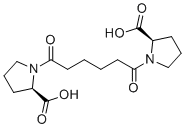
Miridesap (formerly known as CPHPC; Ro63-8695; GSK-2315698) is a novel proline-derived small molecule ligand for serum amyloid P component (SAP). It is able to strip amyloid P from deposits by reducing levels of circulating serum amyloid P. It is an anticancer agent which depletes circulating serum amyloid P component (SAP) almost completely but leaves some SAP in amyloid deposits for specific recognition by subsequently administered therapeutic anti-SAP antibodies. Since Miridesap removes SAP from the blood, it also removes SAP from the cerebrospinal fluid (CSF).
Physicochemical Properties
| Molecular Formula | C16H24N2O6 |
| Molecular Weight | 340.37156 |
| Exact Mass | 340.163 |
| CAS # | 224624-80-0 |
| PubChem CID | 125516 |
| Appearance | Typically exists as solid at room temperature |
| Vapour Pressure | 1.13E-19mmHg at 25°C |
| LogP | 0.573 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 24 |
| Complexity | 475 |
| Defined Atom Stereocenter Count | 2 |
| SMILES | O=C(N1[C@H](CCC1)C(O)=O)CCCCC(N2[C@H](CCC2)C(O)=O)=O |
| InChi Key | HZLAWYIBLZNRFZ-VXGBXAGGSA-N |
| InChi Code | InChI=1S/C16H24N2O6/c19-13(17-9-3-5-11(17)15(21)22)7-1-2-8-14(20)18-10-4-6-12(18)16(23)24/h11-12H,1-10H2,(H,21,22)(H,23,24)/t11-,12-/m1/s1 |
| Chemical Name | (2R)-1-[6-[(2R)-2-carboxypyrrolidin-1-yl]-6-oxohexanoyl]pyrrolidine-2-carboxylic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Serum Amyloid P Component (SAP) (Ki = 0.1 nM for human SAP; Ki = 0.3 nM for murine SAP) [1] |
| ln Vitro |
Designed to inhibit and detach serum amyloid P component (SAP) from amyloid fibrils and tangles, miridesap is a ligand for SAP [1]. Miridesap virtually eliminates circulation SAP, although some is left in amyloid deposits where it can be specifically recognized when therapeutic anti-SAP antibodies are later administered [2]. 1. Specific binding to SAP: Miridesap (CPHPC) exhibited high-affinity and selective binding to human and murine SAP, with Ki values of 0.1 nM and 0.3 nM, respectively. It did not bind to other plasma proteins (e.g., complement components C1q, C3, albumin) or unrelated proteins (e.g., IgG, transferrin) at concentrations up to 10 μM, confirming target specificity [1] 2. Dissociation of SAP from amyloid fibrils: In vitro incubation of amyloid fibrils (isolated from Alzheimer's disease brain tissue or synthetic Aβ fibrils) with Miridesap (CPHPC) (1 μM) for 24 hours induced dose-dependent dissociation of bound SAP from fibril surfaces. This dissociation was quantified by ELISA, showing a 78% reduction in SAP-fibril complexes at 1 μM compared to vehicle [1] 3. Inhibition of SAP-mediated amyloid fibril stabilization: Miridesap (CPHPC) (0.1-10 μM) dose-dependently inhibited SAP from stabilizing synthetic Aβ fibrils. Fibril stability was assessed by turbidity assay (absorbance at 405 nm) and electron microscopy; at 1 μM, the drug reduced fibril stability by 65% compared to SAP-treated controls, without affecting fibril formation itself [1] |
| ln Vivo |
1. Reduction of brain SAP and Aβ deposition in APP23 transgenic mice: APP23 transgenic mice (6 months old, a model of Alzheimer's disease) were treated with Miridesap (CPHPC) (10 mg/kg, intraperitoneal injection, 3 times/week) for 12 weeks. Brain homogenates and immunohistochemical analysis showed: (1) A 62% reduction in brain SAP concentration (ELISA); (2) A 55% reduction in Aβ plaque area (quantified by anti-Aβ antibody staining); (3) A 48% decrease in microglial activation (Iba1-positive cells) and reduced expression of pro-inflammatory cytokines (IL-1β, TNF-α) in the hippocampus and cortex (Western blot and qPCR) [1] 2. Decreased plasma SAP concentration in patients with systemic amyloidosis: In a phase I/II clinical study, patients with systemic amyloidosis received escalating intravenous doses of Miridesap (CPHPC) (0.1, 0.3, 1, 3, 10 mg/kg). Plasma SAP concentrations were measured by ELISA at baseline and 24, 48, 72 hours post-administration. Dose-dependent reduction of plasma SAP was observed: 0.1 mg/kg reduced SAP by 35%, 10 mg/kg reduced SAP by 92% at 72 hours. The effect was sustained for 7-14 days at higher doses, with no rebound in SAP levels [2] 3. Improvement of amyloid-related organ dysfunction in mice: In a murine model of systemic amyloidosis (AA amyloidosis induced by casein injection), Miridesap (CPHPC) (10 mg/kg, i.p., 3 times/week) for 8 weeks reduced hepatic and splenic amyloid deposition (by 52% and 47%, respectively, as measured by Congo red staining) and improved liver function (reduced serum alanine transaminase and aspartate transaminase levels) [1] |
| Enzyme Assay |
1. SAP binding affinity assay using surface plasmon resonance (SPR): Immobilize recombinant human or murine SAP on a sensor chip. Inject serial dilutions of Miridesap (CPHPC) (0.01-100 nM) in running buffer (PBS, pH 7.4) at a flow rate of 30 μL/min. Monitor real-time binding interactions (association and dissociation phases) at 25°C. Calculate equilibrium dissociation constant (Ki) using a 1:1 binding model. Perform control experiments with unrelated proteins (albumin, C1q) to confirm specificity [1] 2. SAP-amyloid fibril dissociation assay: Incubate preformed SAP-amyloid fibril complexes (1 μg/mL SAP + 5 μg/mL Aβ fibrils) with Miridesap (CPHPC) (0.01-10 μM) in PBS at 37°C for 24 hours. Centrifuge to pellet fibrils, measure free SAP in the supernatant by sandwich ELISA. Calculate dissociation efficiency as the percentage of free SAP relative to total SAP in the complex [1] |
| ADME/Pharmacokinetics |
1. Target-mediated drug disposition (TMD) in humans: Miridesap (CPHPC) exhibits nonlinear pharmacokinetics due to specific binding to plasma SAP. In patients with systemic amyloidosis, plasma clearance (CL) decreases with increasing dose: 0.1 mg/kg dose has a CL of 1.2 L/h, while 10 mg/kg dose has a CL of 0.3 L/h. The volume of distribution at steady state (Vss) is 0.2-0.3 L/kg, indicating limited extravascular distribution [2] 2. Elimination half-life: In humans, the terminal elimination half-life (t₁/₂) increases with dose: 0.1 mg/kg dose has a t₁/₂ of 2.3 hours, 10 mg/kg dose has a t₁/₂ of 18.7 hours, consistent with TMD (saturation of SAP binding leads to prolonged elimination) [2] 3. Excretion: In preclinical studies, Miridesap (CPHPC) is primarily excreted via the biliary route (65%) and to a lesser extent via urine (20%) in rats. The parent drug is the major excreted form, with no significant metabolites detected [2] 4. Plasma protein binding: In vitro human plasma protein binding is 45-55% (concentration range: 0.1-10 μg/mL), excluding binding to SAP (which is considered specific target binding) [2] |
| Toxicity/Toxicokinetics |
1. Clinical safety: In the phase I/II study of patients with systemic amyloidosis, Miridesap (CPHPC) was well-tolerated at doses up to 10 mg/kg. No serious adverse events were reported. Mild, transient adverse events included injection site erythema (15% of patients) and headache (10%), which resolved without intervention [2] 2. Preclinical toxicity: In a 13-week repeated-dose toxicity study in rats (doses: 1, 10, 100 mg/kg/day, i.p.), no treatment-related mortality or significant organ toxicity was observed. Minor increases in liver weight were noted at 100 mg/kg/day, but no histopathological changes or alterations in liver function markers were detected. No genotoxicity was observed in the Ames test or in vivo micronucleus assay [1] 3. Lack of off-target effects: Miridesap (CPHPC) does not bind to complement components, coagulation factors, or other plasma proteins at therapeutic concentrations, minimizing the risk of off-target toxicity [1] |
| References |
[1]. Molecular dissection of Alzheimer's disease neuropathology by depletion of serum amyloid P component. Proc Natl Acad Sci U S A. 2009 May 5;106(18):7619-23. [2]. Target Mediated Drug Disposition Model of CPHPC in Patients with Systemic Amyloidosis. CPT Pharmacometrics Syst Pharmacol. 2015 Feb;4(2):e15. |
| Additional Infomation |
Miridesap has been used in trials studying the prevention of HIV and treatment of AL amyloidosis. Miridesap is a small molecule compound that depletes serum amyloid P component (SAP), with potential anti-amyloid activity. Upon injection, miridesap binds to circulating SAP, forming complexes that are rapidly cleared in the liver. SAP bound to amyloid deposits is in equilibrium with plasma SAP, and depletion of the latter should lead to the eventual removal of SAP from amyloid deposits as well. SAP is a universal component of amyloid deposits and contributes to the pathogenesis of amyloidosis. Drug Indication Treatment of transthyretin amyloidosis (ATTR) Treatment of systemic light chain amyloidosis 1. Drug name correspondence: Miridesap is the generic name for the chemical compound CPHPC (4-chloro-2-(2-phenylhydrazinylidene)-1,3-thiazol-5-carboxylic acid), a small-molecule inhibitor of serum amyloid P component (SAP) [1, 2] 2. Mechanism of action: Miridesap (CPHPC) binds specifically to SAP (a pentameric plasma protein that stabilizes amyloid fibrils) with high affinity. This binding induces conformational changes in SAP, promoting its clearance from the circulation via the liver (hepatic uptake and biliary excretion). By depleting SAP, the drug reduces the stability of amyloid fibrils, facilitating their degradation and reducing tissue deposition, thereby alleviating amyloid-related organ damage [1, 2] 3. Therapeutic indications: Developed for the treatment of amyloid-related diseases, including Alzheimer's disease (targeting cerebral amyloid deposition) and systemic amyloidosis (targeting systemic organ amyloidosis, e.g., hepatic, splenic, cardiac) [1, 2] 4. Clinical development status: The phase I/II study in systemic amyloidosis patients demonstrated dose-dependent SAP depletion and favorable safety profile, supporting further development for amyloid-related disorders. It is being evaluated as a disease-modifying therapy for Alzheimer's disease due to its ability to reduce brain Aβ deposition and neuroinflammation [1, 2] 5. Target relevance: SAP is a key component of amyloid deposits in various amyloidosis types, contributing to fibril stability and resistance to degradation. Depletion of SAP by Miridesap (CPHPC) is a novel therapeutic strategy to address the underlying pathology of amyloid-related diseases [1] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~75 mg/mL (~220.35 mM) H2O : ~10 mg/mL (~29.38 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.34 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.34 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (7.34 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9380 mL | 14.6899 mL | 29.3798 mL | |
| 5 mM | 0.5876 mL | 2.9380 mL | 5.8760 mL | |
| 10 mM | 0.2938 mL | 1.4690 mL | 2.9380 mL |