Physicochemical Properties
| Molecular Formula | C24H36O5 |
| Molecular Weight | 404.54 |
| CAS # | 517-33-9 |
| Appearance | Typically exists as solids at room temperature |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 582.2±50.0 °C at 760 mmHg |
| Flash Point | 320.0±26.6 °C |
| Vapour Pressure | 0.0±3.7 mmHg at 25°C |
| Index of Refraction | 1.543 |
| LogP | 2.22 |
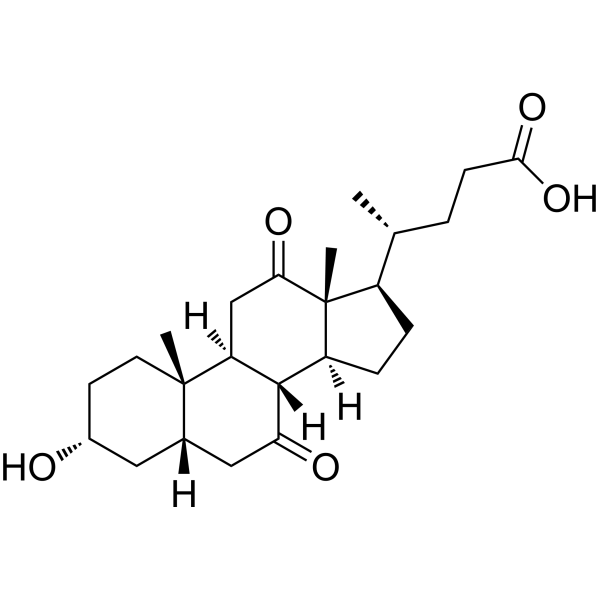
| SMILES | CC(CCC(=O)O)C1CCC2C1(C(=O)CC3C2C(=O)CC4C3(CCC(C4)O)C)C |
| Synonyms | 7,12-Dioxolithocholic acid; 3-Hydroxy-7,12-diketocholanoic acid; 517-33-9; 7,12-Dioxolithocholic acid; 7,12-Diketolithocholic acid; 3alpha-Hydroxy-7,12-diketo-5beta-cholanoic acid; 3alpha-Hydroxy-7,12-dioxo-5beta-cholan-24-oic Acid; (4R)-4-[(3R,5S,8R,9S,10S,13R,14S,17R)-3-hydroxy-10,13-dimethyl-7,12-dioxo-2,3,4,5,6,8,9,11,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl]pentanoic acid; 5beta-Cholanic acid-3alpha-ol-7,12-dione; 3-Hydroxy-7,12-diketocholanoic acid; 7,12-Diketo-LCA |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Lithocholic acid metabolite |
| ln Vitro | 7,12-dioxolithocholic acid is a bile acid that is lithocholic acid carrying two additional oxo substituents at positions 7 and 12. It has a role as a bacterial metabolite. It is a bile acid, a monohydroxy-5beta-cholanic acid, an oxo-5beta-cholanic acid, a 7-oxo steroid, a 12-oxo steroid and a 3alpha-hydroxy steroid. It is functionally related to a lithocholic acid. It is a conjugate acid of a 7,12-dioxolithocholate. |
| References |
[1]. Lan K, et al., Key Role for the 12-Hydroxy Group in the Negative Ion Fragmentation of Unconjugated C24 Bile Acids. Anal Chem. 2016 Jul 19;88(14):7041-8. |
| Additional Infomation | Host-gut microbial interactions contribute to human health and disease states and an important manifestation resulting from this cometabolism is a vast diversity of bile acids (BAs). There is increasing interest in using BAs as biomarkers to assess the health status of individuals and, therefore, an increased need for their accurate separation and identification. In this study, the negative ion fragmentation behaviors of C24 BAs were investigated by UPLC-ESI-QTOF-MS. The step-by-step fragmentation analysis revealed a distinct fragmentation mechanism for the unconjugated BAs containing a 12-hydroxyl group. The unconjugated BAs lacking 12-hydroxylation fragmented via dehydration and dehydrogenation. In contrast, the 12-hydroxylated ones, such as deoxycholic acid (DCA) and cholic acid (CA), employed dissociation routes including dehydration, loss of carbon monoxide or carbon dioxide, and dehydrogenation. All fragmentations of the 12-hydroxylated unconjugated BAs, characterized by means of stable isotope labeled standards, were associated with the rotation of the carboxylate side chain and the subsequent rearrangements accompanied by proton transfer between 12-hydroxyl and 24-carboxyl groups. Compared to DCA, CA underwent further cleavages of the steroid skeleton. Accordingly, the effects of stereochemistry on the fragmentation pattern of CA were investigated using its stereoisomers. Based on the knowledge gained from the fragmentation analysis, a novel BA, 3β,7β,12α-trihydroxy-5β-cholanic acid, was identified in the postprandial urine samples of patients with nonalcoholic steatohepatitis. The analyses used in this study may contribute to a better understanding of the chemical diversity of BAs and the molecular basis of human liver diseases that involve BA synthesis, transport, and metabolism.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4719 mL | 12.3597 mL | 24.7194 mL | |
| 5 mM | 0.4944 mL | 2.4719 mL | 4.9439 mL | |
| 10 mM | 0.2472 mL | 1.2360 mL | 2.4719 mL |