Physicochemical Properties
| Molecular Formula | C27H28CLN5O9S3 |
| Molecular Weight | 698.18 |
| Exact Mass | 697.074 |
| Elemental Analysis | C, 46.45; H, 4.04; Cl, 5.08; N, 10.03; O, 20.62; S, 13.78 |
| CAS # | 92602-21-6 |
| Related CAS # | 97275-40-6;41952-52-7 92602-21-6 (HCl); |
| PubChem CID | 179696 |
| Appearance | Solid powder |
| LogP | 4.102 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 15 |
| Rotatable Bond Count | 13 |
| Heavy Atom Count | 45 |
| Complexity | 1260 |
| Defined Atom Stereocenter Count | 4 |
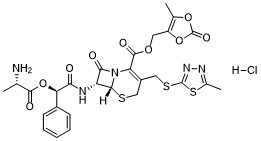
| SMILES | Cl.C[C@@H](C(O[C@@H](C(N[C@@H]1C(=O)N2C(=C(CS[C@H]12)CSC1=NN=C(C)S1)C(OCC1OC(=O)OC=1C)=O)=O)C1C=CC=CC=1)=O)N |
| InChi Key | HFVATKYQUGKLGL-PCQLZLFJSA-N |
| InChi Code | InChI=1S/C27H27N5O9S3.ClH/c1-12(28)24(35)41-20(15-7-5-4-6-8-15)21(33)29-18-22(34)32-19(25(36)38-9-17-13(2)39-27(37)40-17)16(10-42-23(18)32)11-43-26-31-30-14(3)44-26;/h4-8,12,18,20,23H,9-11,28H2,1-3H3,(H,29,33);1H/t12-,18+,20+,23+;/m0./s1 |
| Chemical Name | (5-methyl-2-oxo-1,3-dioxol-4-yl)methyl (6R,7R)-7-((R)-2-((L-alanyl)oxy)-2-phenylacetamido)-3-(((5-methyl-1,3,4-thiadiazol-2-yl)thio)methyl)-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate, hydrochloride |
| Synonyms | Cefcanel daloxate hydrochloride; Cefcanel daloxate HCl; KY-109; KY109; KY 109; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| References | 1: Edwall B, Slettevold L, Thurmann-Nielsen E, Walstad R, Torrång A, Dahl K. Pharmacokinetics of oral cefcanel daloxate hydrochloride in healthy volunteers and patients with various degrees of impaired renal function. J Antimicrob Chemother. 1994 Feb;33(2):281-8. doi: 10.1093/jac/33.2.281. PMID: 8182009. 2: Grunfeld A, Sinclair P, Nicolle L; Canadian Tonsillitis Study Group. Cefcanel daloxate versus penicillin in acute streptococcal pharyngotonsillitis. Can J Infect Dis. 1994 May;5(3):119-24. doi: 10.1155/1994/531572. PMID: 22346486; PMCID: PMC3250823. 3: Holst E, Rollof J, Miörner H. In vitro activities of cefcanel and some other cephalosporins against Pasteurella multocida. Antimicrob Agents Chemother. 1989 Dec;33(12):2142-3. doi: 10.1128/AAC.33.12.2142. PMID: 2619280; PMCID: PMC172837. 4: Bergan T, da Fonseca J. Antibacterial activity of cefcanel. Chemotherapy. 1993 Mar-Apr;39(2):96-104. doi: 10.1159/000239109. PMID: 8458252. 5: Cazzola M. Novel oral cephalosporins. Expert Opin Investig Drugs. 2000 Feb;9(2):237-46. doi: 10.1517/13543784.9.2.237. PMID: 11060674. 6: Nord CE, Lindmark A, Persson I. Comparative antibacterial activity of the new cephalosporin cefcanel against anaerobic bacteria. Eur J Clin Microbiol Infect Dis. 1989 Jun;8(6):550-1. doi: 10.1007/BF01967478. PMID: 2548866. 7: Nicolle LE, Hoepelman AI, Floor M, Verhoef J, Norgard K. Comparison of three days' therapy with cefcanel or amoxicillin for the treatment of acute uncomplicated urinary tract infection. Scand J Infect Dis. 1993;25(5):631-7. doi: 10.3109/00365549309008553. PMID: 8284649. 8: Edwall B, Arvidsson A, Lake-Bakaar D, Lanbeck-Vallén K, Yisak W. Disposition of oral [14C]cefcanel daloxate hydrochloride in healthy male subjects. Drug Metab Dispos. 1993 Jan-Feb;21(1):171-7. PMID: 8095213. 9: Löwdin E, Tornqvist IO, Cars O. The postantibiotic effect of cefcanel on beta-hemolytic streptococci group A in vitro and in vivo. Scand J Infect Dis Suppl. 1990;74:190-4. PMID: 2097708. 10: Chin NX, Gu JW, Neu HC. In vitro activity of cefcanel versus other oral cephalosporins. Eur J Clin Microbiol Infect Dis. 1991 Aug;10(8):676-82. doi: 10.1007/BF01975825. PMID: 1748125. 11: Bergan T, da Fonseca J. Efficacy of cefcanel on staphylocci. Chemotherapy. 1991;37(1):43-9. doi: 10.1159/000238831. PMID: 2013241. 12: Köhler W, Schenk P. Cephalosporinbehandlung der Sinusitis maxillaris [Cephalosporin treatment of maxillary sinusitis]. Laryngorhinootologie. 1995 Jun;74(6):355-60. German. doi: 10.1055/s-2007-997757. PMID: 7662079. 13: Magni L, Lionell C. In vivo efficacy of cefcanel daloxate in comparison with cefaclor. Scand J Infect Dis Suppl. 1990;74:185-9. PMID: 2097707. 14: Lanbeck-Vallén K, Carlqvist J. Determination of cefcanel in plasma and urine by high-performance liquid chromatography using coupled columns, after administration of the new cephalosporin prodrug cefcanel daloxate hydrochloride. J Chromatogr. 1992 Jul 1;578(1):71-6. doi: 10.1016/0378-4347(92)80226-g. PMID: 1400788. 15: Odenholt-Tornqvist I, Löwdin E, Cars O. Pharmacodynamic effects of subinhibitory concentrations of beta-lactam antibiotics in vitro. Antimicrob Agents Chemother. 1991 Sep;35(9):1834-9. doi: 10.1128/AAC.35.9.1834. PMID: 1952854; PMCID: PMC245277. 16: Kakeya N, Nishizawa S, Nishimura K, Yoshimi A, Tamaki S, Mori T, Kitao K. KY-109, a new bifunctional pro-drug of a cephalosporin. Chemistry, physico- chemical and biological properties. J Antibiot (Tokyo). 1985 Mar;38(3):380-9. doi: 10.7164/antibiotics.38.380. PMID: 3839226. 17: Obana Y, Hashizume H, Yoshimi A, Nishino T. In vitro and in vivo antibacterial activity of KY-109, a new orally active cephalosporin. J Antibiot (Tokyo). 1988 Jul;41(7):949-58. doi: 10.7164/antibiotics.41.949. PMID: 3417567. 18: Nishimura K, Nishizawa S, Yoshimi A, Nakamura S, Nishimura M, Kakeya N. KY-109, a new bifunctional prodrug of cephalosporin. II. Mechanism of oral absorption. Chem Pharm Bull (Tokyo). 1988 Jun;36(6):2128-34. doi: 10.1248/cpb.36.2128. PMID: 3240449. 19: Nishizawa S, Yoshimi A, Muro H, Kasai M, Hatano S, Hashizume H, Yamada T, Hashimoto E, Nishimura K, Kakeya N. [Study on orally active cephalosporin pro- drug. Synthesis, oral absorption and antibacterial activity of (5-methyl-2-oxo-1,3-dioxol-4-yl)methyl 7-[D-O-(aminoacyl)mandelamido]-3-[[(5-methyl-1,3,4-thiadiazol -2-yl)-thio]methyl]-3-cephem-4-carboxylates]. Yakugaku Zasshi. 1988 Aug;108(8):745-53. Japanese. doi: 10.1248/yakushi1947.108.8_745. PMID: 3246644. |
Solubility Data
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.4323 mL | 7.1615 mL | 14.3230 mL | |
| 5 mM | 0.2865 mL | 1.4323 mL | 2.8646 mL | |
| 10 mM | 0.1432 mL | 0.7161 mL | 1.4323 mL |