VPS34-IN2 (PIK-III; Vps34-PIK-III) is a novel, potent and selective inhibitor of VPS34 (IC50 = 18 nM) with the ability to modulate autophagy in Vivo. PIK-III blocks autophagy and uncovers a role for NCOA4 in ferritin degradation and iron homeostasis in vivo. In contrast to related kinases like PI(3)K, PIK-III binds to a particular hydrophobic pocket. The acute inhibition of autophagy and de novo lipidation of LC3 by PIK-III results in the stabilization of autophagy substrates.
Physicochemical Properties
| Molecular Formula | C17H17N7 |
| Molecular Weight | 319.3638 |
| Exact Mass | 319.155 |
| Elemental Analysis | C, 63.93; H, 5.37; N, 30.70 |
| CAS # | 1383716-40-2 |
| Related CAS # | 1383716-40-2 |
| PubChem CID | 67983123 |
| Appearance | White to off-white solid powder |
| LogP | 3.261 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 24 |
| Complexity | 396 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | N1C(N([H])[H])=NC([H])=C(C2C([H])=C([H])N=C(N([H])C3C([H])=C([H])N=C([H])C=3[H])N=2)C=1C([H])([H])C1([H])C([H])([H])C1([H])[H] |
| InChi Key | XXSDLQLNIVFIJI-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C17H17N7/c18-16-21-10-13(15(23-16)9-11-1-2-11)14-5-8-20-17(24-14)22-12-3-6-19-7-4-12/h3-8,10-11H,1-2,9H2,(H2,18,21,23)(H,19,20,22,24) |
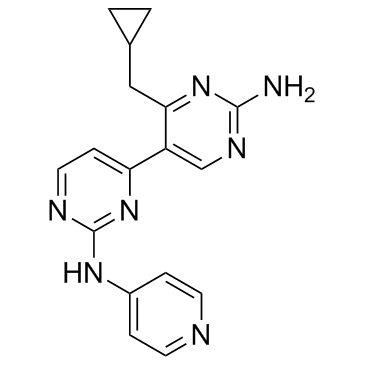
| Chemical Name | 4-(cyclopropylmethyl)-5-[2-(pyridin-4-ylamino)pyrimidin-4-yl]pyrimidin-2-amine |
| Synonyms | Vps34 PIK-III; Vps34-PIK III; VPS34-IN2; VPS34-IN 2; Vps34-PIK-III; PIK-III; PIK III; PIKIII; VPS34-IN-2 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Vps34 (IC50 = 18 nM); PI(3)Kδ (IC50 = 1.2 μM); PI(3)Kγ (IC50 = 3.04 μM); PI(3)Kα (IC50 = 3.96 μM) VPS34 (IC₅₀ = 15 nM)[1] PI3Kδ (IC₅₀ = 820 nM)[1] TAK1 (10-fold less selective than VPS34, IC₅₀ not specified)[1] >280 other kinases including PI3Kα, β, γ (IC₅₀ >>100-fold selectivity over VPS34)[1] |
| ln Vitro |
VPS34 enzymatic function is essential for LC3 lipidation in mammalian cells and PIK-III is a robust inhibitor of autophagy and LC3 lipidation in mammalian cells. In H4 cells, both in basal conditions and when autophagy is induced using the mTOR inhibitor AZD8055, PIK-III reduces the formation of autolysosomes and boosts the cytosolic signal of LC3. PIK-III prevents the removal of mitochondria in a mitophagy model induced by CCCP. In H4 and PSN1 cells, treatment with PIK-III causes LC3-I levels to rise. In Panc10.05 cells, PIK-III raises the levels of LC3-II concurrently with LC3-I, indicating a cell type-specific response[1]. VPS34-IN2 inhibited VPS34 enzymatic activity with an IC₅₀ of 15 nM in a biochemical assay.[1] In a cellular GFP-FYVE reporter assay, which monitors PtdIns(3)P levels as a surrogate for VPS34 activity, VPS34-IN2 showed an IC₅₀ of 25 nM, indicating potent on-target cellular activity.[1] Treatment of DLD1 cells with VPS34-IN2 for 24 hours led to accumulation of autophagy substrates p62, NCOA4, NBR1, NDP52, and FTH1, similar to the known inhibitor PIK-III.[1] In DLD1 cells, treatment with VPS34-IN2 increased the levels of both lipidated (LC3-II) and non-lipidated (LC3-I) forms of LC3, consistent with inhibition of autophagic flux.[1] |
| ln Vivo |
The single-dose, fast-absorbing Vps34-PIK-III (10 mg/kg; valve) has a strong valve biological utilization (F% = 47) and a modest mean systemic clearance (30 mL/min/kg, or around 33% of hepatic blood flow) [1]. In RKO colon cancer tumor-bearing nude mice, oral administration of VPS34-IN2 at 50 mg/kg twice daily for 7 days resulted in a time-dependent accumulation of LC3-II in tumor samples, indicating inhibition of autophagy in vivo.[1] No reduction in tumor volume was observed during this 7-day treatment period.[1] |
| Enzyme Assay |
The biochemical inhibition of VPS34 was measured using a luminescence-based ATP detection assay. The assay determines the IC₅₀ value by measuring the depletion of ATP upon kinase activity.[1] Selectivity profiling against a panel of over 280 kinases (including lipid and protein kinases) was performed to establish the selectivity profile of the compound.[1] |
| Cell Assay |
To determine whether inhibition of VPS34 function impacts autophagy,LC3 and known autophagy substrates such as damaged mitochondria or the autophagy cargo receptor p62 are monitored. H4 cells expressing mCherry-GFP-LC3 are treated overnight with the listed substances, fixed, stained with Hoechst 33342, and imaged by automated acquisition. HeLa cells expressing GFP-Parkin are treated with PIK-III for 12 hours, then added CCCP for another 12 hours. The cells are then fixed, stained for endogenous Tom20, and imaged. A GFP-FYVE reporter assay was used to assess cellular VPS34 inhibition. Cells expressing the reporter were treated with compounds, and the redistribution or intensity of the GFP-FYVE signal, which binds to PtdIns(3)P, was measured to generate IC₅₀ values.[1] For autophagy substrate analysis, DLD1 cells were treated with compounds for 24 hours. Cell lysates were then analyzed by immunoblotting to detect proteins such as p62, NCOA4, and LC3.[1] |
| Animal Protocol |
Animal/Disease Models: C57BL/6 mice[1]. Doses: 10 mg/kg; 2 mg/kg Route of Administration: oral; intravenous (iv) (iv)injection; individual Experimental Results: 1.19 pharmacokinetic/PK/PK parameters of Vps34-PIK-III in C57BL/6 mice [1]. IV (2 mg/kg) PO (10 mg/kg) Tmax (h) 0.7 Cmax (nM) 2994 AUCinf (nM·h) 2855 6725 t1/2 (h) 1.2 CL (mL/min/kg) 30 Vdss ( Liter/kg)1.5F(%)47% The pharmacokinetic profile was determined in C57BL/6 mice. VPS34-IN2 was administered as a single intravenous dose (2 mg/kg) or a single oral dose (10 mg/kg). Blood samples were collected at various time points to determine PK parameters.[1] For the in vivo efficacy/pharmacodynamics study, RKO tumor-bearing nude mice were treated orally with VPS34-IN2 at a dose of 50 mg/kg, twice daily (BID), for 7 days. Tumor samples were collected at the end of the treatment for immunoblot analysis of LC3-II levels.[1] |
| ADME/Pharmacokinetics |
In C57BL/6 mice following a single IV dose (2 mg/kg), the mean systemic clearance (CL) was 30 mL/min/kg, and the volume of distribution at steady state (Vss) was 1.5 L/kg.[1] The terminal half-life (t₁/₂) after IV administration was 1.2 hours.[1] Following a single oral dose (10 mg/kg), the maximum plasma concentration (Cmax) was 2994 nM, and the time to reach Cmax (Tmax) was 0.7 hours.[1] The oral bioavailability (F) was calculated to be 47%.[1] |
| References |
[1]. Potent, Selective, and Orally Bioavailable Inhibitors of VPS34 Provide Chemical Tools to Modulate Autophagy in Vivo. ACS Med Chem Lett. 2015 Nov 13;7(1):72-6. [2]. PIK3C3 regulates the expansion of liver CSCs and PIK3C3 inhibition counteracts liver cancer stem cell activity induced by PI3K inhibitor. Cell Death Dis. 2020 Jun 8;11(6):427. [3]. Selective VPS34 inhibitor blocks autophagy and uncovers a role for NCOA4 in ferritin degradation and iron homeostasis in vivo. Nat Cell Biol. 2014 Nov;16(11):1069-79. |
| Additional Infomation |
VPS34-IN2 (compound 19) was developed through medicinal chemistry optimization of a high-throughput screening hit to achieve high potency and selectivity for VPS34 over other PI3K isoforms and a broad panel of kinases.[1] An X-ray co-crystal structure of VPS34 with VPS34-IN2 confirmed its binding mode. The compound binds to the ATP-binding site, with an aminopyrimidine moiety interacting with the hinge region (Ile685), and a cyclopropyl group occupying a hydrophobic pocket unique to VPS34, contributing to its selectivity.[1] VPS34-IN2 is described as the first disclosed orally bioavailable, selective VPS34 inhibitor shown to inhibit autophagy in vivo, serving as a valuable chemical tool for probing autophagy biology.[1] |
Solubility Data
| Solubility (In Vitro) |
DMSO: 31~63 mg/mL (97~197.3 mM) Ethanol: ~63 mg/mL (~197.3 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.83 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.83 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (7.83 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1313 mL | 15.6563 mL | 31.3126 mL | |
| 5 mM | 0.6263 mL | 3.1313 mL | 6.2625 mL | |
| 10 mM | 0.3131 mL | 1.5656 mL | 3.1313 mL |