ALW-II-41-27 is a potent Eph receptor tyrosine kinase inhibitor with IC50 of 11 nM for Eph2. Tumor growth in vivo was inhibited by ALW-II-41-27'sinhibitionof EPHA2, which also reduced the survival and proliferation of erlotinib-resistant tumor cells. The viability of cells that had developed resistance to the third-generation EGFR TKI AZD9291 was likewise effectively decreased by ALW-II-41-27. All of these findings point to an involvement of EPHA2 in the preservation of cell survival in EGFR-mutant, TKI-resistant lung cancer and suggest that EPHA2 might be a valuable target for therapeutic intervention in these tumors.
Physicochemical Properties
| Molecular Formula | C32H32F3N5O2S |
| Molecular Weight | 607.6890 |
| Exact Mass | 607.223 |
| Elemental Analysis | C, 63.25; H, 5.31; F, 9.38; N, 11.52; O, 5.27; S, 5.28 |
| CAS # | 1186206-79-0 |
| Related CAS # | 1186206-79-0 |
| PubChem CID | 42628503 |
| Appearance | White to light yellow solid powder |
| LogP | 7.423 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 43 |
| Complexity | 933 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | S1C([H])=C([H])C([H])=C1C1C([H])=NC([H])=C(C=1[H])C(N([H])C1=C(C([H])([H])[H])C([H])=C([H])C(=C1[H])C(N([H])C1C([H])=C([H])C(=C(C(F)(F)F)C=1[H])C([H])([H])N1C([H])([H])C([H])([H])N(C([H])([H])C([H])([H])[H])C([H])([H])C1([H])[H])=O)=O |
| InChi Key | HYWXBDQAYLPMIX-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C32H32F3N5O2S/c1-3-39-10-12-40(13-11-39)20-23-8-9-26(17-27(23)32(33,34)35)37-30(41)22-7-6-21(2)28(16-22)38-31(42)25-15-24(18-36-19-25)29-5-4-14-43-29/h4-9,14-19H,3,10-13,20H2,1-2H3,(H,37,41)(H,38,42) |
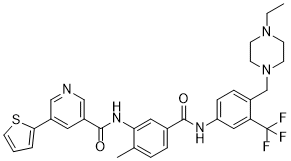
| Chemical Name | N-[5-[[4-[(4-ethylpiperazin-1-yl)methyl]-3-(trifluoromethyl)phenyl]carbamoyl]-2-methylphenyl]-5-thiophen-2-ylpyridine-3-carboxamide |
| Synonyms | ALWII-41-27; ALW-II-41-27; ALW II-41-27; ALW II-4127; ALW-II-4127; ALWII-4127 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | EphA2 (Kd = 12 nM) |
| ln Vitro | ALW-II-41-27 inhibits Ba/F3 cells transformed with Tel fusions of EphA3, Kit, Fms, KDR, FLT1, FGR, Src, Lyn, Bmx, and Bcr-Abl with an EC50 below 500 nM. Bcr-Abl and ALW-II-41-27 show cross-reactivity. Inhibiting b-raf, CSF1R, DDR1, DDR2, EphA2, EphA5, EphA8, EphB1, EphB2, EphB3, Frk, Kit, Lck, p38α, p38β, PDGFRα, PDGFRβ, Raf1, and numerous other kinases, ALW-II-41-27 shows how the addition of a thiophene group can significantly affect kinase selectivity[1]. |
| Cell Assay | In order to ascertain the function of EPHA2, myogenic precursors were exposed to ALW-II-41-27 (0.5 μM) for a duration of 12 hours in GM, after which they underwent myogenic differentiation. |
| References |
[1]. Discovery and structural analysis of Eph receptor tyrosine kinase inhibitors. Bioorganic & Medicinal Chemistry Letters (2009), 19(15), 4467-4470. [2]. Targeting EphA2 impairs cell cycle progression and growth of basal-like/triple-negative breast cancers. Oncogene. 2017 Jun 5. |
Solubility Data
| Solubility (In Vitro) |
DMSO: ~100 mg/mL (~164.6 mM) Ethanol: ~100 mg/mL (~164.6 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.11 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.5 mg/mL (4.11 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (4.11 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6456 mL | 8.2279 mL | 16.4558 mL | |
| 5 mM | 0.3291 mL | 1.6456 mL | 3.2912 mL | |
| 10 mM | 0.1646 mL | 0.8228 mL | 1.6456 mL |