UPGL00004 (UPGL-00004) is a newly developed glutaminase C (GAC, kidney glutaminase isoform C) inhibitor that is strong, allosteric, and has anticancer properties. With an IC50 of 29 nM, UPGL00004 exhibits high selectivity for GAC over GLS2. It also inhibits GAC. With low nanomolar potency, UPGL00004 can inhibit both the enzymatic activity of GAC and the proliferation of highly aggressive triple negative MDA-MB-231 breast cancer cells. When UPGL00004 and its analogs are contrasted with the top GAC inhibitors, BPTES and CB-839, they exhibit better microsomal stability, ligand efficiency, and fewer rotatable bonds. Heteroatom substituted heterocycloalkanes were used to replace the flexible n-diethylthio or n-butyl moiety found in the leading inhibitors in order to improve their properties. Cancer cells have altered glycolytic flux (the "Warburg effect"), which makes them dependent on increased glutamine metabolism (also known as "glutamine addiction") for growth. Overexpression of glutaminase C (GAC), which catalyzes the initial step in glutamine metabolism and thus represents a potential therapeutic target, satisfies this requirement.
Physicochemical Properties
| Molecular Formula | C25H26N8O2S2 | |
| Molecular Weight | 534.656341075897 | |
| Exact Mass | 534.16 | |
| Elemental Analysis | C, 56.16; H, 4.90; N, 20.96; O, 5.98; S, 11.99 | |
| CAS # | 1890169-95-5 | |
| Related CAS # | 1890169-95-5; | |
| PubChem CID | 121256411 | |
| Appearance | Off-white to gray solid powder | |
| LogP | 4 | |
| Hydrogen Bond Donor Count | 3 | |
| Hydrogen Bond Acceptor Count | 10 | |
| Rotatable Bond Count | 9 | |
| Heavy Atom Count | 37 | |
| Complexity | 744 | |
| Defined Atom Stereocenter Count | 0 | |
| InChi Key | MRYCNTHLPRENBA-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C25H26N8O2S2/c34-20(15-17-7-3-1-4-8-17)27-23-30-29-22(36-23)26-19-11-13-33(14-12-19)25-32-31-24(37-25)28-21(35)16-18-9-5-2-6-10-18/h1-10,19H,11-16H2,(H,26,29)(H,27,30,34)(H,28,31,35) | |
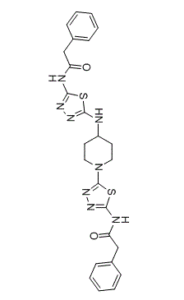
| Chemical Name | 2-phenyl-N-[5-[[1-[5-[(2-phenylacetyl)amino]-1,3,4-thiadiazol-2-yl]piperidin-4-yl]amino]-1,3,4-thiadiazol-2-yl]acetamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.03.00 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Glutaminase C (IC50 = 29 nM); Glutaminase C (Kd = 27 nM) UPGL00004 inhibits TSE, HS578T, and MDA-MB-231 cells with IC50 values of 262 nM, 129 nM, and 70 nM, respectively[1]. |
| ln Vitro |
UPGL00004 inhibits TSE, HS578T, and MDA-MB-231 cells with IC50 values of 262 nM, 129 nM, and 70 nM, respectively[1]. UPGL00004 inhibited the catalytic activity of recombinant human GAC with an IC₅₀ of 29 nM in an enzymatic assay stimulated by inorganic phosphate. [1] UPGL00004 exhibited selectivity for GAC over GLS2, with IC₅₀ values against GLS2 ranging from 10 to 35 μM. [1] In a fluorescence spectroscopic binding assay using a GAC(F327W) mutant, UPGL00004 showed dose-dependent quenching of intrinsic tryptophan fluorescence, indicating direct binding to GAC, with a binding affinity similar to that of CB-839 and higher than BPTES. [1] UPGL00004 inhibited the proliferation of triple-negative breast cancer cell lines: MDA-MB-231 (IC₅₀ = 70 nM), HS578T (IC₅₀ = 129 nM), and TSE (IC₅₀ = 262 nM). It showed minimal inhibition (>1000 nM IC₅₀) against HER2-positive (MDA-MB-453) and estrogen receptor-positive (BT474) breast cancer cell lines. [1] Treatment of triple-negative breast cancer cells (MDA-MB-231, HS578T, TSE) with 70 nM UPGL00004 for 14 hours significantly reduced ammonia production (a product of GAC-catalyzed glutamine hydrolysis), indicating inhibition of intracellular GAC activity. This effect was less pronounced in HER2-positive MDA-MB-453 cells. [1] The inhibitory potency of UPGL00004 against MDA-MB-231 cell growth increased approximately 2-fold under glutamine-deprived conditions. [1] X-ray crystallography revealed that UPGL00004 binds to the same allosteric site as BPTES and CB-839 at the dimer-dimer interface of the GAC tetramer, stabilizing an inactive conformation. The structure showed that UPGL00004 forms 9 high-strength hydrogen bonds with GAC (according to Berndt criteria), contributing to its high binding affinity. [1] |
| ln Vivo |
The combination of UPGL00004 (1 mg/kg body weight) and Bevacizumab (2.5 mg/kg body weight) through intraperitoneal injection totally stop any discernible growth in tumor size in a tumor graft model derived from triple-negative breast cancer patients[1]. In a patient-derived xenograft model of triple-negative breast cancer (HCI-002 graft), intraperitoneal administration of UPGL00004 (1 mg/kg) every other day for 4 weeks, in combination with the anti-VEGF antibody bevacizumab (2.5 mg/kg), completely prevented any detectable increase in tumor size. Neither agent alone at these doses caused a statistically significant reduction in tumor growth. [1] |
| Enzyme Assay |
Recombinant human GAC (residues 73-598) or GLS2 (residues 38-602) was expressed in E. coli and purified using nickel affinity chromatography followed by size-exclusion chromatography. [1] For the enzymatic activity assay, recombinant GAC (50 nM) or GLS2 (100 nM) was combined with 20 mM glutamine and various concentrations of the inhibitor (final DMSO concentration 2%) in 65 mM Tris acetate and 0.25 mM EDTA (pH 8.6) buffer. The reaction was immediately initiated by adding 150 mM potassium phosphate and incubated at room temperature for 10 minutes, then quenched with hydrochloric acid. [1] In a second step, the quenched reaction mixture was combined with a solution containing β-NAD (1.79 mM), glutamate dehydrogenase (GDH, 7.5 units/mL), and hydrazine monohydrate (2%) in 130 mM Tris-HCl (pH 9.4) buffer. The combined mixture was incubated at room temperature for 1 hour, and the absorbance at 340 nm was measured to determine glutamate production. IC₅₀ values were calculated using a four-parameter logistic function. [1] |
| Cell Assay |
Breast cancer cells are kept at 37°C and in an atmosphere with 5% CO2 in complete media (RPMI 1640 supplemented with 10% FBS). For assays, a density of 1 × 10⁴ cells per well is applied to the wells of a 12-well plate. After allowing the cells to adhere to the plate for one night, they are treated for six days in full media with the prescribed dosages of each medication or a DMSO control. Every two days, drugs and media are updated. Trypsin is used to extract the cells from the plates on the sixth day, and a hemocytometer is used to manually count the cells. Breast cancer cells were maintained in RPMI 1640 medium supplemented with 10% fetal bovine serum. For proliferation assays, cells were seeded in 12-well plates at a density of 1×10⁴ cells per well, allowed to attach overnight, and then treated with various concentrations of UPGL00004 or DMSO control in complete medium for 6 days. Media and drugs were refreshed every 2 days. On day 6, cells were trypsinized and counted manually with a hemocytometer. [1] For ammonia production assays, cells were seeded in 6-well plates at 2×10⁵ cells per well and grown for 48 hours. The medium was then replaced with serum-free, phenol red-free, glutamine-free RPMI 1640 supplemented with 2 mM glutamine and the indicated inhibitor or DMSO. After 14 hours of incubation, the conditioned medium was collected, and ammonia content was measured using a commercial ammonia assay kit according to the manufacturer's instructions. [1] |
| Animal Protocol |
triple-negative breast cancer patient-derived tumor graft (PDX) model (NOD/SCID mice) 1 mg/kg i.p. A patient-derived triple-negative breast cancer tumor graft (HCI-002) was cut into pieces and implanted into the mammary glands of NOD/SCID mice. [1] When tumors reached approximately 3 mm in diameter (about 2 weeks post-implantation), mice were randomly divided into groups. [1] UPGL00004 was dissolved in a vehicle consisting of RPMI medium containing 4% DMSO, 5% Cremophor EL, and 5% ethanol (v/v). [1] Mice received intraperitoneal injections every other day for 4 weeks with: vehicle control; bevacizumab (2.5 mg/kg); UPGL00004 (1 mg/kg); or a combination of bevacizumab and UPGL00004. [1] Tumor volumes were monitored, and after 4 weeks, tumors were excised and weighed. [1] |
| ADME/Pharmacokinetics |
The study mentions that selected compounds from the series containing UPGL00004 showed superior metabolic stability compared to BPTES and CB-839 when incubated with human liver microsomes. [1] |
| References |
[1]. Characterization of the interactions of potent allosteric inhibitors with glutaminase C, a key enzyme in cancer cell glutamine metabolism. J Biol Chem. 2018 Mar 9;293(10):3535-3545. |
| Additional Infomation |
UPGL00004 is a novel allosteric inhibitor of GAC, designed as a BPTES analog where the flexible chain moiety is replaced by a rigid saturated heterocyclic core. [1] The compound is reported to have improved microsomal stability compared to BPTES and CB-839. [1] Its mechanism of action involves binding to and stabilizing an inactive tetrameric state of GAC at an allosteric site distant from the catalytic center, thereby inhibiting glutamine metabolism. [1] The study suggests that combining GAC inhibition (UPGL00004) with anti-angiogenic therapy (bevacizumab) may be a beneficial therapeutic strategy for triple-negative breast cancer. [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8703 mL | 9.3517 mL | 18.7035 mL | |
| 5 mM | 0.3741 mL | 1.8703 mL | 3.7407 mL | |
| 10 mM | 0.1870 mL | 0.9352 mL | 1.8703 mL |