Physicochemical Properties
| Molecular Formula | C11H20O2 |
| Molecular Weight | 184.2753 |
| Exact Mass | 184.146 |
| CAS # | 112-38-9 |
| Related CAS # | 10-Undecenoic acid zinc salt;557-08-4 |
| PubChem CID | 5634 |
| Appearance | Off-white to light yellow <23°C powder,>25°C liquid |
| Density | 0.9±0.1 g/cm3 |
| Boiling Point | 300.8±0.0 °C at 760 mmHg |
| Melting Point | 23-25 °C(lit.) |
| Flash Point | 148.9±0.0 °C |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C |
| Index of Refraction | 1.456 |
| LogP | 3.99 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 13 |
| Complexity | 141 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O([H])C(C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])=C([H])[H])=O |
| InChi Key | FRPZMMHWLSIFAZ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C11H20O2/c1-2-3-4-5-6-7-8-9-10-11(12)13/h2H,1,3-10H2,(H,12,13) |
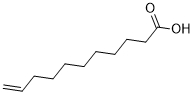
| Chemical Name | undec-10-enoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Intestinal permeation enhancer. [1] |
| ln Vitro |
The sodium salt of 10-undecylenic acid (uC₁₁) was evaluated as an intestinal permeation enhancer in Caco-2 cell monolayers. At 5–10 mM, uC₁₁ reduced transepithelial electrical resistance (TEER) and increased the apparent permeability (Papp) of [¹⁴C]-mannitol and FITC-dextrans (FD4, FD10). Its efficacy was comparable to sodium caprate (C₁₀) and superior to the saturated analogue sodium undecylenate (C₁₁) in terms of lower cytotoxicity. [1] MTT assay in Caco-2 cells showed that uC₁₁ and C₁₀ had similar IC₅₀ values (~5 mM after 24 h exposure), while C₁₁ was more cytotoxic (IC₅₀ < 2.5 mM). [1] High-content analysis (HCA) in Caco-2 cells revealed that uC₁₁ (8.5 mM, 60 min) increased plasma membrane permeability (PMP), intracellular Ca²⁺, and nuclear intensity, indicating a surfactant-like effect correlating with enhanced paracellular flux. [1] In isolated rat colonic mucosa mounted in Ussing chambers, uC₁₁ (10–20 mM) reduced TEER and increased Papp of [¹⁴C]-mannitol and FD4, with efficacy similar to C₁₀ but less mucosal damage than C₁₁. [1] |
| ln Vivo |
In situ instillation of uC₁₁ (100 mM) with FD4 in rat jejunal and colonic loops significantly increased FD4 bioavailability (BA) compared to control. Enhancement ratios were ~10 in jejunum and ~30 in colon, comparable to C₁₀. [1] Mini-tablets containing uC₁₁ (60% w/w) and FD4 instilled into rat jejunum and colon showed higher FD4 absorption (AUC) than C₁₀ mini-tablets, particularly in the colon, with no histological evidence of mucosal damage. [1] |
| Cell Assay |
Caco-2 monolayer flux assay: Caco-2 cells were grown on Transwell filters for 21–28 days. The apical compartment contained Ca²⁺-free HBSS with test compounds and flux markers ([¹⁴C]-mannitol or FITC-dextrans). Samples were taken from the basolateral side over time, and Papp was calculated. Treatments were done in duplicate and repeated three times. [1] MTT cytotoxicity assay: Caco-2 cells were seeded in 96-well plates, exposed to MCFA salts in Ca²⁺-free DMEM for 1, 8, or 24 h, followed by MTT incubation and absorbance measurement. IC₅₀ was defined as the concentration reducing absorbance by 50%. [1] High-content analysis (HCA): Caco-2 cells were seeded in 96-well plates, exposed to MCFA salts, then stained with Hoechst 33342, Fluo-4 AM, TMRM, and TOTO®-3 iodide. Imaging was performed using a high-content analyzer to measure cell number, nuclear area, nuclear intensity, intracellular Ca²⁺, mitochondrial membrane potential, and plasma membrane permeability. [1] |
| Animal Protocol |
In situ intestinal instillation in rats: Male Wistar rats were fasted, anesthetized, and jejunal or colonic loops were surgically prepared. Solutions containing uC₁₁ or C₁₀ (100 mM) with FD4 (40 mg/kg) were injected into the loop. Blood samples were taken over 180 min for FD4 quantification. [1] Mini-tablet instillation: Mini-tablets containing uC₁₁ or C₁₀ (60% w/w) and FD4 were placed into jejunal or colonic loops via a small incision. PBS was added to aid disintegration. Blood was collected over 180 min for pharmacokinetic analysis. [1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Undecylenic acid may be absorbed through the skin [MSDS]. No information regarding route of elimination. No information regarding volume of distribution. No information regarding clearance. Metabolism / Metabolites No information regarding metabolism. Biological Half-Life No information regarding half-life. The oral bioavailability of FD4 was enhanced by co-administration with uC₁₁ in rat intestinal instillations. In jejunum, BA increased from 2.1% (control) to 21.8% (uC₁₁); in colon, from 1.7% to 49.8%. [1] uC₁₁ mini-tablets showed prolonged Tmax (84 min in colon) compared to C₁₀ tablets (45 min), likely due to slower dissolution. [1] |
| Toxicity/Toxicokinetics |
Protein Binding No information regarding protein binding. uC₁₁ showed lower cytotoxicity than the saturated C₁₁ in Caco-2 cells (MTT and HCA). [1] Histological examination of rat intestinal mucosa after 3-h instillation with 100 mM uC₁₁ showed no significant damage, similar to C₁₀. [1] The oral LD₅₀ of undecylenic acid in rats is 2.5 g/kg (cited from Material Safety Data Sheet). [1] |
| References |
[1]. Efficacious intestinal permeation enhancement induced by the sodium salt of 10-undecylenic acid, a medium chain fatty acid derivative. AAPS J. 2014 Sep;16(5):1064-76. |
| Additional Infomation |
10-undecenoic acid is an undecenoic acid having its double bond in the 10-position. It is derived from castor oil and is used for the treatment of skin problems. It has a role as a plant metabolite and an antifungal drug. It is a conjugate acid of a 10-undecenoate. Undecylenate, or undecylenic acid, is an unsaturated fatty acid with a terminal double bond that is derived from castor oil. Undecylenic acid is also found naturally in the human sweat. It is used as a precursor in the manufacture of aromatic chemicals, polymers or modified silicones. Undecylenic acid was first isolated from the products of distillation of castor oil in 1877 via pyrolysis of ricinoleic acid, and has been polymerized for vinyl production. It it suggested that many organic fatty acids exert fungicidal or fungistatic actions. Undecylenic acid also possesses antifungal properties, but is never used on its own for antifungal purposes. Salts of undecylenate are found in topical over-the-counter or mixture products as antifungal agents. Zinc undecylenate is an example of a topical antifungal agent that treats skin infections such as athlete’s foot and relieves itching, burning, and irritation associated with the skin condition. Due to its bifunctional properties, undecylenate is also used as a linking molecule to conjugate other biomolecules such as proteins. It serves as an acid moiety for anabolic steroid boldenone. Undecylenic acid has been reported in Streptomyces and Salvia fruticosa with data available. Undecylenic Acid is a natural or synthetic fungistatic fatty acid, antifungal Undecylenic Acid is used topically as a zinc salt in various creams against fungal infections, eczemas, ringworm, and other cutaneous conditions. The zinc provides an astringent action, reducing rawness and irritation. Undecylenic acid is found in black elderberry. Undecylenic acid is a flavouring ingredient. See also: Triclosan (active moiety of); Zinc Undecylenate (has salt form); Calcium Undecylenate (has salt form) ... View More ... Drug Indication Indicated for the treatment of fungal infections as a salt form. No therapeutic indications on its own. Mechanism of Action Undecylenic acid demonstrated effectiveness against _Candida albicans_, which is an opportunistic pathogenic yeast with two cellular morphologies: the round yeast form and the filamentous form with elongated hyphae. Hyphae formation is associated with active infections and virulence. A study proposed that undecylenic acid inhibits biofilm formation of _Candida albicans_ with optimal concentration above 3 mM and disrupts hyphal growth, which is the morphological transition from yeast to filamentous phase, at concentration above 4 mM. Under the drug treatment, hyphal formation related genes, like HWP1, were significantly reduced in transcriptional level leading to poor biofilm formation. Both biofilm and hyphae formation are critical virulence factors for the initiation of skin infection and late development of disseminated infection. Undecylenic acid may also inhibit enzyme involved in lipid metabolism and abolish germ tube formation by carrying protons across the plasma membrane, thus altering cytoplasmic pH. Undecylenic acid (UA) is an OTC antifungal and nutritional supplement with over 60 years of oral use in humans. Its sodium salt (uC₁₁) is a solid suitable for tablet formulation. [1] uC₁₁ acts as a permeation enhancer by increasing paracellular transport via mild surfactant-like effects on epithelial membranes, possibly involving intracellular Ca²⁺ modulation and tight junction regulation. [1] The unsaturated bond in uC₁₁ reduces lipophilicity and cytotoxicity compared to saturated MCFAs, while maintaining permeation enhancement efficacy. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 50 mg/mL (~271.33 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.4265 mL | 27.1326 mL | 54.2652 mL | |
| 5 mM | 1.0853 mL | 5.4265 mL | 10.8530 mL | |
| 10 mM | 0.5427 mL | 2.7133 mL | 5.4265 mL |