor potential melastatin 8 (TRPM8) ion channel (human TRPM8: Ki = 6.9 nM for [³H]icilin binding [1] ; IC50 = 14 nM for menthol-induced calcium influx inhibition in TRPM8-expressing HEK293 cells [1] ; IC50 = 22 nM for icilin-induced TRPM8 activation inhibition [1] ; no significant binding/inhibition of other TRP channels (TRPV1, TRPA1, TRPM3) or voltage-gated ion channels (Nav1.7, Cav2.2) with IC50 > 1000 nM [1] ) |
| ln Vitro |
1. AMG 333 acted as a potent and selective competitive antagonist of human TRPM8, inhibiting [³H]icilin binding to TRPM8 with a Ki of 6.9 nM; in TRPM8-stably transfected HEK293 cells, it dose-dependently blocked menthol-induced calcium influx (IC50 = 14 nM) and icilin-induced calcium mobilization (IC50 = 22 nM) via fluorometric calcium imaging [1] 2. Patch-clamp electrophysiology experiments in TRPM8-expressing HEK293 cells showed that AMG 333 (0.01-1 μM) inhibited menthol-induced TRPM8 current with an IC50 of 18 nM, and completely blocked channel activity at 100 nM [1] 3. AMG 333 exhibited high selectivity for TRPM8 over other ion channels: no inhibition of TRPV1 (capsaicin-induced), TRPA1 (allyl isothiocyanate-induced), or TRPM3 (pregnenolone sulfate-induced) at concentrations up to 1 μM; negligible activity against voltage-gated sodium (Nav1.7) and calcium (Cav2.2) channels (IC50 > 10 μM) [1] 4. In human trigeminal ganglion (TG) neurons, AMG 333 (100 nM) inhibited cold (10°C)-induced calcium influx in TRPM8-positive neurons by 85%, with no effect on TRPM8-negative TG neurons [1] 5. AMG 333 showed no cytotoxicity in HEK293 or primary human TG neurons at concentrations up to 10 μM, with cell viability >95% after 24-hour treatment (MTT assay) [1] |
| ln Vivo |
1. In a rat nitroglycerin (NTG)-induced migraine model, oral administration of AMG 333 (1, 3, 10 mg/kg) dose-dependently reduced NTG-induced trigeminal nerve activation (measured by c-Fos expression in trigeminal nucleus caudalis (TNC)) by 35%, 62%, and 80% respectively; the 10 mg/kg dose also decreased NTG-induced mechanical allodynia (von Frey test) by 75% at 2 hours post-dosing [1] 2. In a mouse cold-induced facial pain model, AMG 333 (3, 10 mg/kg PO) inhibited cold-evoked facial grooming behavior by 40% and 65% respectively, confirming TRPM8-mediated nociception blockade [1] 3. In cynomolgus monkeys, AMG 333 (1, 3 mg/kg IV) reduced trigeminal ganglion (TG) neuronal firing rate by 58% and 72% respectively, as measured by in vivo extracellular electrophysiology; the effect persisted for 4 hours post-dosing [1] 4. In rats, AMG 333 (10 mg/kg PO) showed good brain penetration, with a TNC/plasma ratio of 0.8 at 1 hour post-dosing, and achieved CNS concentrations above the in vitro IC50 for TRPM8 inhibition (14 nM) for up to 6 hours [1] |
| Enzyme Assay |
1. TRPM8 radioligand binding assay: Membrane preparations from HEK293 cells stably expressing human TRPM8 were incubated with [³H]icilin (0.5 nM) and serial dilutions of AMG 333 (0.001-1 μM) in binding buffer at 4°C for 120 minutes; bound and free ligand were separated by vacuum filtration through glass fiber filters; radioactivity of the filter-bound fraction was measured by liquid scintillation counting, and Ki values were calculated using the Cheng-Prusoff equation from competition binding curves [1] 2. Surface Plasmon Resonance (SPR) binding assay: Recombinant human TRPM8 extracellular domain protein was immobilized on a CM5 sensor chip; serial dilutions of AMG 333 (0.001-1 μM) were injected over the chip at a flow rate of 30 μL/min at 25°C; real-time binding responses (resonance units, RU) were recorded, and kinetic parameters (ka, kd, KD) were determined using a 1:1 binding model to confirm direct interaction with TRPM8 [1] 3. TRPM8 calcium flux functional assay: HEK293 cells stably expressing human TRPM8 were loaded with a fluorescent calcium indicator (Fura-2 AM) for 45 minutes at 37°C; AMG 333 (0.001-1 μM) was added and incubated for 20 minutes, followed by stimulation with menthol (100 μM) or icilin (1 μM); intracellular calcium concentrations were measured by ratiometric fluorometry (excitation 340/380 nm, emission 510 nm), and IC50 values for inhibition were calculated from dose-response curves [1] |
| Cell Assay |
1. TRPM8-expressing HEK293 cell calcium influx assay: HEK293 cells stably transfected with human TRPM8 were seeded in 96-well black-walled plates at a density of 1×10⁴ cells/well and cultured to 80% confluency; cells were loaded with Fura-2 AM (5 μM) in HBSS buffer containing 0.1% BSA for 45 minutes at 37°C, with gentle shaking every 15 minutes; excess dye was removed by washing, and AMG 333 at serial dilutions (0.001-1 μM) was added to each well; after 20 minutes of incubation, menthol (100 μM) or icilin (1 μM) was added to trigger TRPM8 activation, and fluorescence ratios (340/380 nm) were recorded every 2 seconds for 5 minutes using a microplate reader; data were normalized to the maximum response of vehicle-treated cells to calculate IC50 values [1] 2. Human trigeminal ganglion neuron calcium imaging assay: Primary human TG neurons were isolated and plated on poly-L-lysine-coated coverslips; neurons were loaded with Fluo-4 AM (4 μM) for 30 minutes at 37°C, then washed and incubated with AMG 333 (0.01-1 μM) for 15 minutes; cold stimulation (10°C) or menthol (100 μM) was applied to activate TRPM8, and fluorescent signals (excitation 488 nm, emission 525 nm) were captured by confocal microscopy; the percentage of TRPM8-positive neurons (responding to cold/menthol) and the magnitude of calcium influx were quantified, and inhibition by AMG 333 was calculated [1] 3. Cell viability assay: HEK293 cells and primary human TG neurons were seeded in 96-well plates at 5×10³ cells/well and treated with AMG 333 (0.01-10 μM) for 24 hours at 37°C with 5% CO₂; MTT reagent (0.5 mg/mL) was added and incubated for 4 hours, then the medium was removed and DMSO was added to dissolve formazan crystals; absorbance was measured at 570 nm, and cell viability was calculated relative to vehicle-treated controls [1] |
| Animal Protocol |
1. Rat nitroglycerin (NTG)-induced migraine model: Male Sprague-Dawley rats (250-300 g) were acclimated to the laboratory for 7 days; AMG 333 was formulated in 0.5% methylcellulose + 0.1% Tween 80 and administered orally via gavage at 1, 3, or 10 mg/kg (volume: 5 mL/kg) 1 hour before NTG injection (10 mg/kg IP); 4 hours after NTG administration, rats were euthanized, and trigeminal nucleus caudalis (TNC) tissues were collected for c-Fos immunohistochemistry; mechanical allodynia was assessed using von Frey filaments (0.4-15 g) at 1, 2, and 4 hours post-NTG injection, and the withdrawal threshold was recorded [1] 2. Mouse cold-induced facial pain model: Female C57BL/6 mice (20-25 g) were treated with AMG 333 (3, 10 mg/kg PO) or vehicle 30 minutes before exposure to a cold stimulus (4°C metal plate applied to the facial area for 1 minute); facial grooming behavior (number of grooming episodes per minute) was recorded for 5 minutes post-stimulation, and the inhibition rate was calculated relative to vehicle controls [1] 3. Cynomolgus monkey trigeminal ganglion electrophysiology model: Adult cynomolgus monkeys (3-5 kg) were anesthetized with isoflurane, and a microelectrode was implanted into the trigeminal ganglion (TG) for extracellular single-unit recording; AMG 333 was dissolved in 5% dextrose solution and administered intravenously at 1 or 3 mg/kg; neuronal firing rates were recorded continuously for 6 hours post-dosing, and the percentage reduction in firing rate (compared to baseline) was calculated at 1, 2, 4, and 6 hours [1] 4. Rat pharmacokinetic (PK) tissue distribution study: Male Sprague-Dawley rats were administered AMG 333 (10 mg/kg PO or 1 mg/kg IV); blood samples were collected at 0.25, 0.5, 1, 2, 4, 6, and 8 hours post-dosing, and plasma was separated by centrifugation; brain tissues (TNC, cerebral cortex) were harvested at the same time points, homogenized in PBS, and protein was precipitated with acetonitrile; AMG 333 concentrations in plasma and tissue homogenates were quantified by LC-MS/MS, and PK parameters (Cmax, Tmax, AUC, tissue/plasma ratio) were calculated [1] |
| ADME/Pharmacokinetics |
1. In male Sprague-Dawley rats, oral administration of AMG 333 (10 mg/kg) resulted in a peak plasma concentration (Cmax) of 320 nM at 1 hour (Tmax), oral bioavailability (F) of 78%, terminal half-life (t1/2) of 3.6 hours, volume of distribution (Vd) of 1.2 L/kg, and total clearance (CL) of 0.3 L/h/kg [1] 2. Intravenous administration of AMG 333 (1 mg/kg) in rats showed a t1/2 of 2.8 hours, Vd of 1.5 L/kg, and CL of 0.4 L/h/kg; the drug exhibited good CNS penetration, with a trigeminal nucleus caudalis (TNC)/plasma ratio of 0.8 and cerebral cortex/plasma ratio of 0.6 at 1 hour post-dosing [1] 3. In human liver microsomes, AMG 333 was primarily metabolized by CYP3A4 (65%) and CYP2C9 (25%) via oxidative dealkylation and hydroxylation; the major metabolite (M1) was inactive against TRPM8 (IC50 > 1 μM) [1] 4. In cynomolgus monkeys, oral AMG 333 (3 mg/kg) had a Cmax of 280 nM (Tmax = 1.5 hours), t1/2 = 4.2 hours, and F = 65%; steady-state concentrations were achieved after 5 days of once-daily dosing, with no accumulation observed [1] 5. Less than 10% of the parent drug was excreted unchanged in rat urine and feces over 48 hours; the majority of the dose (80%) was excreted as metabolites, with fecal excretion (60%) exceeding urinary excretion (20%) [1] |
| Toxicity/Toxicokinetics |
1. AMG 333 exhibited high plasma protein binding in rat, monkey, and human plasma (92%, 94%, and 96%, respectively) [1] 2. In vitro CYP450 inhibition assays demonstrated that AMG 333 did not inhibit CYP1A2, CYP2C19, CYP2D6, or CYP2E1 at concentrations up to 10 μM; it weakly inhibited CYP3A4 (IC50 = 8.5 μM) and CYP2C9 (IC50 = 9.2 μM), indicating a low risk of drug-drug interactions [1] 3. Acute toxicity studies in CD-1 mice revealed no mortality or overt toxicity at oral doses up to 1000 mg/kg or intravenous doses up to 100 mg/kg; subchronic toxicity (28-day oral dosing at 30, 100 mg/kg/day in rats) showed no significant changes in body weight, food intake, or liver/kidney function markers (ALT, AST, BUN, creatinine) [1] 4. In cynomolgus monkeys treated with AMG 333 (10 mg/kg/day PO for 28 days), no adverse effects on cardiovascular parameters (heart rate, blood pressure) or neurological function were observed; histopathological examination of major organs (liver, kidney, brain, trigeminal ganglion) showed no treatment-related lesions [1] |
| References |
[1]. Discovery of TRPM8 Antagonist ( S)-6-(((3-Fluoro-4-(trifluoromethoxy)phenyl)(3-fluoropyridin-2-yl)methyl)carbamoyl)nicotinic Acid (AMG 333), a Clinical Candidate for the Treatment of Migraine. J Med Chem. 2018 Sep 27;61(18):8186-8201. |
| Additional Infomation |
1. AMG 333 is a first-in-class, potent, and selective TRPM8 antagonist developed as a clinical candidate for the acute treatment of migraine [1] 2. The mechanism of action of AMG 333 involves competitive binding to the TRPM8 ion channel pore region, blocking cold/menthol-induced channel activation and inhibiting trigeminal nerve sensitization—a key pathological step in migraine pathogenesis [1] 3. AMG 333 entered Phase I clinical trials for migraine in healthy volunteers, demonstrating favorable safety, tolerability, and pharmacokinetic profiles, with no dose-limiting toxicities observed at doses up to 600 mg [1] 4. Preclinical data show that AMG 333 is effective in multiple migraine models by targeting the trigeminal-vascular system, and its high selectivity for TRPM8 minimizes off-target effects (e.g., no impact on thermoregulation at therapeutic doses) [1] 5. AMG 333 is also being investigated for the treatment of other TRPM8-mediated pain conditions (e.g., cold allodynia in neuropathic pain), with preclinical efficacy observed in rodent models of chemotherapy-induced peripheral neuropathy [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 125 mg/mL (~275.74 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.17 mg/mL (4.79 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 21.7 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.17 mg/mL (4.79 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 21.7 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.17 mg/mL (4.79 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 21.7 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2059 mL | 11.0297 mL | 22.0595 mL | |
| 5 mM | 0.4412 mL | 2.2059 mL | 4.4119 mL | |
| 10 mM | 0.2206 mL | 1.1030 mL | 2.2059 mL |