Physicochemical Properties
| Molecular Formula | C20H24N10O13P2 |
| Molecular Weight | 674.417 |
| Exact Mass | 674.1 |
| Elemental Analysis | C, 35.62; H, 3.59; N, 20.77; O, 30.84; P, 9.19 |
| CAS # | 849214-04-6 |
| Related CAS # | cGAMP disodium;2407516-83-8;cGAMP diammonium; 849214-04-6 (free acid) |
| PubChem CID | 135471108 |
| Appearance | White to yellow solid powder |
| Density | 2.6±0.1 g/cm3 |
| Index of Refraction | 2.071 |
| Source | Endogenous Metabolite |
| LogP | -5.9 |
| Hydrogen Bond Donor Count | 7 |
| Hydrogen Bond Acceptor Count | 19 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 45 |
| Complexity | 1300 |
| Defined Atom Stereocenter Count | 8 |
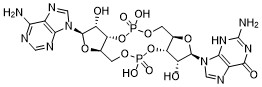
| SMILES | O[C@@H]1[C@@H]2OP(OC[C@H]3O[C@@H](N4C=NC5C(=NC=NC4=5)N)[C@H](O)[C@@H]3OP(OC[C@H]2O[C@H]1N1C=NC2C(N=C(NC1=2)N)=O)(O)=O)(O)=O |
| InChi Key | RFCBNSCSPXMEBK-INFSMZHSSA-N |
| InChi Code | InChI=1S/C20H24N10O13P2/c21-14-8-15(24-3-23-14)29(4-25-8)18-10(31)12-6(40-18)1-38-45(36,37)43-13-7(2-39-44(34,35)42-12)41-19(11(13)32)30-5-26-9-16(30)27-20(22)28-17(9)33/h3-7,10-13,18-19,31-32H,1-2H2,(H,34,35)(H,36,37)(H2,21,23,24)(H3,22,27,28,33)/t6-,7-,10-,11-,12-,13-,18-,19-/m1/s1 |
| Chemical Name | 2-amino-9-((2R,3R,3aS,7aR,9R,10R,10aS,14aR)-9-(6-amino-9H-purin-9-yl)-3,5,10,12-tetrahydroxy-5,12-dioxidooctahydro-2H,7H-difuro[3,2-d:3',2'-j][1,3,7,9]tetraoxa[2,8]diphosphacyclododecin-2-yl)-3,9-dihydro-6H-purin-6-one |
| Synonyms | 3′3′-cGAMP; Cyclic AMP-GMP; c-GMP-AMP; Cgamp; 849214-04-6; c-GMP-AMP; 3',5'-cyclic GMP-AMP; CHEMBL4449584; 3',3'-cGAMP; 2-amino-9-[(1S,6R,8R,9R,10S,15R,17R,18R)-17-(6-aminopurin-9-yl)-3,9,12,18-tetrahydroxy-3,12-dioxo-2,4,7,11,13,16-hexaoxa-3lambda5,12lambda5-diphosphatricyclo[13.3.0.06,10]octadecan-8-yl]-1H-purin-6-one; 3'3'-Cyclic guanosine monophosphate-adenosine monophosphate; cyclic GMP-AMP; cGAMP; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Endogenous Metabolite; second messenger; STING/stimulator of interferon genes |
| ln Vitro | Mouse splenocytes' capacity for antigen-specific proliferation is enhanced by cGAMP [2]. In vitro, cGAMP directly stimulates human and mouse dendritic cells [2]. Patient fibroblasts exhibit enhanced IFNB1 transcription with cGAMP stimulation, but not transcription of genes encoding TNF, IL6, or interleukin 1 (IL1) [3]. Type I IFN is secreted and an antiviral state is induced when cGAMP activates the receptor STING, which is located in the endoplasmic reticulum (ER) [4]. |
| ln Vivo |
cGAMP promotes the production of antigen-specific cytokines in spleen cells of immunized mice [2]. The recently discovered mammalian enzyme cyclic GMP-AMP synthase produces cyclic GMP-AMP (cGAMP) after being activated by pathogen-derived cytosolic double stranded DNA. The product can stimulate STING-dependent interferon type I signaling. Here, we explore the efficacy of cGAMP as a mucosal adjuvant in mice. In this study, researchers show that cGAMP can enhance the adaptive immune response to the model antigen ovalbumin. It promotes antigen specific IgG and a balanced Th1/Th2 lymphocyte response in immunized mice. A characteristic of the cGAMP-induced immune response is the slightly reduced induction of interleukin-17 as a hallmark of Th17 activity – a distinct feature that is not observed with other cyclic di-nucleotide adjuvants. We further characterize the innate immune stimulation activity in vitro on murine bone marrow-derived dendritic cells and human dendritic cells. The observed results suggest the consideration of cGAMP as a candidate mucosal adjuvant for human vaccines[2]. |
| Enzyme Assay | Cytosolic DNA induces type I interferons and other cytokines that are important for antimicrobial defense but can also result in autoimmunity. This DNA signaling pathway requires the adaptor protein STING and the transcription factor IRF3, but the mechanism of DNA sensing is unclear. We found that mammalian cytosolic extracts synthesized cyclic guanosine monophosphate-adenosine monophosphate (cyclic GMP-AMP, or cGAMP) in vitro from adenosine triphosphate and guanosine triphosphate in the presence of DNA but not RNA. DNA transfection or DNA virus infection of mammalian cells also triggered cGAMP production. cGAMP bound to STING, leading to the activation of IRF3 and induction of interferon-β. Thus, cGAMP functions as an endogenous second messenger in metazoans and triggers interferon production in response to cytosolic DNA[1]. |
| Cell Assay |
In vitro stimulation of primary cells[2] The culture medium of primary cells was supplemented with 5 µg/ml (murine cells) or 60 µg/ml (human cells) of c-di-AMP or cGAMP or left without additive. Cells were incubated for 24 h at 37°C. Scrape loading[4] HEK STING cells were seeded at a density of 2.5 × 105 cells ml−1 in 96-well plates. After 16 h cGAMP(2′-5′) was added to the medium to a final concentration of 50 μg ml−1. Monolayers of cells were manually wounded by six scratches per well using an 18G needle. Images were acquired after 4–8 h. |
| Animal Protocol |
Animal/Disease Models: 6-8 weeks old female C57BL/6 (H-2b) mice [2] Doses: 5 µg Route of Administration: Nostril mucosa Adjuvant Experimental Results: Ovalbumin (OVA) specific IgA and total IgG and IgG1 and IgG1 titers were higher compared to IgG2c in sera from OVA-immunized mice with cGAMP adjuvant. |
| References |
[1]. Cyclic GMP-AMP is an endogenous second messenger in innate immune signaling by cytosolic DNA. Science. 2013 Feb 15;339(6121):826-30. [2]. Cyclic GMP-AMP displays mucosal adjuvant activity in mice. PLoS One. 2014 Oct 8;9(10):e110150. [3]. Activated STING in a vascular and pulmonary syndrome. N Engl J Med. 2014 Aug 7;371(6):507-18. [4]. Cell intrinsic immunity spreads to bystander cells via the intercellular transfer of cGAMP. Nature. 2013 Nov 28;503(7477):530-4. |
| Additional Infomation | C-GMP-AMP is a cyclic purine dinucleotide that consists of AMP and GMP units cyclised via 3',5'-linkages. It is an adenyl ribonucleotide, a cyclic purine dinucleotide and a guanyl ribonucleotide. It is a conjugate acid of a c-GMP-AMP(2-). |
Solubility Data
| Solubility (In Vitro) | H2O : ~180 mg/mL (~266.90 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.4828 mL | 7.4138 mL | 14.8276 mL | |
| 5 mM | 0.2966 mL | 1.4828 mL | 2.9655 mL | |
| 10 mM | 0.1483 mL | 0.7414 mL | 1.4828 mL |