Physicochemical Properties
| Molecular Formula | C₁₈H₃₀O₂ |
| Molecular Weight | 278.43 |
| Exact Mass | 278.224 |
| CAS # | 463-40-1 |
| Related CAS # | 463-40-1 |
| PubChem CID | 5280934 |
| Appearance | Colorless to light yellow liquid |
| Density | 0.9±0.1 g/cm3 |
| Boiling Point | 443.4±0.0 °C at 760 mmHg |
| Melting Point | -11 °C(lit.) |
| Flash Point | 275.7±14.4 °C |
| Vapour Pressure | 0.0±2.3 mmHg at 25°C |
| Index of Refraction | 1.491 |
| LogP | 6.5 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 13 |
| Heavy Atom Count | 20 |
| Complexity | 301 |
| Defined Atom Stereocenter Count | 0 |
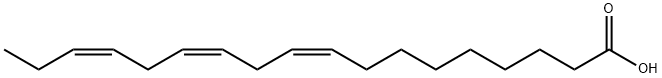
| SMILES | O([H])C(C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])/C(/[H])=C(/[H])\C([H])([H])/C(/[H])=C(/[H])\C([H])([H])/C(/[H])=C(/[H])\C([H])([H])C([H])([H])[H])=O |
| InChi Key | DTOSIQBPPRVQHS-PDBXOOCHSA-N |
| InChi Code | InChI=1S/C18H30O2/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18(19)20/h3-4,6-7,9-10H,2,5,8,11-17H2,1H3,(H,19,20)/b4-3-,7-6-,10-9- |
| Chemical Name | (9Z,12Z,15Z)-octadeca-9,12,15-trienoic acid |
| Synonyms | αLinolenic acid α Linolenic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | PI3K; Akt |
| ln Vitro | α-Linolenic acid converses into the longer chain fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA)[1]. |
| ln Vivo | In mice, α-Linolenic acid (50, 100, or 250 mg/kg; for 10 days) completely inhibits collagen- and adrenaline-induced thrombosis at 250 mg/kg[1]. Rats weighing 250–300 g have less A-V thrombus formation when given 35, 70, or 175 mg/kg of α-Linolenic acid[1]. In rats, α-Linolenic acid (70 or 175 mg/kg) inhibits collagen stimulated platelet aggregation[1]. |
| Animal Protocol |
Mice weighing at 18 ~ 22 g[1] 50, 100, 250 mg/kg For 10 days |
| Toxicity/Toxicokinetics |
Toxicity Summary Alpha Linolenic Acid or ALA is considered an essential fatty acid because it is required for human health, but cannot be synthesized by humans. It is in fact a plant-derived fatty acid. Humans can synthesize other omega-3 fatty acids from ALA, including eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). EPA is a precursor of the series-3 prostaglandins, the series-5 leukotrienes and the series-3 thromboxanes. These eicosanoids have anti-inflammatory and anti-atherogenic properties. ALA metabolites may also inhibit the production of the pro-inflammatory eicosanoids, prostaglandin E2 (PGE2) and leukotriene B4 (LTB4), as well as the pro-inflammatory cytokines, tumor necrosis factor-alpha (TNF-alpha) and interleukin-1 beta (IL-1 beta). Omega-3 fatty acids like ALA and its byproducts can modulate the expression of a number of genes, including those involved with fatty acid metabolism and inflammation. They regulate gene expression through their effects on the activity of transcription factors including NF-kappa B and members of the peroxisome proliferator-activated receptor (PPAR) family. Incorporation of ALA and its metabolites in cell membranes can affect membrane fluidity and may play a role in anti-inflammatory activity, inhibition of platelet aggregation and possibly in anti-proliferative actions of ALA. ALA is first metabolized by delta6 desaturease into steridonic acid. |
| References |
[1]. Anti-thrombotic effects of α-linolenic acid isolated from Zanthoxylum bungeanum Maxim seeds. BMC Complement Altern Med. 2014 Sep 23;14:348. |
| Additional Infomation |
Linolenic acid is a clear colorless liquid. (NTP, 1992) Alpha-linolenic acid is a linolenic acid with cis-double bonds at positions 9, 12 and 15. Shown to have an antithrombotic effect. It has a role as a micronutrient, a nutraceutical and a mouse metabolite. It is an omega-3 fatty acid and a linolenic acid. It is a conjugate acid of an alpha-linolenate and a (9Z,12Z,15Z)-octadeca-9,12,15-trienoate. Alpha-linolenic acid (ALA) is a polyunsaturated omega-3 fatty acid. It is a component of many common vegetable oils and is important to human nutrition. alpha-Linolenic acid is a metabolite found in or produced by Escherichia coli (strain K12, MG1655). Linolenic Acid has been reported in Salvia miltiorrhiza, Calodendrum capense, and other organisms with data available. Linolenic Acid is an essential fatty acid belonging to the omega-3 fatty acids group. It is highly concentrated in certain plant oils and has been reported to inhibit the synthesis of prostaglandin resulting in reduced inflammation and prevention of certain chronic diseases. Alpha-linolenic acid (ALA) is a polyunsaturated omega-3 fatty acid. It is a component of many common vegetable oils and is important to human nutrition. A fatty acid that is found in plants and involved in the formation of prostaglandins. See also: Cod Liver Oil (part of); Evening Primrose Oil (part of); Krill oil (part of) ... View More ... Drug Indication For nutritional supplementation and for treating dietary shortage or imbalance. Mechanism of Action Alpha Linolenic Acid or ALA is considered an essential fatty acid because it is required for human health, but cannot be synthesized by humans. It is in fact a plant-derived fatty acid. Humans can synthesize other omega-3 fatty acids from ALA, including eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). EPA is a precursor of the series-3 prostaglandins, the series-5 leukotrienes and the series-3 thromboxanes. These eicosanoids have anti-inflammatory and anti-atherogenic properties. ALA metabolites may also inhibit the production of the pro-inflammatory eicosanoids, prostaglandin E2 (PGE2) and leukotriene B4 (LTB4), as well as the pro-inflammatory cytokines, tumor necrosis factor-alpha (TNF-alpha) and interleukin-1 beta (IL-1 beta). Omega-3 fatty acids like ALA and its byproducts can modulate the expression of a number of genes, including those involved with fatty acid metabolism and inflammation. They regulate gene expression through their effects on the activity of transcription factors including NF-kappa B and members of the peroxisome proliferator-activated receptor (PPAR) family. Incorporation of ALA and its metabolites in cell membranes can affect membrane fluidity and may play a role in anti-inflammatory activity, inhibition of platelet aggregation and possibly in anti-proliferative actions of ALA. ALA is first metabolized by delta6 desaturease into steridonic acid. Pharmacodynamics Alpha Linolenic Acid (ALA) is an 18-carbon polyunsaturated fatty acid with three double bonds. It is also called an omega-3 fatty acid, and is essential for all mammals. Alpha-linolenic acid (or omega 3 fatty acid) intake can decrease the risk of cardiovascular diseases by 1) preventing arrhythmias that can lead to sudden cardiac death, 2) decreasing the risk of thrombosis (blood clot formation) that can lead to heart attack or stroke, 3) decreasing serum triglyceride levels, 4) slowing the growth of atherosclerotic plaque, 5) improving vascular endothelial function, 6) lowering blood pressure slightly, and 7) decreasing inflammation. ALA deficiencies can lead to visual problems and sensory neuropathy. Scaly and hemorrhagic skin or scalp inflammations may also develop. |
Solubility Data
| Solubility (In Vitro) |
Ethanol: ~100 mg/mL (~359.2 mM) DMSO: ~100 mg/mL (~359.2 mM) H2O: <0.1 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.98 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (8.98 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (8.98 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.5916 mL | 17.9578 mL | 35.9157 mL | |
| 5 mM | 0.7183 mL | 3.5916 mL | 7.1831 mL | |
| 10 mM | 0.3592 mL | 1.7958 mL | 3.5916 mL |