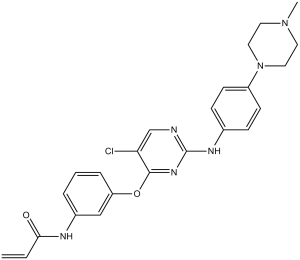
WZ3146 (WZ-3146) is a covalent / irreversible and mutant-selective inhibitor of EGFR (L858R and E746_A750) with potential anticancer activity. It exhibits no effects on ERBB2 phosphorylation (T798I), but it inhibits EGFR (L858R and E746_A750) with IC50s of 2 nM and 2 nM, respectively. When applied to EGFR T790M-containing cell lines in vitro, WZ3146 demonstrates strong anti-proliferative activity.
Physicochemical Properties
| Molecular Formula | C24H25CLN6O2 | |
| Molecular Weight | 464.95 | |
| Exact Mass | 464.172 | |
| Elemental Analysis | C, 62.00; H, 5.42; Cl, 7.63; N, 18.08; O, 6.88 | |
| CAS # | 1214265-56-1 | |
| Related CAS # |
|
|
| PubChem CID | 44607360 | |
| Appearance | White to off-white solid powder | |
| Density | 1.3±0.1 g/cm3 | |
| Index of Refraction | 1.661 | |
| LogP | 3.6 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 7 | |
| Rotatable Bond Count | 7 | |
| Heavy Atom Count | 33 | |
| Complexity | 638 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | ClC1=C([H])N=C(N=C1OC1=C([H])C([H])=C([H])C(=C1[H])N([H])C(C([H])=C([H])[H])=O)N([H])C1C([H])=C([H])C(=C([H])C=1[H])N1C([H])([H])C([H])([H])N(C([H])([H])[H])C([H])([H])C1([H])[H] |
|
| InChi Key | APHGZZPEOCCYNO-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C24H25ClN6O2/c1-3-22(32)27-18-5-4-6-20(15-18)33-23-21(25)16-26-24(29-23)28-17-7-9-19(10-8-17)31-13-11-30(2)12-14-31/h3-10,15-16H,1,11-14H2,2H3,(H,27,32)(H,26,28,29) | |
| Chemical Name | N-[3-[5-chloro-2-[4-(4-methylpiperazin-1-yl)anilino]pyrimidin-4-yl]oxyphenyl]prop-2-enamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
EGFR L858R (IC50 = 2 nM); EGFR L858R/T790M (IC50 = 5 nM); EGFR E746_A750 (IC50 = 2 nM); EGFR E746_A750/T790M (IC50 = 14 nM); EGFR (IC50 = 24 nM) WZ3146 is a mutant-selective inhibitor of the epidermal growth factor receptor (EGFR) tyrosine kinase, specifically targeting the EGFR T790M mutation with higher potency compared to wild-type EGFR. It demonstrated preferential inhibition of EGFR T790M-driven signaling pathways [1] |
| ln Vitro |
WZ3146 markedly inhibits the growth of EGFR mutant cell lines, with an IC50 of 3 nM in EGFR Del E746_A750 containing HCC827 cells, 15 nM in EGFR Del E746_A750 containing PC9 cells, 29 nM in EGFR L858R/T790M containing H1975 cells, and 3 nM in EGFR Del E746_A750/T790M containing PC9 GR cells.[1] WZ3146 dose-dependently inhibited proliferation of EGFR T790M-mutant tumor cell lines (e.g., H1975) by blocking EGFR autophosphorylation and downstream AKT/ERK signaling. It induced apoptosis in these cells as evidenced by caspase-3 activation and PARP cleavage. The drug showed minimal activity against wild-type EGFR-expressing cells at concentrations up to 10 μM [1] |
| ln Vivo |
WZ4002 significantly reduces tumor growth and inhibits EGFR phosphorylation in murine models of EGFR T790M. Oral administration of WZ3146 significantly suppressed tumor growth in nude mice bearing H1975 xenografts. A daily dose of 25 mg/kg for 21 days resulted in a 60% reduction in tumor volume compared to vehicle controls. Intratumoral EGFR T790M phosphorylation was potently inhibited, confirming target engagement [1] |
| Enzyme Assay |
The ATP/NADH coupled assay system in 96-well format is used to perform the assays in triplicate. The final reaction mixture includes the following: 0.5 mg/mL bovine serum albumin (BSA), 2 mM MnCl2, 1 mM phospho(enol) pyruvic acid (PEP), 1 mM TCEP, 0.1 M Hepes 7.4, 2.5 mM poly-[Glu4Tyr1] peptide, 1/50 of the final reaction mixture volume of rabbit muscle-derived pyruvate kinase/lactic dehydrogenase enzymes, 0.5 mM NADH, 0.5 μM EGFR kinase, 100 μM ATP, and varying amounts of WZ3146. To initiate the reaction, ATP and WZ3146 are combined, made into a separate stock from the mixture containing all other ingredients, and then added last to the latter. The slopes of the A340 curves provide steady state initial velocity data. Recombinant EGFR kinase domains (wild-type and T790M mutant) were incubated with WZ3146 (0.01–10 μM) in kinase buffer containing ATP and a synthetic peptide substrate. Phosphorylation levels were measured via radiometric assay. WZ3146 displayed higher binding affinity for the T790M mutant compared to wild-type EGFR, with a >10-fold selectivity ratio [1] |
| Cell Assay |
The MTS assay measures growth and growth inhibition. A fixed number of NSCLC or Ba/F3 cells are used in each experiment, and the cells are subjected to a 72-hour treatment. Three repetitions are required for every experiment, and each experimental point is set up in six to twelve wells. Windows version 5.0 of GraphPad Prism is used to display the data graphically. Non-linear regression model with sigmoidal dose response is used to fit the curves. H1975 cells were treated with WZ3146 (0.1–5 μM) for 72 hours, and cell viability was assessed using MTT assay. Western blot analysis confirmed dose-dependent inhibition of p-EGFR (T790M), p-AKT, and p-ERK. Apoptosis was evaluated by Annexin V/PI staining and caspase-3 activation [1] |
| Animal Protocol |
The MTS assay is used to evaluate growth and growth inhibition. Ba/F3 cells are treated with WZ3146 for a duration of 72 hours. By using the MTS assay, growth and growth inhibition are evaluated. Nude mice with H1975 xenografts (100–150 mm³) received WZ3146 orally (25 mg/kg/day) in 0.5% carboxymethylcellulose for 21 days. Tumor volume was measured thrice weekly. At sacrifice, tumors were analyzed for p-EGFR (T790M) expression by immunohistochemistry [1] |
| ADME/Pharmacokinetics |
WZ3146 exhibited moderate oral bioavailability (~40%) in mice, with a plasma half-life of 3.5 hours. Maximum plasma concentration (Cmax) of 2.1 μg/mL was achieved 1 hour post-dose. The drug distributed preferentially to tumor tissues, with a tumor-to-plasma concentration ratio of 2.8 [1] |
| Toxicity/Toxicokinetics |
No significant toxicity was observed in mice treated with WZ3146 at doses up to 50 mg/kg/day for 14 days. Serum ALT, AST, and creatinine levels remained within normal ranges. Histopathological analysis of liver and kidneys showed no structural damage [1] |
| References |
[1]. Novel mutant-selective EGFR kinase inhibitors against EGFR T790M.Nature. 2009 Dec 24;462(7276):1070-4. |
| Additional Infomation |
N-[3-[[5-chloro-2-[4-(4-methyl-1-piperazinyl)anilino]-4-pyrimidinyl]oxy]phenyl]-2-propenamide is a member of piperazines. WZ3146 represents a novel class of EGFR T790M-selective inhibitors that overcome resistance to first- and second-generation EGFR TKIs. Its mechanism involves covalent binding to the cysteine residue in the EGFR kinase domain, specifically stabilizing the inactive conformation of the T790M mutant. Preclinical data support its potential as a therapeutic agent for EGFR T790M-positive non-small cell lung cancer [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.38 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.38 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.38 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1508 mL | 10.7538 mL | 21.5077 mL | |
| 5 mM | 0.4302 mL | 2.1508 mL | 4.3015 mL | |
| 10 mM | 0.2151 mL | 1.0754 mL | 2.1508 mL |