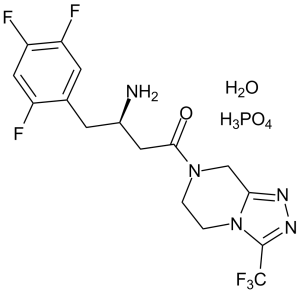
Sitagliptin phosphate (formerly also known as MK-431; trade name Januvia; Xelevia; Janumet) monohydrate, is an oral bioavailable, triazolopyrazine-based, potent inhibitor of DPP-IV (dipeptidyl peptidase-4) with an IC50 of 19 nM in Caco-2 cell extracts. It is an antihyperglycemic and antidiabetic medication.
Physicochemical Properties
| Molecular Formula | C16H20F6N5O6P |
| Molecular Weight | 523.32 |
| Exact Mass | 523.105 |
| Elemental Analysis | C, 36.72; H, 3.85; F, 21.78; N, 13.38; O, 18.34; P, 5.92 |
| CAS # | 654671-77-9 |
| Related CAS # | Sitagliptin;486460-32-6;Sitagliptin phosphate;654671-78-0;(S)-Sitagliptin phosphate;823817-58-9;(Rac)-Sitagliptin;823817-56-7 |
| PubChem CID | 11591741 |
| Appearance | White to off-white solid powder |
| Boiling Point | 529.9ºC at 760 mmHg |
| Flash Point | 274.3ºC |
| LogP | 1.661 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 15 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 34 |
| Complexity | 616 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | [H]O[H].O=C(N1CC2=NN=C(C(F)(F)F)N2CC1)C[C@H](N)CC3=CC(F)=C(F)C=C3F.O=P(O)(O)O |
| InChi Key | GQPYTJVDPQTBQC-KLQYNRQASA-N |
| InChi Code | InChI=1S/C16H15F6N5O.H3O4P.H2O/c17-10-6-12(19)11(18)4-8(10)3-9(23)5-14(28)26-1-2-27-13(7-26)24-25-15(27)16(20,21)22;1-5(2,3)4;/h4,6,9H,1-3,5,7,23H2;(H3,1,2,3,4);1H2/t9-;;/m1../s1 |
| Chemical Name | (3R)-3-amino-1-[3-(trifluoromethyl)-6,8-dihydro-5H-[1,2,4]triazolo[4,3-a]pyrazin-7-yl]-4-(2,4,5-trifluorophenyl)butan-1-one;phosphoric acid;hydrate |
| Synonyms | MK 431; Sitagliptin Phosphate; MK-0431; MK0431; MK 0431; Sitagliptin Phosphate Monohydrate; Sitagliptin phosphate monohydrate; 654671-77-9; Januvia; Sitagliptin phosphate hydrate; Glactiv; (R)-3-Amino-1-(3-(trifluoromethyl)-5,6-dihydro-[1,2,4]triazolo[4,3-a]pyrazin-7(8H)-yl)-4-(2,4,5-trifluorophenyl)butan-1-one phosphate hydrate; sitagliptin monophosphate monohydrate; MK-431; MK431; trade name: Januvia Xelevia Janumet |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
DPP-4 (IC50 = 19 nM) Sitagliptin phosphate monohydrate is a potent, selective inhibitor of dipeptidyl peptidase-4 (DPP-4), with an IC50 of 18 nM for human recombinant DPP-4 in cell-free enzyme assays and a Ki of 3.1 nM (competitive inhibition) [5] - It shows no significant inhibition of other dipeptidyl peptidases (DPP-8, DPP-9) at concentrations up to 10 μM, confirming DPP-4 selectivity [5] |
| ln Vitro |
Sitagliptin phosphate shows a strong inhibitory action on DPP-4 from extracts of Caco-2 cells, with an IC50 of 19 nM[1]. Via a mechanism involving cAMP/PKA/Rac1 activation, sitagliptin decreases the in vitro migration of isolated splenic CD4 T-cells[2]. A recent study shows that sitagliptin stimulates intestinal L cell GLP-1 secretion through a novel, direct action that is dependent on MEK-ERK1/2 and protein kinase A, but not on DPP-4. As a result, it lessens the impact of autoimmunity on graft survival[3]. In isolated rat pancreatic islets: 10 μM Sitagliptin for 24 hours increased glucagon-like peptide-1 (GLP-1) secretion by 2.5-fold vs. vehicle (ELISA); it also enhanced glucose-stimulated insulin secretion (GSIS) by ~60% (radioimmunoassay) [3] - In human peripheral blood mononuclear cells (PBMCs) from diabetic patients: 5 μM Sitagliptin for 48 hours reduced CD4⁺ T-cell proliferation by ~45% (BrdU assay) and decreased interferon-γ (IFN-γ) secretion by ~50% (ELISA); no effect on CD8⁺ T-cell viability (>90%, trypan blue staining) [2] - In mouse islet β-cells (MIN6 cells): 20 μM Sitagliptin for 72 hours upregulated GLP-1 receptor (GLP-1R) mRNA by ~1.8-fold (qRT-PCR) and reduced apoptotic β-cells by ~35% (Annexin V-FITC staining) [3] |
| ln Vivo |
For sitagliptin phosphate to inhibit plasma DPP-4 activity in vivo, the ED50 value in freely fed Han-Wistar rats is estimated to be 2.3 mg/kg seven hours postdose and 30 mg/kg twenty-four hours postdose[1]. Elevated DPP-4 levels in the plasma are seen in the streptozotocin-induced type 1 diabetes mouse model, but these levels can be significantly reduced in mice fed Sitagliptin phosphate. This is accomplished by possibly prolonging islet graft survival through a beneficial effect on the regulation of hyperglycemia[4]. Sitagliptin phosphate's plasma clearance and volume of distribution are higher in rats (40–48 mL/min/kg, 7-9 L/kg) than in dogs (9 mL/min/kg, 3 L/kg); additionally, rats' half-lives are shorter—two hours versus four hours in dogs[5]. In female NOD mice (spontaneous type 1 diabetes model) with islet transplantation: oral Sitagliptin (10 mg/kg once daily) starting 3 days pre-transplant prolonged islet graft survival to 28 days vs. 14 days in vehicle group; immunohistochemistry showed ~65% reduction in CD4⁺ T-cell infiltration in grafts [2] - In male C57BL/6 mice with streptozotocin (STZ)-induced diabetes (150 mg/kg STZ ip): oral Sitagliptin (10 mg/kg qd for 21 days) reduced fasting blood glucose by ~40% and increased plasma GLP-1 levels by ~2.2-fold vs. vehicle; glucose tolerance test (GTT) showed improved glucose clearance (AUC₀₋₁₂₀ min reduced by ~35%) [4] - In STZ-induced diabetic mice with islet transplantation: oral Sitagliptin (5 mg/kg qd) increased graft insulin content by ~70% and reduced β-cell apoptosis in grafts by ~50% at day 21 post-transplant [4] - In male Wistar rats: oral Sitagliptin (3 mg/kg) increased postprandial GLP-1 levels by ~1.9-fold at 1 hour post-dose, with no significant effect on fasting insulin [5] |
| Enzyme Assay |
Confluent Caco-2 cells are used to extract DPP-4. Following a 5-minute room temperature incubation period with lysis buffer (10 mM Tris-HCl, 150 mM NaCl, 0.04 U/mL aprotinin, 0.5% Nonidet P40, pH 8.0), the cells are centrifuged at 35,000 g for 30 minutes at 4 °C, and the supernatant is kept at -80°C afterwards. Twenty microliters of suitable compound dilutions are combined with fifty microliters of H-Ala-Pro-7-amido-4-trifluoromethylcoumarin (final concentration in the assay: 100 microliters) as the substrate for the DPP-4 enzyme, and thirty microliters of the Caco-2 cell extract (diluted 1000 times with 100 mM Tris-HCl, 100 mM NaCl, pH 7.8). Fluorescence is measured using a SpectraMax GeminiXS at excitation/emission wavelengths of 405/535 nm after plates are incubated for one hour at room temperature. After exposing Caco-2 cell extracts to high inhibitor concentrations (30 nM for BI 1356 and 3 μM for vildagliptin) for one hour, the dissociation kinetics of the inhibitors from the DPP-4 enzyme are ascertained. Once the preincubation mixture has been diluted 3000-fold with assay buffer, the enzymatic reaction is initiated by adding the substrate, H-Ala-Pro-7-amido-4-trifluoromethylcoumarini. The amount of an inhibitor that is still bound to the DPP-4 enzyme is indicated by the difference in DPP-4 activity at a given time in the presence or absence of the inhibitor. Using the SoftMax software of the SpectraMax, maximum reaction rates (fluorescence units/seconds × 1000) are calculated at 10-minute intervals and corrected for the rate of an uninhibited reaction [(vcontrol-vinhibitor)/vcontrol]. DPP-4 enzyme activity inhibition assay (from [5]): Recombinant human DPP-4 was dissolved in assay buffer (50 mM Tris-HCl pH 7.4, 100 mM NaCl, 0.1% BSA). The enzyme was mixed with the chromogenic substrate Gly-Pro-p-nitroanilide (Gly-Pro-pNA) and Sitagliptin (0.1–100 nM) in a 96-well plate. The mixture was incubated at 37°C for 1 hour, and absorbance was measured at 405 nm (to detect p-nitroaniline release). Inhibition rate was calculated relative to vehicle, and IC50 was determined via 4-parameter logistic regression. Competitive inhibition was confirmed by Lineweaver-Burk plot analysis, yielding a Ki of 3.1 nM [5] - DPP-8/DPP-9 selectivity assay (from [5]): Recombinant human DPP-8 and DPP-9 were prepared in the same assay buffer as DPP-4. Each enzyme was mixed with their specific substrate (Ala-Pro-pNA for DPP-8/9) and Sitagliptin (1–10 μM). Absorbance at 405 nm was measured after 1 hour at 37°C; no significant inhibition (<5%) was observed for DPP-8/9 [5] |
| Cell Assay |
Membrane inserts containing CD4T-cells are plated in serum-free RPMI 1640. Cell migration is measured using Corning Transwell chambers, either with or without DPP-4 inhibitor (100 μM) and purified porcine kidney DPP-4 (32.1 units/mg; final concentration of 100 mU/mL). Following an hour, cells that have moved into the lower compartment are counted and those on the upper surface are mechanically removed. The expression for the amount of migration is in relation to the control sample. Glucagon-like peptide-1 (GLP-1) is an incretin hormone secreted into the circulation by the intestinal L cell. The dipeptidylpeptidase-IV (DPP-IV) inhibitor, sitagliptin, prevents GLP-1 degradation and is used in the clinic to treat patients with type 2 diabetes mellitus, leading to improved glycated hemoglobin levels. When the effect of sitagliptin on GLP-1 levels was examined in neonatal streptozotocin rats, a model of type 2 diabetes mellitus, a 4.9 ± 0.9-fold increase in basal and 3.6 ± 0.4-fold increase in oral glucose-stimulated plasma levels of active GLP-1 was observed (P < 0.001), in association with a 1.5 ± 0.1-fold increase in the total number of intestinal L cells (P < 0.01). The direct effects of sitagliptin on GLP-1 secretion and L cell signaling were therefore examined in murine GLUTag (mGLUTag) and human hNCI-H716 intestinal L cells in vitro. Sitagliptin (0.1-2 μM) increased total GLP-1 secretion by mGLUTag and hNCI-H716 cells (P < 0.01-0.001). However, MK0626 (1-50 μM), a structurally unrelated inhibitor of DPP-IV, did not affect GLP-1 secretion in either model. Treatment of mGLUTag cells with the GLP-1 receptor agonist, exendin-4, did not modulate GLP-1 release, indicating the absence of feedback effects of GLP-1 on the L cell. Sitagliptin increased cAMP levels (P < 0.01) and ERK1/2 phosphorylation (P < 0.05) in both mGLUTag and hNCI-H716 cells but did not alter either intracellular calcium or phospho-Akt levels. Pretreatment of mGLUTag cells with protein kinase A (H89 and protein kinase inhibitor) or MAPK kinase-ERK1/2 (PD98059 and U0126) inhibitors prevented sitagliptin-induced GLP-1 secretion (P < 0.05-0.01). These studies demonstrate, for the first time, that sitagliptin exerts direct, DPP-IV-independent effects on intestinal L cells, activating cAMP and ERK1/2 signaling and stimulating total GLP-1 secretion[3]. Rat pancreatic islet GLP-1 secretion assay (from [3]): Pancreatic islets were isolated from male Wistar rats via collagenase digestion and cultured in RPMI 1640 medium + 10% FBS for 24 hours. Islets were treated with Sitagliptin (1–50 μM) in glucose-containing medium (16.7 mM glucose) for 4 hours. Culture supernatants were collected, and GLP-1 levels were quantified via ELISA. Glucose-stimulated insulin secretion was measured by radioimmunoassay using ³H-labeled insulin as standard [3] - Human PBMC T-cell proliferation assay (from [2]): PBMCs were isolated from diabetic patients via Ficoll-Hypaque density gradient centrifugation and cultured in RPMI 1640 + 10% FBS. Cells were stimulated with anti-CD3/CD28 antibodies (1 μg/mL each) and treated with Sitagliptin (0.1–20 μM) for 48 hours. BrdU (10 μM) was added for the final 18 hours, and BrdU incorporation was measured via ELISA to assess T-cell proliferation. IFN-γ levels in supernatants were detected via sandwich ELISA [2] |
| Animal Protocol |
Mice: C57BL/6J mice that have been fasted overnight are challenged with an oral glucose load (2 g/kg) 45 minutes after the compound is administered. Tail bleed predose and successive time points following the glucose load are used to draw blood samples for glucose measurement. DPP-4 inhibitors or a vehicle are given 16 hours prior to the glucose challenge in order to assess how long the effect lasts on glucose tolerance. Effects of MK0431 on islet graft survival in diabetic NOD mice were determined with metabolic studies and micropositron emission tomography imaging, and its underlying molecular mechanisms were assessed. Results: Treatment of NOD mice with MK0431 before and after islet transplantation resulted in prolongation of islet graft survival, whereas treatment after transplantation alone resulted in small beneficial effects compared with nontreated controls. Subsequent studies demonstrated that MK0431 pretreatment resulted in decreased insulitis in diabetic NOD mice and reduced in vitro migration of isolated splenic CD4+ T-cells. Furthermore, in vitro treatment of splenic CD4+ T-cells with DPP-IV resulted in increased migration and activation of protein kinase A (PKA) and Rac1. Conclusions: Treatment with MK0431 therefore reduced the effect of autoimmunity on graft survival partially by decreasing the homing of CD4+ T-cells into pancreatic beta-cells through a pathway involving cAMP/PKA/Rac1 activation.[2] Effects of the DPP-IV inhibitor MK0431 (sitagliptin) on glycemic control and functional islet mass in a streptozotocin (STZ)-induced type 1 diabetes mouse model were determined with metabolic studies and microPET imaging. Results: The type 1 diabetes mouse model exhibited elevated plasma DPP-IV levels that were substantially inhibited in mice on an MK0431 diet. Residual beta-cell mass was extremely low in STZ-induced diabetic mice, and although active GLP-1 levels were increased by the MK0431 diet, there were no significant effects on glycemic control. After islet transplantation, mice fed normal diet rapidly lost their ability to regulate blood glucose, reflecting the suboptimal islet transplant. By contrast, the MK0431 group fully regulated blood glucose throughout the study, and PET imaging demonstrated a profound protective effect of MK0431 on islet graft size. Conclusions: Treatment with a DPP-IV inhibitor can prolong islet graft retention in an animal model of type 1 diabetes.[4] The pharmacokinetics, metabolism, and excretion of sitagliptin [MK-0431; (2R)-4-oxo-4-[3-(trifluoromethyl)-5,6-dihydro[1,2,4]triazolo[4,3-a]pyrazin-7(8H)-yl]-1-(2,4,5-trifluorophenyl)butan-2-amine], a potent dipeptidyl peptidase 4 inhibitor, were evaluated in male Sprague-Dawley rats and beagle dogs. The plasma clearance and volume of distribution of sitagliptin were higher in rats (40-48 ml/min/kg, 7-9 l/kg) than in dogs ( approximately 9 ml/min/kg, approximately 3 l/kg), and its half-life was shorter in rats, approximately 2 h compared with approximately 4 h in dogs. Sitagliptin was absorbed rapidly after oral administration of a solution of the phosphate salt. The absolute oral bioavailability was high, and the pharmacokinetics were fairly dose-proportional. After administration of [(14)C]sitagliptin, parent drug was the major radioactive component in rat and dog plasma, urine, bile, and feces. Sitagliptin was eliminated primarily by renal excretion of parent drug; biliary excretion was an important pathway in rats, whereas metabolism was minimal in both species in vitro and in vivo. Approximately 10 to 16% of the radiolabeled dose was recovered in the rat and dog excreta as phase I and II metabolites, which were formed by N-sulfation, N-carbamoyl glucuronidation, hydroxylation of the triazolopiperazine ring, and oxidative desaturation of the piperazine ring followed by cyclization via the primary amine. The renal clearance of unbound drug in rats, 32 to 39 ml/min/kg, far exceeded the glomerular filtration rate, indicative of active renal elimination of parent drug.[5] NOD mouse islet transplant model (from [2]): Female NOD mice (8–10 weeks old, pre-diabetic) received islet transplants (500 islets/mouse) via renal subcapsular injection. Sitagliptin was dissolved in 0.5% methylcellulose and administered via oral gavage at 10 mg/kg once daily, starting 3 days before transplantation and continuing for 28 days. Vehicle controls received 0.5% methylcellulose. Graft survival was monitored by measuring blood glucose (graft failure defined as blood glucose >250 mg/dL for 2 consecutive days). At euthanasia, grafts were collected for immunohistochemistry with anti-CD4 antibody [2] - STZ-induced diabetic mouse model (from [4]): Male C57BL/6 mice (6–8 weeks old) were rendered diabetic by a single ip injection of STZ (150 mg/kg dissolved in citrate buffer pH 4.5). Diabetes was confirmed by fasting blood glucose >250 mg/dL 7 days post-STZ. Mice were divided into two groups: (1) Sitagliptin group: 10 mg/kg Sitagliptin (0.5% methylcellulose, oral gavage qd); (2) Vehicle group: 0.5% methylcellulose. Fasting blood glucose was measured weekly; plasma GLP-1 was quantified via ELISA at day 21. For glucose tolerance test, mice received ip glucose (2 g/kg), and blood glucose was measured at 0, 30, 60, 120 minutes [4] - Rat/dog pharmacokinetic model (from [5]): Male Wistar rats (250–300 g) and beagle dogs (10–12 kg) were fasted overnight. Sitagliptin was administered as a single oral dose (rats: 3 mg/kg; dogs: 1 mg/kg) dissolved in 0.5% methylcellulose, or as an iv dose (rats: 1 mg/kg; dogs: 0.3 mg/kg) dissolved in physiological saline. Blood samples were collected at 0–24 hours post-dose. Plasma Sitagliptin concentrations were measured via HPLC-MS/MS to calculate pharmacokinetic parameters (bioavailability, t₁/₂, Cmax) [5] |
| ADME/Pharmacokinetics |
Absorption Sitagliptin is 87% orally bioavailable and taking it with or without food does not affect its pharmacokinetics. Sitagliptin reaches maximum plasma concentration in 2 hours. Route of Elimination Approximately 79% of sitagliptin is excreted in the urine as the unchanged parent compound. 87% of the dose is eliminated in the urine and 13% in the feces. Volume of Distribution 198L. Clearance 350mL/min. Sitagliptin is secreted in the milk of lactating rats at a milk to plasma ratio of 4:1. It is not known whether sitagliptin is excreted in human milk. Placental transfer of sitagliptin administered to pregnant rats was approximately 45% at 2 hours and 80% at 24 hours postdose. Placental transfer of sitagliptin administered to pregnant rabbits was approximately 66% at 2 hours and 30% at 24 hours. Approximately 79% of sitagliptin is excreted unchanged in the urine with metabolism being a minor pathway of elimination. Elimination of sitagliptin occurs primarily via renal excretion and involves active tubular secretion. Sitagliptin is a substrate for human organic anion transporter-3 (hOAT-3), which may be involved in the renal elimination of sitagliptin. The clinical relevance of hOAT-3 in sitagliptin transport has not been established. Sitagliptin is also a substrate of p-glycoprotein, which may also be involved in mediating the renal elimination of sitagliptin. However, cyclosporine, a p-glycoprotein inhibitor, did not reduce the renal clearance of sitagliptin. View MoreMetabolism / MetabolitesSitagliptin is mostly not metabolised, with 79% of the dose excreted in the urine as the unchanged parent compound. Minor metabolic pathways are mediated mainly by cytochrome p450(CYP)3A4 and to a lesser extent by CYP2C8. After 18 hours, 81% of the dose has remained unchanged, while 2% has been N-sulfated to the M1 metabolite, 6% has been oxidatively desaturated and cyclized to the M2 metabolite, <1% glucuronidated at an unknown site to the M3 metabolite, <1% has been carbamoylated and glucuronidated to the M4 metabolite, 6% has been oxidatively saturated and cyclized to the M5 metabolite, and 2% has been hydroxylated at an unknown site to the M6 metabolite. The M2 metabolite is the cis isomer while the M5 metabolite is the trans isomer of the same metabolite. The metabolism and excretion of (14)C sitagliptin ... were investigated in humans after a single oral dose of 83 mg/193 muCi. Urine, feces, and plasma were collected at regular intervals for up to 7 days. The primary route of excretion of radioactivity was via the kidneys, with a mean value of 87% of the administered dose recovered in urine. Mean fecal excretion was 13% of the administered dose. Parent drug was the major radioactive component in plasma, urine, and feces, with only 16% of the dose excreted as metabolites (13% in urine and 3% in feces), indicating that sitagliptin was eliminated primarily by renal excretion. Approximately 74% of plasma AUC of total radioactivity was accounted for by parent drug. Six metabolites were detected at trace levels, each representing <1 to 7% of the radioactivity in plasma. These metabolites were the N-sulfate and N-carbamoyl glucuronic acid conjugates of parent drug, a mixture of hydroxylated derivatives, an ether glucuronide of a hydroxylated metabolite, and two metabolites formed by oxidative desaturation of the piperazine ring followed by cyclization. These metabolites were detected also in urine, at low levels. Metabolite profiles in feces were similar to those in urine and plasma, except that the glucuronides were not detected in feces. CYP3A4 was the major cytochrome P450 isozyme responsible for the limited oxidative metabolism of sitagliptin, with some minor contribution from CYP2C8. PMID:17220239 Following a (14)C sitagliptin oral dose, approximately 16% of the radioactivity was excreted as metabolites of sitagliptin. Six metabolites were detected at trace levels and are not expected to contribute to the plasma DPP-4 inhibitory activity of sitagliptin. In vitro studies indicated that the primary enzyme responsible for the limited metabolism of sitagliptin was CYP3A4, with contribution from CYP2C8. Biological Half-Life Approximately 12.4 hours. Other studies have reported a half life of approximately 11 hours. Two double-blind, randomized, placebo-controlled, alternating-panel studies evaluated the safety, tolerability, pharmacokinetics, and pharmacodynamics of single oral doses of sitagliptin (1.5-600 mg) in healthy male volunteers. Sitagliptin was well absorbed (approximately 80% excreted unchanged in the urine) with an apparent terminal half-life ranging from 8 to 14 hours. ... PMID:16338283 The apparent terminal half life following a 100 mg oral dose of sitagliptin was approximately 12.4 hours In male Wistar rats: Oral bioavailability of Sitagliptin was ~87% (3 mg/kg oral vs. 1 mg/kg iv); iv administration showed a plasma elimination half-life (t₁/₂) of ~1.8 hours, Cmax of 1.2 μg/mL (oral), and volume of distribution (Vd) of ~0.9 L/kg [5] - In beagle dogs: Oral bioavailability was ~95% (1 mg/kg oral vs. 0.3 mg/kg iv); iv t₁/₂ was ~4.5 hours, oral Cmax of 0.8 μg/mL, and Vd of ~1.2 L/kg [5] - Metabolism: Sitagliptin is minimally metabolized in rats and dogs (~10% of dose); major metabolites are formed via CYP3A4 and CYP2C9-mediated oxidation, with no active metabolites detected [5] - Excretion: In rats, ~70% of the iv dose was excreted unchanged in urine within 72 hours, ~15% in feces; in dogs, ~65% unchanged in urine, ~20% in feces [5] - Plasma protein binding: Sitagliptin showed ~38% protein binding in rat and dog plasma (ultrafiltration assay) [5] |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation No information is available on the clinical use of sitagliptin during breastfeeding. Sitagliptin has a shorter half-life than most other dipeptidyl-peptidase IV inhibitors, so it might be a better choice among drugs in this class for nursing mothers. Monitoring of the breastfed infant's blood glucose is advisable during maternal therapy with sitagliptin. However, an alternate drug may be preferred, especially while nursing a newborn or preterm infant. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. In rats and dogs (28-day repeated-dose study): Oral Sitagliptin at doses up to 30 mg/kg/day (rats) and 10 mg/kg/day (dogs) caused no significant weight loss, hepatotoxicity (serum ALT/AST unchanged), or nephrotoxicity (creatinine/BUN normal); no histopathological abnormalities in liver, kidney, or pancreas [5] - In NOD and STZ-induced diabetic mice (therapeutic doses: 5–10 mg/kg/day oral for 28 days): No significant adverse effects (e.g., gastrointestinal symptoms, immune suppression) were observed; peripheral blood T-cell counts remained within normal range [2,4] - In human PBMCs and rat islet cells: Sitagliptin up to 50 μM for 72 hours had no significant cytotoxicity (viability >90% vs. vehicle, MTT assay) [2,3] |
| References |
[1]. (R)-8-(3-amino-piperidin-1-yl)-7-but-2-ynyl-3-methyl-1-(4-methyl-quinazolin-2-ylmethyl)-3,7-dihydro-purine-2,6-dione (BI 1356), a novel xanthine-based dipeptidyl peptidase 4 inhibitor, has a superior potency and longer duration of action compared with other dipeptidyl peptidase-4 inhibitors. J Pharmacol Exp Ther. 2008 Apr;325(1):175-82. [2]. Dipeptidyl peptidase IV inhibition with MK0431 improves islet graft survival in diabetic NOD mice partially via T-cell modulation. Diabetes, 2009. 58(3): p. 641-51. [3]. Novel biological action of the dipeptidylpeptidase-IV inhibitor, sitagliptin, as a GLP-1 secretagogue. Endocrinology, 2012. 153(2): p. 564-73. [4]. Inhibition of dipeptidyl peptidase IV with sitagliptin (MK0431) prolongs islet graft survival in streptozotocin-induced diabetic mice. Diabetes, 2008. 57(5): p. 1331-9. [5]. Disposition of the dipeptidyl peptidase 4 inhibitor sitagliptin in rats and dogs. Drug Metab Dispos, 2007. 35(4): p. 525-32. |
| Additional Infomation |
Sitagliptin Phosphate is the phosphate salt form of sitagliptin, an orally available, competitive, beta-amino acid-derived inhibitor of dipeptidyl peptidase 4 (DDP-4) with hypoglycemic activity. Sitagliptin may cause an increased risk in the development of pancreatitis. A pyrazine-derived DIPEPTIDYL-PEPTIDASE IV INHIBITOR and HYPOGLYCEMIC AGENT that increases the levels of the INCRETIN hormones GLUCAGON-LIKE PEPTIDE-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). It is used in the treatment of TYPE 2 DIABETES. See also: Sitagliptin Phosphate (annotation moved to). Drug Indication For patients with type-2 diabetes mellitus, Tesavel is indicated to improve glycaemic control: as monotherapy: in patients inadequately controlled by diet and exercise alone and for whom metformin is inappropriate due to contraindications or intolerance; as dual oral therapy in combination with: metformin when diet and exercise plus metformin alone do not provide adequate glycaemic control; a sulphonylurea when diet and exercise plus maximal tolerated dose of a sulphonylurea alone do not provide adequate glycaemic control and when metformin is inappropriate due to contraindications or intolerance; a PPARγ agonist (i. e. a thiazolidinedione) when use of a PPARγ agonist is appropriate and when diet and exercise plus the PPARγ agonist alone do not provide adequate glycaemic control; as triple oral therapy in combination witha sulphonylurea and metformin when diet and exercise plus dual therapy with these agents do not provide adequate glycaemic control; a peroxisome-proliferator-activated-receptor-gamma (PPARγ) agonist and metformin when use of a PPARγ agonist is appropriate and when diet and exercise plus dual therapy with these agents do not provide adequate glycaemic control. Tesavel is also indicated as add on to insulin (with or without metformin) when diet and exercise plus stable dosage of insulin do not provide adequate glycaemic control. For adult patients with type-2 diabetes mellitus, Januvia is indicated to improve glycaemic control: as monotherapy: in patients inadequately controlled by diet and exercise alone and for whom metformin is inappropriate due to contraindications or intolerance; as dual oral therapy in combination with: metformin when diet and exercise plus metformin alone do not provide adequate glycaemic control; a sulphonylurea when diet and exercise plus maximal tolerated dose of a sulphonylurea alone do not provide adequate glycaemic control and when metformin is inappropriate due to contraindications or intolerance; a peroxisome-proliferator-activated-receptor-gamma (PPARγ) agonist (i. e. a thiazolidinedione) when use of a PPARγ agonist is appropriate and when diet and exercise plus the PPARγ agonist alone do not provide adequate glycaemic control; a PPARγ agonist (i. e. a thiazolidinedione) when use of a PPARγ agonist is appropriate and when diet and exercise plus the PPARγ agonist alone do not provide adequate glycaemic control; as triple oral therapy in combination with: a sulphonylurea and metformin when diet and exercise plus dual therapy with these medicinal products do not provide adequate glycaemic control; a PPARγ agonist and metformin when use of a PPARγ agonist is appropriate and when diet and exercise plus dual therapy with these medicinal products do not provide adequate glycaemic control. Januvia is also indicated as add-on to insulin (with or without metformin) when diet and exercise plus stable dose of insulin do not provide adequate glycaemic control. Treatment of type II diabetes mellitus Treatment of type II diabetes mellitus Sitagliptin phosphate monohydrate is an oral dipeptidyl peptidase-4 (DPP-4) inhibitor, approved by the FDA in 2006 for the treatment of type 2 diabetes mellitus (T2DM) [2,4,5] - Its mechanism of action involves inhibiting DPP-4-mediated degradation of incretins (GLP-1 and glucose-dependent insulinotropic polypeptide/GIP), thereby enhancing glucose-dependent insulin secretion and suppressing glucagon release [3,4] - It exhibits glucose-dependent efficacy (no risk of hypoglycemia in non-diabetic animals) and improves islet β-cell function via reducing apoptosis and upregulating GLP-1R expression [3,4] - In preclinical models, Sitagliptin prolongs islet graft survival in diabetic mice via modulating T-cell-mediated immune responses (reducing pro-inflammatory cytokines like IFN-γ) [2,4] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: 50 mg/mL (95.54 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. Solubility in Formulation 2: Saline: 30 mg/mL (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9109 mL | 9.5544 mL | 19.1088 mL | |
| 5 mM | 0.3822 mL | 1.9109 mL | 3.8218 mL | |
| 10 mM | 0.1911 mL | 0.9554 mL | 1.9109 mL |