Physicochemical Properties
| Molecular Formula | C22H28N2O3 |
| Molecular Weight | 368.47 |
| Exact Mass | 368.21 |
| CAS # | 510-22-5 |
| PubChem CID | 73255 |
| Appearance | White to off-white solid powder |
| Density | 1.25g/cm3 |
| Boiling Point | 518.4ºC at 760mmHg |
| Melting Point | 223-224ºC (dec.) |
| Flash Point | 267.3ºC |
| Index of Refraction | 1.631 |
| LogP | 3.201 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 27 |
| Complexity | 598 |
| Defined Atom Stereocenter Count | 4 |
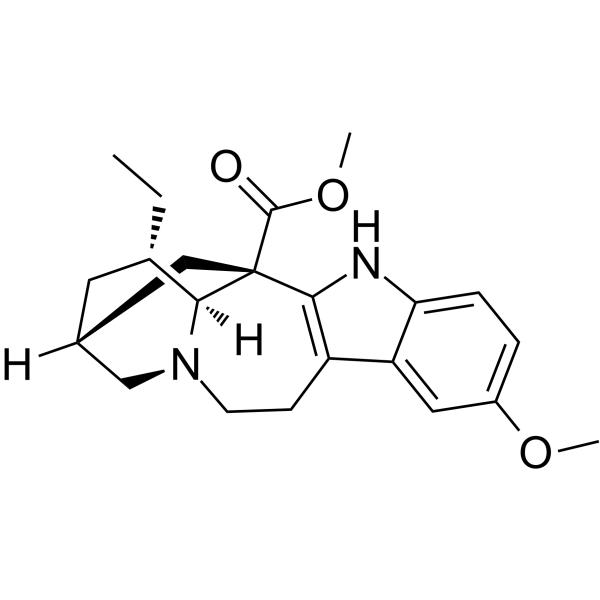
| SMILES | CC[C@H]1C[C@H]2C[C@@]3([C@H]1N(C2)CCC4=C3NC5=C4C=C(C=C5)OC)C(=O)OC |
| InChi Key | MMAYTCMMKJYIAM-RUGRQLENSA-N |
| InChi Code | InChI=1S/C22H28N2O3/c1-4-14-9-13-11-22(21(25)27-3)19-16(7-8-24(12-13)20(14)22)17-10-15(26-2)5-6-18(17)23-19/h5-6,10,13-14,20,23H,4,7-9,11-12H2,1-3H3/t13-,14-,20-,22+/m0/s1 |
| Chemical Name | methyl (1S,15S,17S,18S)-17-ethyl-7-methoxy-3,13-diazapentacyclo[13.3.1.02,10.04,9.013,18]nonadeca-2(10),4(9),5,7-tetraene-1-carboxylate |
| Synonyms | Voacangine; (-)-Voacangine; Carbomethoxyibogaine; 510-22-5; CHEMBL182120; 12-Methoxyibogamine-18-carboxylic acid methyl ester; methyl (1S,15S,17S,18S)-17-ethyl-7-methoxy-3,13-diazapentacyclo[13.3.1.02,10.04,9.013,18]nonadeca-2(10),4(9),5,7-tetraene-1-carboxylate; 9SY76D3YUK; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | EC50: 8 μM (TRPA1)[1]. IC50: 9 μM (TRPM8)[1]. IC50: 50 μM (TRPV1)[1]. IC50: 7 μM (icilin)[1] |
| ln Vitro | On TRPA1-expressing cells, vacangine (100 μM; HEK cells) causes a Ca2+ influx, but not on the other TRPs. Voacangine inhibits the activation of TRPV1 induced by capsaicin (CAP) as well as the activation of TRPM8 induced by icilin and menthol. Voacangine reduces the activation of TRPV1 produced by CAP. Voacangine exhibits a CAP response inhibition that is dose-dependent. Voacangine inhibits CAP on TRPV1 in a competitive manner. Voacangine acts at the same recognition site on hTRPV1 as CAP, making it a competitive antagonist. Voacangine is an agonist for TRPA1 but an antagonist for TRPV1 and TRPM8. Voacangine specifically prevents TRPV1 from being activated by heat and CAP. The first TRPM8 antagonist that exists naturally and competes with menthol is vacangine. Voacangine specifically prevents TRPM8 activation generated by pharmacological agonists. [1]. At an IC50 of 18 μM, vacangine prevents HUVECs from proliferating while having no cytotoxic effects. In a dose-dependent way, vocacangine reduces the expression levels of VEGF, the target gene of hypoxia inducible factor-1α[2]. |
| ln Vivo | Voacangine strongly inhibits angiogenesis in vitro, including chemoinvasion and tube formation produced by VEGF[2]. |
| Enzyme Assay | Voacangine (1) is an alkaloid found in the root bark of Voacanga africana. Our previous work has suggested that 1 is a novel transient receptor potential vanilloid type 1 (TRPV1) antagonist. In this study, the agonist and antagonist activities of 1 were examined against thermosensitive TRP channels. Channel activity was evaluated mainly using TRP channel-expressing HEK cells and calcium imaging. Herein, it was shown that 1 acts as an antagonist for TRPV1 and TRPM8 but as an agonist for TRPA1 (EC50, 8 μM). The compound competitively blocked capsaicin binding to TRPV1 (IC50, 50 μM). Voacangine (1) competitively inhibited the binding of menthol to TRPM8 (IC50, 9 μM), but it showed noncompetitive inhibition against icilin (IC50, 7 μM). Moreover, the compound selectively abrogated chemical agonist-induced TRPM8 activation and did not affect cold-induced activation. Among these effects, the TRPM8 inhibition profile is unique and noteworthy, because to date no studies have reported a menthol competitive inhibitor of TRPM8 derived from a natural source. Furthermore, this is the first report of a stimulus-selective TRPM8 antagonist. Accordingly, 1 may contribute to the development of a novel class of stimulus-selective TRPM8 blockers.[1] |
| Cell Assay | The crude extract of V. africana inhibits the proliferation of HUVECs in a dose-dependent manner (Fig. 1B). Voacangine is a known principal component of this extract. Therefore, we investigated whether voacangine is responsible for the observed anti-proliferative activity of the extract. As shown in Fig. 1C, voacangine inhibited cell growth at 10 μM. Notably, it exerted a greater growth inhibition effect on HUVECs than on other normal and cancer cell lines (Table 1).[2] |
| References |
[1]. Activation and inhibition of thermosensitive TRP channels by voacangine, an alkaloid present in Voacanga africana, an African tree. J Nat Prod. 2014;77(2):285-297. [2]. A natural small molecule voacangine inhibits angiogenesis both in vitro and in vivo. Biochem Biophys Res Commun. 2012;417(1):330-334. |
| Additional Infomation | (-)-voacangine is a monoterpenoid indole alkaloid with formula C22H28N2O3, isolated from several plant species. It has a role as an angiogenesis inhibitor, an antineoplastic agent and a plant metabolite. It is a monoterpenoid indole alkaloid, a tertiary amino compound, a methyl ester, an organic heteropentacyclic compound and an alkaloid ester. It is a conjugate base of a (-)-voacangine(1+). |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7139 mL | 13.5696 mL | 27.1393 mL | |
| 5 mM | 0.5428 mL | 2.7139 mL | 5.4279 mL | |
| 10 mM | 0.2714 mL | 1.3570 mL | 2.7139 mL |