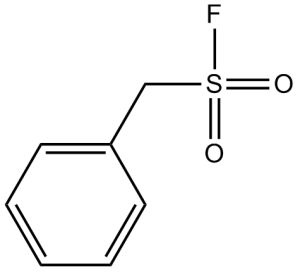
PMSF (full name: Phenylmethanesulfonyl fluoride) is a potent and irreversible inhibitor of serine/cysteine protease, which includes chymotrypsin, thrombin, and trypsin. It may also have analgesic properties. PMSF exhibited antinociception, hypothermia, and immobility as cannabinoid effects.
Physicochemical Properties
| Molecular Formula | C7H7FO2S | |
| Molecular Weight | 174.19 | |
| Exact Mass | 174.015 | |
| Elemental Analysis | C, 48.27; H, 4.05; F, 10.91; O, 18.37; S, 18.41 | |
| CAS # | 329-98-6 | |
| Related CAS # |
|
|
| PubChem CID | 4784 | |
| Appearance | White to off-white solid powder | |
| Density | 1.3±0.1 g/cm3 | |
| Boiling Point | 285.7±19.0 °C at 760 mmHg | |
| Melting Point | 92-95 °C | |
| Flash Point | 126.6±21.5 °C | |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C | |
| Index of Refraction | 1.522 | |
| LogP | 2.33 | |
| Hydrogen Bond Donor Count | 0 | |
| Hydrogen Bond Acceptor Count | 3 | |
| Rotatable Bond Count | 2 | |
| Heavy Atom Count | 11 | |
| Complexity | 199 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | O=S(CC1=CC=CC=C1)(F)=O |
|
| InChi Key | YBYRMVIVWMBXKQ-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C7H7FO2S/c8-11(9,10)6-7-4-2-1-3-5-7/h1-5H,6H2 | |
| Chemical Name | phenylmethanesulfonyl fluoride | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
cysteine protease; chymotrypsin PMSF (Phenylmethylsulfonyl Fluoride) is a reversible inhibitor of serine proteases, including trypsin (Ki = 1.3 μM) and chymotrypsin (Ki = 0.15 μM) [3] - PMSF inhibits calpain (a calcium-dependent cysteine protease with serine protease-like activity) with an IC50 value of 20 μM [4] |
| ln Vitro |
PMSF quickly inactivates purified chymotrypsin from the human pancreas, while human trypsin is less susceptible to inhibition by PMSF. Acetylcholinesterase from human red blood cells is also quickly inhibited by PMSF. (Source: ) At 2 mM, PMSF treatment almost completely inhibits carbachol-stimulated inositol incorporation into phosphatidylinositol (PI) of the longitudinal smooth muscle of the guinea pig ileum. It has no effect on potassium-stimulated inositol incorporation.PMSF causes a temporary inhibition of contraction by both potassium and carbachol, in contrast to its specific inhibition of carbachol-stimulated phosphoinositide turnover.[3] In Trypanosoma brucei, it has been demonstrated that PMSF inhibits the addition of ethanolamine phosphate to glycosylphosphatidylinositol (GPI) intermediates. Moreover, PMSF prevents T. brucei from acylating the inositol residue of GPI intermediates in the bloodstream. In procyclic forms of T. brucei, PMSF inhibits the addition of ethanolamine phosphate and inositol acylation, but not in mammalian HeLa cells. In [4] As an 8-fold higher BSF concentration is required to achieve even a 6-fold slower inactivation than that using PMSF, PMSF is the more reactive inactivator of mouse acetylcholinesterase (AChE). In a cell-free system containing plasma serine proteases, pretreatment with 10 μM PMSF inhibited the activation of plasminogen to plasmin by ~80%, as measured by fibrinolytic assay [1] - In mouse liver homogenates, incubation with 5 μM PMSF reduced the activity of serine-dependent carboxylesterase by ~65% (detected via p-nitrophenyl acetate hydrolysis assay) [2] - In purified enzyme systems, PMSF inhibited trypsin and chymotrypsin in a dose-dependent manner: at 5 μM, trypsin activity was reduced by ~70%, and chymotrypsin activity by ~90% [3] - In isolated rat neonatal cardiomyocytes treated with 100 μM H₂O₂ (oxidative stress), 20 μM PMSF inhibited calpain-mediated troponin I degradation by ~55% (via Western blot) and decreased LDH release (a marker of cell death) by ~40% [4] - In primary cultured rat cortical neurons exposed to oxygen-glucose deprivation (OGD), 15 μM PMSF reduced OGD-induced neuronal death by ~35% (MTT assay) and inhibited the cleavage of α-spectrin (a serine protease substrate) by ~50% (Western blot) [5] - In mouse neuroblastoma N2a cells treated with 50 μM glutamate (excitotoxicity), 10 μM PMSF decreased caspase-3 activation by ~45% (fluorometric assay) and increased cell viability from ~30% (control) to ~60% [6] |
| ln Vivo |
In Sprague-Dawley rats, intraperitoneal injection of PMSF results in dose-dependent analgesia. In rats, PMSF dramatically increases the analgesic effect of beta-endorphin (END). With ED50 values of 86 mg/kg, 224 mg/kg, and 206 mg/kg, respectively, mice receiving intraperitoneal injections of PMSF display a range of cannabinoid effects, including antinociception, hypothermia, and immobility. Anandamide increases its effects on tail-flick response (antinociception), spontaneous activity, and mobility by five, ten, and eight times, respectively, when pretreated with an inactive dose of PMSF (30 mg/kg).[5] When administered 12 hours before PSP, PMSF completely protects hens from organophosphorus ester-induced delayed neuropathy (OPIDN); however, when administered 4 hours after PSP, PMSF intensifies its neurotoxic effects.[6] Five minutes after injecting 1 or 10 mg/kg of 3H-anandamide, pretreatment with PMSF (30 mg/kg, i.p.) elevates anandamide levels in the brain in comparison to the injection of 3H-anandamide plus vehicle. Hens protected against the development of organophosphate-induced delayed neuropathy (OPIDN) are protected by pretreatment with PMSF, which inhibits the degradation of neurofilament (NF) induced by tri-ortho-cresyl phosphate (TOCP). PMSF administration increases the distinct cannabimimetic effects of anandamide (AEA) or Δ(9)-tetrahydrocannabinol (THC) in ICR mice by blocking the fatty acid amide hydrolase enzyme. In male Sprague-Dawley rats (250-300 g) administered intravenous PMSF (10 mg/kg), plasma plasmin activity was inhibited by ~75% within 30 minutes, and this inhibition persisted for ~2 hours; no significant changes in mean arterial pressure were observed [1] - In female ICR mice (20-25 g) given intraperitoneal PMSF (5 mg/kg), liver carboxylesterase activity was reduced by ~60% 1 hour post-administration, and recovered to ~80% of control levels after 6 hours [2] - In a rat model of transient middle cerebral artery occlusion (tMCAO), intracerebroventricular injection of 1 μmol PMSF (administered 30 minutes post-reperfusion) reduced cerebral infarct volume by ~30% (TTC staining, 24 hours post-reperfusion) and improved neurological deficit scores (0-5 scale) by ~1.5 points [5] - In a mouse model of kainic acid-induced seizures (intraperitoneal kainic acid, 30 mg/kg), pretreatment with subcutaneous PMSF (2 mg/kg, 1 hour before kainic acid) reduced the seizure severity score (0-4 scale) from ~3.5 (control) to ~1.8 and decreased the number of TUNEL-positive hippocampal neurons by ~40% (48 hours post-seizure) [6] |
| Enzyme Assay |
Phenylmethanesulfonyl fluoride (PMSF) (2 mM), a putative inhibitor of phosphatidylinositol-specific phospholipase C, almost completely inhibited carbachol-stimulated inositol incorporation into phosphatidylinositol (PI) of longitudinal smooth muscle of guinea pig ileum, while it had no effect on potassium-stimulated inositol incorporation. This suggests that the two stimuli may affect phosphoinositide turnover by different mechanisms, distinguishable by PMSF. In contrast to its specific inhibition of carbachol-stimulated phosphoinositide turnover, PMSF produced a transient inhibition of contraction by both carbachol and potassium. The non-selective effect of PMSF on contraction suggests that it is not the result of its inhibitory effect on phosphoinositide breakdown. PMSF (2 mM) inhibited carbachol-stimulated inositol phosphate accumulation in the presence of Li+ by only 15%-19%, indicating that PMSF inhibition of phosphoinositide turnover was not due to its inhibition of phosphoinositide phosphodiesterase, but to one or more steps following phosphoinositide breakdown[3]. Trypsin/chymotrypsin activity assay: Purified trypsin or chymotrypsin was mixed with their respective chromogenic substrates (Nα-benzoyl-DL-arginine-p-nitroanilide for trypsin, N-benzoyl-L-tyrosine ethyl ester for chymotrypsin) in Tris-HCl buffer (pH 8.0 for trypsin, pH 7.5 for chymotrypsin). PMSF was added at concentrations ranging from 0.1 μM to 10 μM, and the mixture was incubated at 37°C for 60 minutes. Absorbance was measured at 405 nm (trypsin) or 256 nm (chymotrypsin) to calculate enzyme activity; inhibition rates were compared to vehicle controls, and Ki values were determined via Lineweaver-Burk plots [3] - Calpain activity assay: Partially purified calpain from rat skeletal muscle was mixed with a fluorescent substrate (Suc-Leu-Tyr-AMC) in buffer containing 2 mM CaCl₂. PMSF was added at 5-50 μM, and the mixture was incubated at 30°C for 45 minutes. Fluorescence intensity (excitation: 360 nm, emission: 460 nm) was measured to quantify calpain activity; IC50 was calculated by fitting inhibition rates to a dose-response curve [4] |
| Cell Assay |
PMSF blocks T. brucei's ability to acylate the inositol residue of GPI intermediates in the bloodstream. Though it does not prevent fatty acid remodeling in vitro, PMSF inhibits the formation of glycolipid C. Hela cells are not affected by PMSF, but procyclic trypanosomes are inhibited in GPI acylation and ethanolamine phosphatp addition. Cardiomyocyte oxidative stress assay: Neonatal rat cardiomyocytes (P1-P3) were cultured in DMEM with 10% FBS for 48 hours. Cells were pretreated with PMSF (5-40 μM) for 1 hour, then exposed to 100 μM H₂O₂ for 6 hours. After treatment, culture supernatant was collected to measure LDH activity (colorimetric assay, 490 nm); cells were lysed for Western blot analysis using anti-troponin I antibody to assess protein degradation [4] - Cortical neuron OGD assay: Rat embryonic cortical neurons (E18) were cultured in neurobasal medium with B27 supplement for 7 days. Neurons were subjected to OGD (95% N₂ + 5% CO₂, glucose-free medium) for 2 hours, with PMSF (5-25 μM) added during reoxygenation. After 24 hours of reoxygenation, MTT reagent was added to measure cell viability (absorbance 570 nm); cell lysates were analyzed via Western blot with anti-α-spectrin antibody to detect protease-mediated cleavage [5] - Neuroblastoma cell excitotoxicity assay: Mouse N2a cells were cultured in MEM with 10% FBS until 70% confluence. Cells were pretreated with PMSF (2-20 μM) for 1 hour, then exposed to 50 μM glutamate for 24 hours. Caspase-3 activity was measured using a fluorogenic substrate (Ac-DEVD-AMC, excitation 380 nm, emission 460 nm); cell viability was assessed via trypan blue exclusion assay [6] |
| Animal Protocol |
In the experiment, male ICR mice weighing between 18 and 25 g are employed. After dissolving PMSF in sesame oil, 0.1 mL/10 g b.wt. of the solution is injected intraperitoneally. Ten minutes should always elapse between intravenous anandamide or vehicle injections before administering PMSF. Overnight, food and water are not provided to the mice as they become used to the assessment area. Each animal is assessed as follows after receiving anandamide intravenously or a vehicle: tail-flick latency (antinociception) response at 5 minutes and spontaneous (locomotor) activity at 5 to 15 minutes; or core (rectal) temperature at 5 minutes and ring-immobility (catalepsy) at 5 to 10 minutes. Rat plasma plasmin inhibition study: Male Sprague-Dawley rats (250-300 g) were anesthetized with sodium pentobarbital (50 mg/kg, intraperitoneal). PMSF was dissolved in 0.9% physiological saline containing 0.1% DMSO (to improve solubility) and administered via tail vein injection at a dose of 10 mg/kg. Blood samples were collected from the femoral artery at 0, 30, 60, 120, and 180 minutes post-administration to measure plasma plasmin activity via fibrinolytic assay. Mean arterial pressure was monitored continuously via a carotid artery catheter [1] - Mouse liver carboxylesterase study: Female ICR mice (20-25 g) were fasted for 12 hours before experimentation. PMSF was dissolved in 5% DMSO + 95% corn oil and administered via intraperitoneal injection at 5 mg/kg. Mice were euthanized at 1, 3, 6, and 12 hours post-administration; livers were harvested, homogenized in Tris-HCl buffer (pH 7.4), and carboxylesterase activity was measured via p-nitrophenyl acetate hydrolysis [2] - Rat tMCAO model: Male Wistar rats (300-350 g) were anesthetized with isoflurane. Focal cerebral ischemia was induced via middle cerebral artery occlusion (MCAO) using a 6-0 nylon suture. After 60 minutes of occlusion, the suture was removed for reperfusion. Thirty minutes post-reperfusion, PMSF (1 μmol) was dissolved in 5 μL artificial cerebrospinal fluid (aCSF) and injected into the lateral ventricle (stereotaxic coordinates: AP -0.8 mm, ML +1.5 mm, DV -3.5 mm) at 1 μL/min. Vehicle controls received 5 μL aCSF. Twenty-four hours post-reperfusion, rats were euthanized, and brains were stained with TTC to measure infarct volume; neurological deficits were scored based on motor function (0 = normal, 5 = severe deficit) [5] - Mouse kainic acid seizure model: Male C57BL/6 mice (20-25 g) were acclimated for 7 days before experimentation. PMSF was dissolved in 0.1 mL PBS (0.1% DMSO) and administered via subcutaneous injection at 2 mg/kg, 1 hour before intraperitoneal injection of kainic acid (30 mg/kg). Seizure severity was scored every 30 minutes for 6 hours (0 = no seizure, 4 = tonic-clonic seizure). Forty-eight hours post-kainic acid injection, mice were euthanized, and hippocampi were collected for TUNEL staining to count apoptotic neurons [6] |
| Toxicity/Toxicokinetics |
Adverse Effects
Dermatotoxin - Skin burns. Toxic Pneumonitis - Inflammation of the lungs induced by inhalation of metal fumes or toxic gases and vapors. 4784 rat LD50 intraperitoneal 150 mg/kg Nature., 173(33), 1954 [PMID:13119739] 4784 mouse LD50 intraperitoneal 215 mg/kg BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD Life Sciences., 31(1193), 1982 [PMID:6292607] 4784 mouse LD50 oral 200 mg/kg Farmakologiya i Toksikologiya, 39(265), 1976 [PMID:1026506] In male Sprague-Dawley rats, intravenous administration of PMSF at 20 mg/kg (twice the therapeutic dose used in [1]) caused transient tremors in 3 out of 5 rats, which resolved within 1 hour; no mortality was observed [1] - In female ICR mice, intraperitoneal injection of PMSF at 30 mg/kg (6 times the dose in [2]) resulted in 20% mortality (1 out of 5 mice) within 24 hours; surviving mice showed reduced locomotor activity for ~4 hours [2] - In the rat tMCAO model, intracerebroventricular injection of 1 μmol PMSF did not cause significant changes in serum ALT, AST, or creatinine levels (markers of liver and kidney toxicity) 24 hours post-administration [5] - In the mouse kainic acid model, subcutaneous injection of 2 mg/kg PMSF (twice weekly for 2 weeks) did not affect body weight gain or cause abnormal histopathological changes in the liver, kidney, or brain [6] |
| References |
[1]. J Pharmacol Exp Ther. 1969 May;167(1):98-104. [2]. Life Sci. 1982 Sep;31(12-13):1193-6. [3]. Cell Calcium. 1984 Jun;5(3):191-203. [4]. J Biol Chem. 1994 Jul 15;269(28):18694-701. [5]. J Pharmacol Exp Ther. 1997 Dec;283(3):1138-43. [6]. Neurotoxicology. 1999 Oct;20(5):749-59. |
| Additional Infomation |
Phenylmethanesulfonyl fluoride is an acyl fluoride with phenylmethanesulfonyl as the acyl group. It has a role as a serine proteinase inhibitor. It is functionally related to a phenylmethanesulfonic acid. Phenylmethylsulfonyl fluoride has been reported in Ixora coccinea with data available. An enzyme inhibitor that inactivates IRC-50 arvin, subtilisin, and the fatty acid synthetase complex. See also: 4-Toluenesulfonyl fluoride (annotation moved to). Phenylmethanesulfonyl fluoride (PMSF) (2 mM), a putative inhibitor of phosphatidylinositol-specific phospholipase C, almost completely inhibited carbachol-stimulated inositol incorporation into phosphatidylinositol (PI) of longitudinal smooth muscle of guinea pig ileum, while it had no effect on potassium-stimulated inositol incorporation. This suggests that the two stimuli may affect phosphoinositide turnover by different mechanisms, distinguishable by PMSF. In contrast to its specific inhibition of carbachol-stimulated phosphoinositide turnover, PMSF produced a transient inhibition of contraction by both carbachol and potassium. The non-selective effect of PMSF on contraction suggests that it is not the result of its inhibitory effect on phosphoinositide breakdown. PMSF (2 mM) inhibited carbachol-stimulated inositol phosphate accumulation in the presence of Li+ by only 15%-19%, indicating that PMSF inhibition of phosphoinositide turnover was not due to its inhibition of phosphoinositide phosphodiesterase, but to one or more steps following phosphoinositide breakdown.[3] Phenylmethylsulfonyl fluoride (PMSF) has been shown to inhibit the addition of ethanolamine phosphate to glycosylphosphatidylinositol (GPI) intermediates in Trypanosoma brucei (Masterson, W. J., and Ferguson, M. A. J. (1991) EMBO J. 10, 2041-2045). Here we show that the Man3-GlcN-PI intermediate that accumulates in the presence of PMSF can undergo fatty acid remodeling, suggesting that the fatty acid remodeling enzymes are not specific for ethanolamine phosphate-containing GPI intermediates. We also show that PMSF inhibits the acylation of the inositol residue of GPI intermediates in bloodstream form T. brucei. Pulse-chase experiments demonstrate that glycolipid C (ethanolamine-PO4-Man3-GlcN-(acyl)PI) is not an obligatory precursor of glycolipid A (ethanolamine-PO4-Man3-GlcN-PI) and that glycolipid C can be converted to glycolipid A. These data suggest a model where glycolipid C is the terminal product of the GPI biosynthetic pathway, in dynamic equilibrium with glycolipid A. The inhibition of ethanolamine phosphate addition and inositol acylation by PMSF was also observed for procyclic forms of T. brucei but not for mammalian HeLa cells. These results suggest differences between the relevant parasite and mammalian enzymes.[4] Anandamide is an putative endogenous cannabinoid ligand that produces pharmacological effects similar to those of Delta9-tetrahydrocannabinol, the principle psychoactive constituent in marijuana. There is considerable evidence that the enzyme inhibitor phenylmethylsulfonyl fluoride (PMSF) is capable of altering the actions of anandamide in vitro by blocking its metabolism. Therefore, studies were conducted in mice to determine whether PMSF could produce cannabinoid effects by altering endogenous levels of anandamide as well as determining whether PMSF could potentiate the effects of exogenously administered anandamide. Mice receiving i.p. injections of PMSF exhibited cannabinoid effects that included antinociception, hypothermia and immobility with ED50 values of 86, 224 and 206 mg/kg, respectively. Spontaneous activity was reduced at doses greater than 100 mg/kg. However, none of these effects was blocked by the cannabinoid antagonist SR 141716A. On the other hand, pretreatment with an inactive dose of PMSF (30 mg/kg) potentiated the effects of anandamide on tail-flick response (antinociception), spontaneous activity and mobility by 5-, 10- and 8-fold, respectively. PMSF did not alter anandamide's hypothermic effects. Overall, these findings with PMSF underscore the importance of metabolism in the actions of anandamide. It still must be established whether metabolites of anandamide contribute to its pharmacological activity. [5] The serine/cysteine protease inhibitor phenylmethylsulfonyl fluoride (PMSF) has been used both to promote and to protect against neuropathic events of organophosphorus-induced delayed neuropathy (OPIDN) in hens (Veronesi and Padilla, 1985; Pope and Padilla, 1990; Lotti et al., 1991; Pope et al., 1993; Randall et al., 1997). This study is the first to expand upon this work by using high resolution microscopy provided by epoxy resin embedding and thin sectioning to evaluate neuropathological manifestations of promotion and protection, and to correlate them with associated clinical modifications. To evaluate dose-related effects of OPIDN, single phenyl saligenin phosphate (PSP) dosages of 0.5, 1.0, or 2.5 mg/kg were administered to adult hens. PMSF (90 mg/kg) was given either 4 hours after (for promotion) or 12 hours prior to (for protection) PSP administration. Clinical signs and pathologic changes in the biventer cervicis nerve, which is uniquely sensitive to OPIDN (El-Fawal et al., 1988), were monitored. PSP alone, 2.5 mg/kg, caused severe OPIDN (terminal clinical score 7.5 +/- 1.0 [0-8 scale]; neuropathology score 2.7 +/- 0.3 [0-4 scale, based on myelinated fiber degeneration]). PMSF given 12 hours prior to PSP gave complete protection (clinical and neuropathology scores of 0; p<0.0001 compared to PSP alone). Signs and lesions of OPIDN were absent following 0.5 mg/kg PSP alone, but PMSF given 4 hours after PSP potentiated its neurotoxic effects (all hens had clinical scores of 4.0 and the average neuropathology score was 3.5 +/- 0.3; p<0.0001 compared to PSP alone). Although quantitative differences were noted, qualitative differences among nerves from hens with OPIDN were not evident, either with light or electron microscopy. At the time of sacrifice, there was a statistically linear relationship (r2 = 0.76) between the clinical scores on the last day of observation and the neuropathology scores (p<0.0001). This study demonstrates that the degree of peripheral nerve myelinated fiber degeneration correlates with clinical deficits in PMSF-induced potentiation of and protection against OPIDN.[6] PMSF is a synthetic, irreversible (at high concentrations) or reversible (at low concentrations) serine protease inhibitor, widely used as a research tool to study the role of serine proteases in enzymatic reactions, cell signaling, and pathological processes (e.g., inflammation, tissue damage) [3,4] - Early studies ([1], [2]) focused on the ability of PMSF to inhibit plasma and tissue serine proteases (e.g., plasmin, carboxylesterase), providing a basis for its application in studying coagulation and metabolic pathways [1,2] - In cardiovascular research, PMSF is used to inhibit calpain (a serine protease-related enzyme) and reduce myocardial cell damage induced by oxidative stress [4] - In neuroscience, PMSF exhibits neuroprotective effects in models of cerebral ischemia and excitotoxicity by inhibiting serine protease-mediated neuronal death, supporting its use as a tool to investigate neuro degenerative disease mechanisms [5,6] - PMSF is unstable in aqueous solutions (half-life ~30 minutes at neutral pH), requiring fresh preparation before use in experiments [3] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2 mg/mL (11.48 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2 mg/mL (11.48 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2 mg/mL (11.48 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 20 mg/mL (114.82 mM) in Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.7409 mL | 28.7043 mL | 57.4086 mL | |
| 5 mM | 1.1482 mL | 5.7409 mL | 11.4817 mL | |
| 10 mM | 0.5741 mL | 2.8704 mL | 5.7409 mL |