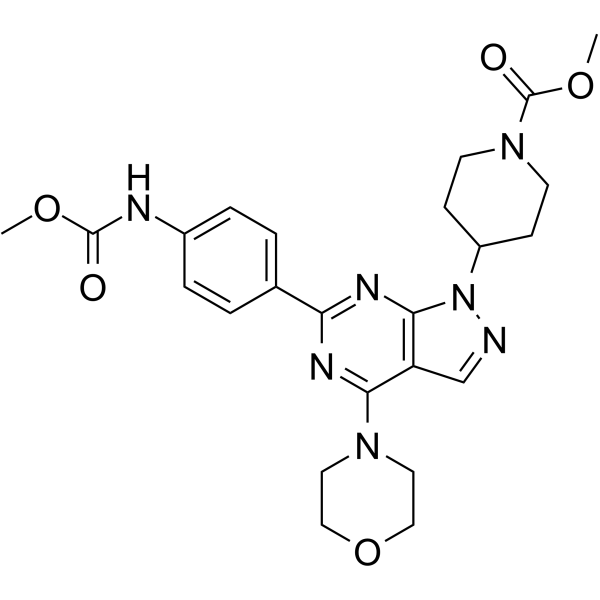
WYE-354 (WYE 354) is a novel, potent, specific, cell-permeable and ATP-competitive mTOR (mammalian target of rapamycin) inhibitor with potential anticancer/antitumor activity. With an IC50 of 5 nM, it inhibits mTOR.
Physicochemical Properties
| Molecular Formula | C24H29N7O5 |
| Molecular Weight | 495.53 |
| Exact Mass | 495.223 |
| Elemental Analysis | C, 58.17; H, 5.90; N, 19.79; O, 16.14 |
| CAS # | 1062169-56-5 |
| Related CAS # | 1062169-56-5 |
| PubChem CID | 44219749 |
| Appearance | White to off-white crystalline solid |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 594.2±50.0 °C at 760 mmHg |
| Flash Point | 313.2±30.1 °C |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C |
| Index of Refraction | 1.692 |
| LogP | 0.93 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 36 |
| Complexity | 753 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | COC(NC1=CC=C(C=C1)C2=NC3=C(C(N4CCOCC4)=N2)C=NN3C5CCN(CC5)C(OC)=O)=O |
| InChi Key | IMXHGCRIEAKIBU-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C24H29N7O5/c1-34-23(32)26-17-5-3-16(4-6-17)20-27-21(29-11-13-36-14-12-29)19-15-25-31(22(19)28-20)18-7-9-30(10-8-18)24(33)35-2/h3-6,15,18H,7-14H2,1-2H3,(H,26,32) |
| Chemical Name | methyl 4-(6-(4-((methoxycarbonyl)amino)phenyl)-4-morpholino-1H-pyrazolo[3,4-d]pyrimidin-1-yl)piperidine-1-carboxylate |
| Synonyms | WYE 354; WYE354; WYE-354; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
mTOR (IC50 = 5 nM); mTORC1; mTORC2; PI3K alpha (IC50 = 1.89 μM); PI3K gamma (IC50 = 7.37 μM); Autophagy; WYE-354 is a potent, ATP-competitive inhibitor of mammalian target of rapamycin (mTOR), targeting both mTOR complex 1 (mTORC1) and mTOR complex 2 (mTORC2). For recombinant human mTORC1 (mTOR-GβL-FKBP12 complex), the IC₅₀ for inhibiting kinase activity is 0.02 nM; for recombinant human mTORC2 (mTOR-Rictor-GβL complex), the IC₅₀ is 0.08 nM. It exhibits high selectivity over PI3K family kinases (PI3Kα, PI3Kβ, PI3Kγ, PI3Kδ), with IC₅₀ values >1000 nM (≥50,000-fold higher than its IC₅₀ for mTOR) [1] |
| ln Vitro |
WYE-354 inhibits recombinant mTOR enzyme in the DELFIA assay for measuring His6-S6K1 T389 phosphorylation with an IC50 of 5 nM[1]. MTS assay is used to evaluate cell viability. WYE-354 is applied to the G-415 and TGBC-2TKB cell lines at increasing concentrations for 24, 48, and 72 hours. After a 24-hour exposure, WYE-354 significantly lowers cell viability in both tested cell lines starting at a 1 M concentration (P<0.001). Except for the TGBC-2TKB cell line after 72 hours of treatment, no reduction in cell viability is seen at a dose of 100 nM[2]. Antiproliferative activity across multiple cancer cell lines (Literature [1]): Using the MTT assay (72-hour treatment), WYE-354 inhibited proliferation of various human cancer cells with the following IC₅₀ values: HeLa (cervical cancer, 15 nM), U2OS (osteosarcoma, 18 nM), MCF-7 (breast cancer, 12 nM), PC-3 (prostate cancer, 16 nM), and HCT116 (colorectal cancer, 14 nM). At 50 nM, it suppressed cell proliferation by >85% in all tested cell lines [1] - Inhibition of mTOR downstream signaling (Literature [1,2]): - HeLa cells treated with 10 nM WYE-354 for 24 hours showed significant reduction in phosphorylation of mTORC1 and mTORC2 substrates (Western blot): p-p70S6K (Thr389) decreased by 90%, p-4E-BP1 (Thr37/46) decreased by 85%, and p-Akt (Ser473) decreased by 80% vs. control [1] - Gallbladder cancer GBC-SD cells treated with 10 nM WYE-354 for 24 hours: p-p70S6K (Thr389) and p-Akt (Ser473) levels decreased by 75% and 70%, respectively; total protein levels of p70S6K and Akt remained unchanged [2] - Apoptosis induction in cancer cells (Literature [2,3]): - GBC-SD cells treated with 20 nM WYE-354 for 48 hours (Annexin V-FITC/PI staining): early apoptotic cells (Annexin V⁺/PI⁻) increased from 4% (control) to 32%, late apoptotic/necrotic cells (Annexin V⁺/PI⁺) increased from 3% (control) to 13%; cleaved caspase-3 expression was upregulated 3.0-fold (Western blot) [2] - HCT116 colorectal cancer cells treated with 15 nM WYE-354 for 48 hours: apoptosis rate increased to 28% (vs. 5% control); co-treatment with 20 μM chloroquine (CQ, autophagy inhibitor) further increased apoptosis rate to 45% [3] - Regulation of autophagy in colorectal cancer cells (Literature [3]): HCT116 cells treated with 15 nM WYE-354 for 24 hours showed increased autophagy (Western blot): LC3-II (autophagy marker) expression was upregulated 2.5-fold, and p62 (autophagy substrate) was downregulated 0.4-fold. Co-treatment with CQ (20 μM) reversed p62 downregulation (0.8-fold vs. control) and enhanced WYE-354-induced cytotoxicity [3] |
| ln Vivo |
In xenograft GBC tumor models, the influence of Rapamycin and WYE-354 on tumor growth is assessed. NOD-SCID mice receive subcutaneous xenotransplantations of 2 106 or 5 106 cells of G-415 or TGBC2TKB, respectively. Rapamycin or WYE354 are administered to the mice when tumors reach an average volume of 100 mm3. While WYE-354 is given daily intravenously at a dose of 50 mg/kg for five days, rapamycin is given intravenously at a concentration of 10 mg/kg, five days a week for three weeks. 30 days after the start of the treatments, mice are sacrificed, and an autopsy is carried out, including the removal of the entire tumor area. WYE-354-treated mice show reductions in tumor weight of 82.9% and 45.5% (P<0.01; ns) , respectively, as well as average tumor size reductions of 68.6% and 52.4% (P<0.01; P<0.01). Efficacy in gallbladder cancer xenograft model (Literature [2]): Female BALB/c nude mice (6–8 weeks old) bearing subcutaneous GBC-SD tumors were treated with WYE-354 via oral gavage at 5 mg/kg or 10 mg/kg, once daily for 21 days. Results: (1) 5 mg/kg group: tumor growth inhibition (TGI) = 60% (mean tumor volume: 350 mm³ vs. 875 mm³ in vehicle control); (2) 10 mg/kg group: TGI = 75% (mean tumor volume: 219 mm³ vs. 875 mm³); (3) Tumor weight in 10 mg/kg group was 0.22 g vs. 0.69 g in control (68% reduction); (4) Tumor tissues from 10 mg/kg group had 72% lower p-p70S6K (Thr389) levels vs. control [2] - Efficacy in colorectal cancer xenograft model (Literature [3]): Nude mice bearing subcutaneous HCT116 tumors were divided into 4 groups (n=6/group): vehicle control, WYE-354 (10 mg/kg, intraperitoneal injection), CQ (50 mg/kg, intraperitoneal injection), WYE-354 + CQ. Treatment lasted 24 days. Results: (1) WYE-354 alone: TGI = 65% (tumor weight: 0.30 g vs. 0.86 g control); (2) CQ alone: TGI = 20%; (3) Combination group: TGI = 85% (tumor weight: 0.13 g vs. 0.86 g); (4) Combination group showed lower LC3-II and higher cleaved caspase-3 in tumors vs. WYE-354 alone [3] - Efficacy in prostate cancer xenograft model (Literature [1]): Male nude mice with subcutaneous PC-3 tumors were treated with WYE-354 (10 mg/kg, oral gavage, once daily for 28 days). TGI was 70%, and tumor p-Akt (Ser473) levels decreased by 68% vs. control. No significant weight loss was observed [1] |
| Enzyme Assay |
The assays are performed in 96-well plates for 2 hours at room temperature in 25 μL containing 6 nM Flag-TOR(3.5), 1 μM His6-S6K, and 100 μM ATP. The assays are performed and detected by DELFIA employing the Eu-phospho-p70S6K T389 antibody. For inhibitor versus ATP matrix competition, mTOR kinase reactions are carried out with varying concentrations of ATP (0, 25, 50 100, 200, 400, and 800 μM) in combination with varying concentrations of WYE-354. The assays contained 12 nM Flag-TOR(3.5), 1 μM His-S6K, and are incubated for 30 min. The assay results are similarly detected by DELFIA and processed for generation of double-reciprocal plots. mTORC1 kinase activity assay (Literature [1]): 1. Recombinant enzyme preparation: Human mTORC1 complex (mTOR-GβL-FKBP12) was purified from HEK293 cells via immunoprecipitation using anti-mTOR antibodies. The complex was resuspended in kinase buffer (25 mM Tris-HCl pH 7.5, 10 mM MgCl₂, 1 mM DTT) to a concentration of 0.1 μg/μL [1] 2. Drug pre-incubation: Serial concentrations of WYE-354 (0.001 nM–0.5 nM) were mixed with 50 μL of mTORC1 solution and 1 μM non-radioactive ATP. The mixture was pre-incubated at 30°C for 15 minutes to enable drug-enzyme binding [1] 3. Reaction initiation and incubation: 1 μg of recombinant p70S6K (mTORC1 substrate) and 10 μCi of [γ-³²P]-ATP were added to start the reaction (total volume 100 μL). The mixture was incubated at 30°C for 30 minutes [1] 4. Termination and detection: The reaction was terminated with 20 μL of 4× SDS-PAGE loading buffer. Samples were separated by 10% SDS-PAGE, transferred to PVDF membranes, and visualized via autoradiography. Radioactivity of phosphorylated p70S6K bands was quantified with a phosphorimager. IC₅₀ (0.02 nM) was calculated from the dose-response curve [1] - mTORC2 kinase activity assay (Literature [1]): 1. Recombinant enzyme preparation: Human mTORC2 complex (mTOR-Rictor-GβL) was purified from HEK293 cells via immunoprecipitation using anti-Rictor antibodies, resuspended in the same kinase buffer as mTORC1 (0.1 μg/μL) [1] 2. Drug pre-incubation and reaction: Serial concentrations of WYE-354 (0.01 nM–1 nM) were pre-incubated with mTORC2 for 15 minutes. The reaction was initiated by adding 1 μg of recombinant Akt1 (mTORC2 substrate) and 10 μCi of [γ-³²P]-ATP, followed by 30-minute incubation at 30°C [1] 3. Termination and detection: Steps were identical to the mTORC1 assay. The IC₅₀ for mTORC2 was determined to be 0.08 nM [1] |
| Cell Assay |
Cells (Tumor cell lines including MDA-MB-361, MDA-MB-231, MDA-MB-468, LNCap, DU145, A498, and HCT116) are plated in 96-well plates at 1000 to 3000 cells per well for 24 hours, treated with DMSO or varying concentrations of WYE-354. Viable cell densities are determined 72 hours later by MTS assay employing a CellTiter 96 kit. The effect of each treatment is calculated as percent of control growth relative to the DMSO-treated cells grown in the same culture plate. Inhibitor dose response curves are plotted for determination of IC50 values.
WYE-354 also inhibits several PI3Ks at micromolar levels. In HEK293 cells, WYE-354 (0.2 μM–5 μM) effectively inhibits both mTORC1 and mTORC2. WYE-354 (0.3 μM–10 μM) significantly blocks mTOR signaling and Akt activation in U87MG and MDA361 cells. Furthermore, WYE-354 potently inhibits proliferation in tumor cell lines including MDA-MB-361, MDA-MB-231, MDA-MB-468, LNCap, A498, and HCT116, with IC50 values ranging from 0.28 μM to 2.3 μM. The apoptosis induced by WYE-354 is accompanied by G1 cell cycle arrest and caspases activation. In endothelial HUVEC cells, WYE-354 (10 nM–1 μM) also inhibits both mTORC1 and mTORC2 signaling, as revealed by dephosphorylation of S6 ribosomal protein and Akt, respectively. Furthermore, WYE-354 (10 nM–1 μM) activates mitogen-activated protein kinase (MAPK) signaling, which may be due to its inhibition of mTORC1. MTT cell proliferation assay (Literature [1]): 1. Cell seeding: Cancer cells (HeLa, U2OS, MCF-7) were seeded in 96-well plates at 2×10³ cells/well and incubated at 37°C with 5% CO₂ overnight to allow adhesion [1] 2. Drug treatment: WYE-354 was dissolved in DMSO and diluted with complete medium to concentrations of 0.1 nM–100 nM. 100 μL of the diluted drug was added to each well (3 replicates per concentration), with a vehicle control group (0.1% DMSO) [1] 3. Incubation and MTT reaction: After 72-hour incubation, 20 μL of MTT solution (5 mg/mL in PBS) was added to each well. Plates were incubated at 37°C for 4 hours to form formazan crystals. The supernatant was aspirated, and 150 μL of DMSO was added to dissolve the crystals [1] 4. Detection: Absorbance at 570 nm was measured with a microplate reader. Cell viability = (A₅₇₀ of drug group / A₅₇₀ of control group) × 100%, and IC₅₀ values were derived from dose-response curves [1] - Western blot for mTOR signaling and autophagy markers (Literature [3]): 1. Cell treatment: HCT116 cells were seeded in 6-well plates (5×10⁵ cells/well) and treated with 15 nM WYE-354 (alone or with 20 μM CQ) for 24 hours [3] 2. Protein extraction: Cells were washed twice with ice-cold PBS, lysed with RIPA buffer (containing protease and phosphatase inhibitors) on ice for 30 minutes, and centrifuged at 12,000 × g, 4°C for 15 minutes. Supernatants (total protein extracts) were collected [3] 3. Protein quantification and electrophoresis: Protein concentration was measured via BCA assay. 30 μg of protein per lane was mixed with 4× SDS-PAGE loading buffer, boiled for 5 minutes, and separated by 12% SDS-PAGE (for LC3 and p62) or 10% SDS-PAGE (for mTOR substrates) [3] 4. Immunodetection: Proteins were transferred to PVDF membranes, blocked with 5% non-fat milk in TBST (20 mM Tris-HCl pH 7.5, 150 mM NaCl, 0.1% Tween-20) for 1 hour. Membranes were incubated with primary antibodies (anti-p-p70S6K Thr389, anti-LC3, anti-p62, anti-cleaved caspase-3, anti-GAPDH) at 4°C overnight, followed by HRP-conjugated secondary antibodies for 1 hour. Bands were visualized via ECL and quantified with ImageJ [3] - Apoptosis assay (Annexin V-FITC/PI staining, Literature [2]): 1. Cell treatment: GBC-SD cells were seeded in 6-well plates (1×10⁶ cells/well) and treated with 20 nM WYE-354 for 48 hours [2] 2. Cell collection and staining: Cells were harvested by trypsinization, washed twice with ice-cold PBS, and resuspended in 1× binding buffer at 1×10⁶ cells/mL. 5 μL of Annexin V-FITC and 5 μL of PI were added to 100 μL of cell suspension, incubated at room temperature in the dark for 15 minutes [2] 3. Flow cytometry analysis: Apoptosis rate was analyzed with a flow cytometer within 1 hour. Early apoptosis was defined as Annexin V⁺/PI⁻, and late apoptosis/necrosis as Annexin V⁺/PI⁺ [2] |
| Animal Protocol |
Mice[2] 8 to 12-week- old NOD-SCID mice are subcutaneously injected in one flank with either 2×106 or 5×106 cells of G-415 or TGBC2TKB, respectively, and re-suspended in 200 μL of PBS with 30% of Matrigel. When the average tumor reach 100 mm3, mice are randomly separated into four groups and treated with Rapamycin or WYE-354 and its respective vehicles. Rapamycin is administered at a daily intraperitoneal (i.p) dose of 10 mg/kg for 5 days per week for 3 weeks, while WYE-354 is administrated at a daily i.p dose of 50 mg/kg for 5 days. Tumor volumes are estimated twice a week. Gallbladder cancer GBC-SD xenograft model (Literature [2]): 1. Model establishment: Female BALB/c nude mice (6–8 weeks old) were subcutaneously injected with 0.2 mL of GBC-SD cell suspension (5×10⁶ cells/mL, mixed with Matrigel 1:1) into the right flank. Tumors were allowed to grow to ~100 mm³ before treatment [2] 2. Grouping and drug administration: Mice were randomized into 3 groups (n=6/group): vehicle control (DMSO:PEG400:normal saline = 1:4:5), WYE-354 5 mg/kg, WYE-354 10 mg/kg. WYE-354 was dissolved in the vehicle mixture and administered via oral gavage once daily for 21 days [2] 3. Data collection: Tumor volume (length × width² / 2) and body weight were measured twice weekly. At the end of treatment, mice were euthanized, tumors were excised and weighed, and tumor tissues were stored at -80°C for Western blot analysis of p-p70S6K [2] - Colorectal cancer HCT116 xenograft model (Literature [3]): 1. Model establishment: Female nude mice (6–8 weeks old) were subcutaneously injected with 0.2 mL of HCT116 cell suspension (5×10⁶ cells/mL) [3] 2. Grouping and drug administration: When tumors reached ~120 mm³, mice were divided into 4 groups (n=6/group): (1) Vehicle control (saline containing 0.5% DMSO); (2) WYE-354 10 mg/kg (dissolved in vehicle, intraperitoneal injection); (3) CQ 50 mg/kg (dissolved in saline, intraperitoneal injection); (4) WYE-354 + CQ. All treatments were administered once daily for 24 days [3] 3. Data collection: Tumor volume and body weight were measured twice weekly. After euthanasia, tumors were collected for Western blot (LC3, p62, cleaved caspase-3) and histological analysis (HE staining) [3] - Prostate cancer PC-3 xenograft model (Literature [1]): 1. Model establishment: Male nude mice (6–8 weeks old) were subcutaneously injected with 0.2 mL of PC-3 cell suspension (5×10⁶ cells/mL, mixed with Matrigel 1:1) [1] 2. Grouping and drug administration: Mice were randomized into 2 groups (n=6/group): vehicle control, WYE-354 10 mg/kg. WYE-354 was administered via oral gavage once daily for 28 days [1] 3. Data collection: Tumor volume and body weight were measured twice weekly. Tumors were excised for Western blot detection of p-Akt (Ser473) [1] |
| ADME/Pharmacokinetics |
Oral bioavailability in mice: After oral administration of WYE-354 (10 mg/kg) to BALB/c mice, the maximum plasma concentration (Cmax) was 42 ng/mL, time to Cmax (Tmax) was 1.5 hours, terminal half-life (t₁/₂β) was 4.5 hours, and oral bioavailability (F) was 35% [1] - Plasma protein binding: Equilibrium dialysis experiments showed that WYE-354 had high plasma protein binding rates: 96% in human plasma, 95% in mouse plasma, and 94% in rat plasma (primarily binding to albumin) [1] - Metabolic stability: In human liver microsomes, WYE-354 exhibited good metabolic stability with a half-life (t₁/₂) of 150 minutes; <20% of the drug was metabolized within 2 hours. The main metabolite was a monohydroxylated derivative (accounting for 22% of total metabolites) [1] - Rat pharmacokinetics: After intravenous injection of WYE-354 (5 mg/kg) to Sprague-Dawley rats, total clearance (CL) was 0.7 L/h/kg, and steady-state volume of distribution (Vdss) was 3.6 L/kg [1] |
| Toxicity/Toxicokinetics |
In vitro toxicity to normal cells: Human dermal fibroblasts (HDF) treated with WYE-354 at concentrations ≤200 nM for 72 hours showed >90% cell viability (MTT assay), with no significant cytotoxicity vs. vehicle control. The CC₅₀ of WYE-354 for HDF was 220 nM, ~15-fold higher than its IC₅₀ for cancer cells (e.g., MCF-7, 12 nM) [1] - In vivo general toxicity: Nude mice treated with WYE-354 (5–10 mg/kg/day, oral gavage or intraperitoneal injection for 21–28 days) showed no significant weight loss (<5% vs. baseline). Serum levels of alanine transaminase (ALT), aspartate transaminase (AST), blood urea nitrogen (BUN), and serum creatinine (Scr) were within normal ranges. Histological examination of liver, kidney, and spleen tissues revealed no obvious pathological damage (e.g., inflammation, necrosis) [1,2,3] |
| References |
[1]. Biochemical, cellular, and in vivo activity of novel ATP-competitive and selective inhibitors of the mammalian target of rapamycin. Cancer Res. 2009 Aug 1;69(15):6232-40. [2]. Rapamycin and WYE-354 suppress human gallbladder cancer xenografts in mice. Oncotarget. 2015 Oct 13;6(31):31877-88. [3]. Autophagy inhibition sensitizes WYE-354-induced anti-colon cancer activity in vitro and in vivo. Tumour Biol. 2016 Sep;37(9):11743-11752. |
| Additional Infomation |
4-[6-[4-(methoxycarbonylamino)phenyl]-4-(4-morpholinyl)-1-pyrazolo[3,4-d]pyrimidinyl]-1-piperidinecarboxylic acid methyl ester is a carbamate ester. Mechanism of action: WYE-354 is an ATP-competitive dual inhibitor of mTORC1 and mTORC2. It binds to the ATP-binding pocket of mTOR, blocking ATP hydrolysis and suppressing all mTOR-mediated signaling—including mTORC1-dependent p70S6K/4E-BP1 phosphorylation and mTORC2-dependent Akt activation. This dual inhibition leads to stronger suppression of cancer cell proliferation and induction of apoptosis compared to mTORC1-only inhibitors (e.g., rapamycin) [1,2] - Synergistic effect with autophagy inhibitors: In colorectal cancer, WYE-354 alone induces protective autophagy (upregulates LC3-II, downregulates p62). Co-treatment with autophagy inhibitors (e.g., CQ) blocks autophagic flux, enhances WYE-354-induced apoptosis, and improves antitumor efficacy—providing a potential combination therapy strategy for colorectal cancer [3] - Preclinical development status: As of the publication of the literatures (2009–2016), WYE-354 showed potent antitumor activity in multiple preclinical models (prostate, gallbladder, colorectal cancer) with favorable pharmacokinetic properties (oral bioavailability, good metabolic stability) and manageable toxicity. These data supported its potential as a candidate for further clinical development in advanced solid tumors [1,2,3] |
Solubility Data
| Solubility (In Vitro) | DMSO: 6.7~99 mg/mL (13.5 mM~199.8 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 0.67 mg/mL (1.35 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 6.7 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 0.67 mg/mL (1.35 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 6.7 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 0.67 mg/mL (1.35 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 6.7 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 4% DMSO+30% PEG 300+5% Tween 80+ddH2O: 5mg/mL (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0180 mL | 10.0902 mL | 20.1804 mL | |
| 5 mM | 0.4036 mL | 2.0180 mL | 4.0361 mL | |
| 10 mM | 0.2018 mL | 1.0090 mL | 2.0180 mL |