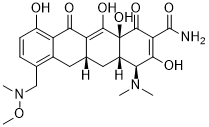
Sarecycline (formerly known as WC-3035 and P005672; trade name: Seysara) is the first narrow spectrum tetracycline-class antibiotic being developed for acne treatment. In Oct 2018, FDA approved Sarecycline for the treatment of inflammatory lesions of non-nodular moderate to severe acne vulgaris in patients 9 years of age and older. In addition to exhibiting activity against important skin/soft tissue pathogens, sarecycline exhibits targeted antibacterial activity against clinical isolates of Cutibacterium acnes In the current study, sarecycline was 16 to 32-fold less active than broad spectrum tetracyclines-such as minocycline and doxycycline-against aerobic Gram-negative bacilli associated with normal human intestinal microbiome. Also, reduced activity against Escherichia coliwas observed in vivo in a murine septicemia model with PD50 values at >40 mg/kg and 5.72 mg/kg for sarecycline and doxycycline, respectively. Sarecycline was also 4 to 8-fold less active against representative anaerobic bacteria that also comprise normal human intestinal microbiome. Additionally, sarecycline displayed a low propensity for resistance development in C. acnes strains, with spontaneous mutation frequencies of 10-10 at 4 to 8-times the MIC, similar to minocycline and vancomycin. When tested against Gram-positive pathogens with defined tetracycline resistance mechanisms, sarecycline was more active than tetracycline against the tet(K) and tet(M) strains, with MIC ranging from 0.125 to 1.0 ug/mL and 8 ug/mL, respectively, compared with 16 to 64 ug/mL and 64 ug/mL for tetracycline, respectively. However, sarecycline activity in the tet(K) and tet(M) strains were decreased compared to wildtype, which demonstrated MIC ranging from 0.06 to 0.25 ug/mL, though not as pronounced as tetracycline.
Physicochemical Properties
| Molecular Formula | C24H29N3O8 |
| Molecular Weight | 487.51 |
| Exact Mass | 487.195 |
| CAS # | 1035654-66-0 |
| Related CAS # | Sarecycline hydrochloride;1035979-44-2 |
| PubChem CID | 54681908 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 1.106 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 35 |
| Complexity | 1010 |
| Defined Atom Stereocenter Count | 4 |
| SMILES | CN([C@H]1[C@@H]2C[C@@H]3CC4=C(C(C3=C(O)[C@]2(O)C(C(C(N)=O)=C1O)=O)=O)C(O)=CC=C4CN(OC)C)C |
| InChi Key | AYUMVPHUMFKFPJ-SBAJWEJLSA-N |
| InChi Code | InChI=1S/C24H29N3O8/c1-26(2)18-13-8-11-7-12-10(9-27(3)35-4)5-6-14(28)16(12)19(29)15(11)21(31)24(13,34)22(32)17(20(18)30)23(25)33/h5-6,11,13,18,28-29,32,34H,7-9H2,1-4H3,(H2,25,33)/t11-,13-,18-,24-/m0/s1 |
| Chemical Name | (4S,4aS,5aR,12aS)-4-(dimethylamino)-3,10,12,12a-tetrahydroxy-7-((methoxy(methyl)amino)methyl)-1,11-dioxo-1,4,4a,5,5a,6,11,12a-octahydrotetracene-2-carboxamide |
| Synonyms | WC-3035; WC 3035; WC3035; P005672; P-005672; P 005672; P005672; trade name: Seysara |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | MIC values for 55 clinical strains of cosmetic acne on stick culture medium are 0.5~16 µg/ml, 0.5 µg/ml for MIC50, and 4 µg/ml for MIC90 [1]. The drug sarecycline exhibits efficacy against macrolide organisms [1]. The degrading aerobic Gram-null bacteria are not much affected by sarecycline hydrochloride [2]. |
| ln Vivo | In a neutropenic mouse infection paradigm, seromycin hydrochloride (0.33–9 mg/kg; intravenously) has demonstrated strong efficacy against Staphylococcus aureus [1]. The ability of serocycline to inhibit E. aureus (PD50=0.25 mg/kg) in an abdominal systemic infection model in CD-1 mice, but not E. Coli at the maximum concentration (>40 mg/kg). The impact is obscure [6]. |
| Animal Protocol |
Animal/Disease Models: Carrageenan-induced footpad edema male Sprague Dawley rat model [6] Doses: 1 mg/kg, 5 mg/kg. kg; intraperitoneal; injection single dose) Anti-inflammatory in the Sprague Dawley rat model activity[6]. ,10 mg/kg, 25 mg/kg, 50 mg/kg, 75 mg/kg, 100 mg/kg, 150 mg/kg Route of Administration: intraperitoneal (ip) injection; single dose. Before injecting a sterile 1 mg/0.1 mL carrageenan solution into the plantar part of the right hind paw. Experimental Results: Compared to baseline, inflammation was diminished to 55.7% and 53.1% at the 75 mg/kg and 100 mg/kg doses, respectively. Animal/Disease Models: Male Sprague Dawley rat model of carrageenan-induced footpad edema [6] Doses: 0.33, 1, 3 or 9 mg/kg Route of Administration: intravenous (iv) (iv)injection Experimental Results: At one dose, thigh bacterial load was diminished 2-log10 is comparable to doxycycline, with ED50 of 8.23 and 8.32 mg/kg respectively. |
| References |
[1]. Zhanel G, et al. Microbiological Profile of Sarecycline, a Novel Targeted Spectrum Tetracycline for the Treatment of Acne Vulgaris. Antimicrob Agents Chemother. 2018 Dec 21;63(1):e01297-18. [2]. Moore AY, et al. Sarecycline: a narrow spectrum tetracycline for the treatment of moderate-to-severe acne vulgaris. Future Microbiol. 2019 Sep;14(14):1235-1242. [3]. Batool Z, et al. Sarecycline interferes with tRNA accommodation and tethers mRNA to the 70S ribosome. Proc Natl Acad Sci U S A. 2020 Aug 25;117(34):20530-20537. [4]. Butler MS, et al. Antibiotics in the clinical pipeline in 2013. J Antibiot (Tokyo). 2013 Oct;66(10):571-91. [5]. Bunick CG, et al. Antibacterial Mechanisms and Efficacy of Sarecycline in Animal Models of Infection and Inflammation. Antibiotics (Basel). 2021 Apr 15;10(4):439. |
| Additional Infomation |
Sarecycline is a Tetracycline-class Drug. The mechanism of action of sarecycline is as a P-Glycoprotein Inhibitor. See also: Sarecycline (annotation moved to); Sarecycline Hydrochloride (annotation moved to). |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.13 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.13 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0512 mL | 10.2562 mL | 20.5124 mL | |
| 5 mM | 0.4102 mL | 2.0512 mL | 4.1025 mL | |
| 10 mM | 0.2051 mL | 1.0256 mL | 2.0512 mL |