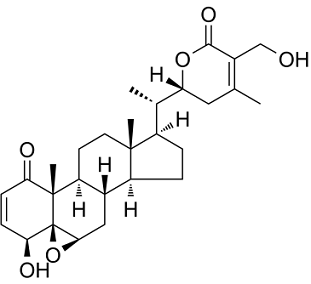
Withaferin A, a steroidal lactone class of NFkB/IkB Inhibitor, is a natural product extracted from Acnistus arborescens, Withania somnifera (Indian Winter cherry or Ashwagandha in Sanskrit) and other members of Solanaceae family. Ayurvedic medicine has historically used it. It is the earliest discovered ergostane-type product belonging to the withanolide class. This natural product has a wide range of pharmacological properties, including cardioprotective, anti-inflammatory, immuno-modulatory, anti-angiogenesis, anti-metastasis, and anti-carcinogenic properties.
Physicochemical Properties
| Molecular Formula | C28H38O6 |
| Molecular Weight | 470.5977 |
| Exact Mass | 470.266 |
| Elemental Analysis | C, 71.46; H, 8.14; O, 20.40 |
| CAS # | 5119-48-2 |
| Related CAS # | 5119-48-2 |
| PubChem CID | 265237 |
| Appearance | White to off-white solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 680.7±55.0 °C at 760 mmHg |
| Melting Point | 252-253ºC |
| Flash Point | 226.7±25.0 °C |
| Vapour Pressure | 0.0±4.8 mmHg at 25°C |
| Index of Refraction | 1.599 |
| LogP | 3.8 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 34 |
| Complexity | 999 |
| Defined Atom Stereocenter Count | 11 |
| SMILES | O1[C@]2([H])C([H])([H])[C@@]3([H])[C@]4([H])C([H])([H])C([H])([H])[C@]([H])([C@]([H])(C([H])([H])[H])[C@@]5([H])C([H])([H])C(C([H])([H])[H])=C(C([H])([H])O[H])C(=O)O5)[C@@]4(C([H])([H])[H])C([H])([H])C([H])([H])[C@]3([H])[C@]3(C(C([H])=C([H])[C@@]([H])([C@]132)O[H])=O)C([H])([H])[H] |
| InChi Key | DBRXOUCRJQVYJQ-CKNDUULBSA-N |
| InChi Code | InChI=1S/C28H38O6/c1-14-11-21(33-25(32)17(14)13-29)15(2)18-5-6-19-16-12-24-28(34-24)23(31)8-7-22(30)27(28,4)20(16)9-10-26(18,19)3/h7-8,15-16,18-21,23-24,29,31H,5-6,9-13H2,1-4H3/t15-,16-,18+,19-,20-,21+,23-,24+,26+,27-,28+/m0/s1 |
| Chemical Name | (1S,2R,6S,7R,9R,11S,12S,15R,16S)-6-hydroxy-15-[(1S)-1-[(2R)-5-(hydroxymethyl)-4-methyl-6-oxo-2,3-dihydropyran-2-yl]ethyl]-2,16-dimethyl-8-oxapentacyclo[9.7.0.02,7.07,9.012,16]octadec-4-en-3-one |
| Synonyms | NSC 101088; NSC101088; NSC-101088; NSC-273757; NSC273757; NSC 273757; Withaferin A |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | NF-κB; vimentin |
| ln Vitro |
Withaferin A inhibits NF-kB activation effectively and has anti-inflammatory properties by preventing Ik-B kinase beta from being activated by TNF through a thioalkylation-sensitive redox mechanism[1]. Withaferin A has anticancer properties as well. The IF protein vimentin is the target of withaferin A, which also causes vimentin filaments to aggregate in bovine aortic endothelial cells (BAECs) at 3 μM and fragment in endothelial cells at 10 μM[2]. The tumorigenic potential of ALDH1 positive cancer stem cells (CSCs) is decreased in a dose-dependent manner by withaferin A (0.5, 1.5 μM) alone or in combination with cisplatin (CIS)[3]. |
| ln Vivo | In mice, withaferin A (2 mg/kg, i.p.) exhibits strong angiogenesis inhibitory activity through vimentin[2]. In tumors taken from mice, the combination of withaferin A (2 mg/kg) and cisplatin (CIS) controls the expression of the ALDH1 marker and inhibits the expression of securin[3]. |
| Cell Assay | The RPMI1640 medium is used to support the ovarian epithelial cancer cell line A2780. It also contains insulin (5 g/mL), penicillin/streptomycin (100 IU/mL and 100 g/mL, respectively), and 10% fetal bovine serum (FBS) from Hyclone. In DMSO, other reagents such as withaferin A and cisplatin (CIS) are prepared. Each time it is made, cisplatin is fresh[3]. |
| Animal Protocol | Mice: In the assay, mice with either a vimentin homozygous defect (Vim−/−) or a vimentin heterozygous defect (Vim−/−) on the 129/Svev background are used. The intraperitoneal (i.p.) injection of ketamine and xylazine is used to anesthetize mice between the ages of 4 and 6 weeks. Proparacain eye drop is used to topically anesthetize the corneas before 1 μL of diluted sodium hydroxide solution (0.15 M) is applied for 1 minute. The cornea is immediately thoroughly washed in saline solution, followed by the delicate removal of the corneal and limbal epithelium by scraping. Atropine eye drops are used topically to treat the cornea, and antibiotic eye ointments containing tobramycin and erythromycin are used to protect it. In the drug or control groups of mice, withaferin A or 12-D WS (2 mg/kg solubilized in DMSO) or vehicle (DMSO) are injected intraperitoneally (i.p.) after the mice have recovered from corneal injury, and then once a day for the following ten days. Eyes are removed from mice that have been killed humanely. In order to prepare the corneal buttons, the anterior segment of the eyes is divided in half. Corneal tissues are fixed in 100% acetone for 20 minutes, cleaned in PBS for 1 hour, and blocked for 18 hours at 4°C in 1% BSA-PBS. Cornea whole-mount staining involves soaking tissues in FITC-conjugated rat anti-mouse CD31 antibody (1:333 dilution in 1% BSA-PBS) for 12 hours, rinsing them off for 24 hours at 4°C in the same solution, and attaching them to glass slides with coverslips. By importing digital images into NIH ImageJ, fluorescent staining can be quantified after being visualized on a microscope[2]. |
| References |
[1]. Withaferin a strongly elicits IkappaB kinase beta hyperphosphorylation concomitant with potent inhibition of its kinase activity. J Biol Chem. 2007 Feb 16;282(7):4253-64. Epub 2006 Dec 6. [2]. The tumor inhibitor and antiangiogenic agent withaferin A targets the intermediate filament protein vimentin. Chem Biol. 2007 Jun;14(6):623-34. [3]. Withaferin A (WFA) inhibits tumor growth and metastasis by targeting ovarian cancer stem cells. Oncotarget. 2017 Aug 10;8(43):74494-74505. |
| Additional Infomation |
Withaferin A is a withanolide that is 5,6:22,26-diepoxyergosta-2,24-diene-1,26-dione substituted by hydroxy groups at positions 4 and 27 (the 4beta,5beta,6beta,22R stereoisomer). Isolated from Physalis longifolia, it exhibits cytotoxic activity. It has a role as an antineoplastic agent and an apoptosis inducer. It is a delta-lactone, a 4-hydroxy steroid, an enone, an ergostanoid, a secondary alcohol, a withanolide, a 27-hydroxy steroid, a primary alcohol and an epoxy steroid. Withaferin A has been reported in Iochroma gesnerioides, Withania coagulans, and other organisms with data available. |
Solubility Data
| Solubility (In Vitro) |
DMSO: 50~94 mg/mL (106.3~199.75 mM) Ethanol: ~4 mg/mL (~8.50 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 0.83 mg/mL (1.76 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 8.3 mg/mL clear DMSO stock solution to 400 μL of PEG300 and mix evenly; then add 50 μL of Tween-80 to the above solution and mix evenly; then add 450 μL of normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 0.83 mg/mL (1.76 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 8.3 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 0.83 mg/mL (1.76 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 8.3 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1249 mL | 10.6247 mL | 21.2495 mL | |
| 5 mM | 0.4250 mL | 2.1249 mL | 4.2499 mL | |
| 10 mM | 0.2125 mL | 1.0625 mL | 2.1249 mL |