Pibrentasvir (formerly ABT-530; ABT530, A-1325912; A1325912; trade name: Mavyret; Maviret), an HCV NS5A protease inhibitor, is an approved antiviral (anti-HCV) drug developed by Abbvie for treating HCV infections. It can be used to treat hepatitis C in the US and EU in combination with glecaprevir as glecaprevir/pibrentasvir (Mavyret).
Physicochemical Properties
| Molecular Formula | C57H65F5N10O8 |
| Molecular Weight | 1113.2 |
| Exact Mass | 1112.49 |
| Elemental Analysis | C, 61.50; H, 5.89; F, 8.53; N, 12.58; O, 11.50 |
| CAS # | 1353900-92-1 |
| Related CAS # | 1821461-48-6 (butanamine hydrate);1353900-92-1 (free); |
| PubChem CID | 58031952 |
| Appearance | White to off-white solid powder |
| Density | 1.4±0.1 g/cm3 |
| Index of Refraction | 1.614 |
| LogP | 8.71 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 17 |
| Rotatable Bond Count | 17 |
| Heavy Atom Count | 80 |
| Complexity | 2000 |
| Defined Atom Stereocenter Count | 8 |
| SMILES | FC1C=C2C(=CC=1[C@H]1CCC(C3=C(C=C4C(=C3)NC([C@@H]3CCCN3C([C@H]([C@@H](C)OC)NC(=O)OC)=O)=N4)F)N1C1C=C(C(=C(C=1)F)N1CCC(C3C=CC(=CC=3)F)CC1)F)NC([C@@H]1CCCN1C([C@H]([C@@H](C)OC)NC(=O)OC)=O)=N2 |
| InChi Key | VJYSBPDEJWLKKJ-NLIMODCCSA-N |
| InChi Code | InChI=1S/C57H65F5N10O8/c1-29(77-3)49(67-56(75)79-5)54(73)70-19-7-9-47(70)52-63-41-25-35(37(59)27-43(41)65-52)45-15-16-46(72(45)34-23-39(61)51(40(62)24-34)69-21-17-32(18-22-69)31-11-13-33(58)14-12-31)36-26-42-44(28-38(36)60)66-53(64-42)48-10-8-20-71(48)55(74)50(30(2)78-4)68-57(76)80-6/h11-14,23-30,32,45-50H,7-10,15-22H2,1-6H3,(H,63,65)(H,64,66)(H,67,75)(H,68,76)/t29-,30-,45-,46-,47+,48+,49+,50+/m1/s1 |
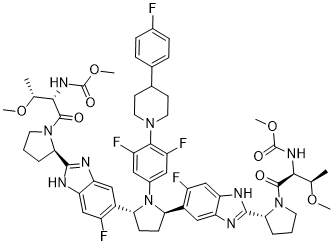
| Chemical Name | methyl N-[(2S,3R)-1-[(2S)-2-[6-[(2R,5R)-1-[3,5-difluoro-4-[4-(4-fluorophenyl)piperidin-1-yl]phenyl]-5-[6-fluoro-2-[(2S)-1-[(2S,3R)-3-methoxy-2-(methoxycarbonylamino)butanoyl]pyrrolidin-2-yl]-3H-benzimidazol-5-yl]pyrrolidin-2-yl]-5-fluoro-1H-benzimidazol-2-yl]pyrrolidin-1-yl]-3-methoxy-1-oxobutan-2-yl]carbamate |
| Synonyms | ABT-530; ABT 530; ABT530; A-1325912; A1325912; A 1325912; A 1325912.0; A1325912.0; A-1325912.0; Pibrentasvir |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
GT5a (EC50 = 1.4 pM); GT1a H77 (EC50 = 1.8 pM); GT2b (EC50 = 1.9 pM); GT4a (EC50 = 1.9 pM); GT3a (EC50 = 2.1 pM) Pibrentasvir is a potent pan-genotypic next-generation HCV NS5A inhibitor that retains activity against common amino acid substitutions of HCV genotypes 1–6, which are known to confer resistance to NS5A inhibitors that are currently approved.[1] |
| ln Vitro |
Pibrentasvir is a potent pan-genotypic next-generation HCV NS5A inhibitor that retains activity against common amino acid substitutions of HCV genotypes 1–6, which are known to confer resistance to NS5A inhibitors that are currently approved.[1] Pibrentasvir demonstrated potent, pan-genotypic antiviral activity against a panel of stable HCV subgenomic replicons containing NS5A from genotypes 1 to 6. The 50% effective concentration (EC50) values in 0% human plasma ranged from 1.4 to 5.0 pM: 1.8 pM against genotype 1a-H77, 4.3 pM against genotype 1b-Con1, 5.0 pM against genotype 2a-JFH-1, and between 1.4 and 2.8 pM against chimeric replicons containing NS5A from genotypes 2a, 2b, 3a, 4a, 5a, and 6a. The antiviral activity ofpibrentasvirwas attenuated 35- to 47-fold in the presence of 40% human plasma due to plasma protein binding, with EC50s increasing to 64 pM (genotype 1a) and 200 pM (genotype 1b). Pibrentasvir showed similar activity (median EC50s ranging from 0.50 to 2.7 pM) against a panel of 64 replicons containing NS5A genes derived from HCV-infected patient samples across genotypes 1a, 1b, 2a, 2b, 3a, 4a, 5a, and 6a/e/p, including samples with baseline polymorphisms at positions associated with resistance to other NS5A inhibitors. Pibrentasvirmaintained full activity against a wide range of single-position NS5A amino acid substitutions known to confer resistance to other NS5A inhibitors (e.g., daclatasvir, elbasvir, ledipasvir, ombitasvir, velpatasvir) in genotypes 1 to 6, with EC50 fold changes typically ≤7. For example, against genotype 1a single substitutions: M28T (2.1-fold), Q30E (2.4-fold), L31M (1.1-fold), Y93H (6.7-fold), Y93N (7.1-fold). Against genotype 3a Y93H, it showed only a 2.3-fold increase in EC50. It also retained activity against replicons containing key resistance-associated substitutions for NS3/4A protease inhibitors (e.g., R155K, D168V) and NS5B polymerase inhibitors (e.g., S282T, C316Y), with EC50 fold changes ≤1.7, indicating no cross-resistance. Pibrentasvir had no measurable antiviral activity against human immunodeficiency virus type 1 (HIV-1 EC50 >900,000 pM) or hepatitis B virus (HBV EC50 >32,000,000 pM). Combination studies in genotype 1b replicon cells using checkerboard assays and MacSynergy II analysis showed that pibrentasvirproduced minor to moderate synergistic antiviral activity when combined with interferon-alpha (synergy volume 43 ± 4.7 μM²%), ribavirin (synergy volume 29 ± 7.4 μM²%), or the HCV NS3/4A protease inhibitor glecaprevir (synergy volume 73 ± 17 μM²%). No antagonism was observed. [1] |
| ln Vivo |
The study mentions that the potent in vitro activity of pibrentasvir translated to robust antiviral activity in a 3-day monotherapy clinical trial. In genotype 1-infected, treatment-naive adults, daily doses of 40 to 400 mg resulted in a mean maximal decrease in HCV plasma RNA from baseline of ≥4.1 log₁₀ IU/ml at the end of the 3-day period. The 120 mg dose, selected for phase 3 studies, achieved a 4.5 log₁₀ IU/ml decrease. During this monotherapy study, NS5A resistance-associated substitutions emerged in only 3 out of 40 (7.5%) patients. [1] |
| Cell Assay |
Southern Research Institute conducts assays for antiviral activity against HBV and HIV-1. Pibrentasvir is evaluated in an HIV-1 antiviral cytoprotection assay with the IIIB strain of the virus and CEM-SS cells. In short, the virus and cells are combined and incubated for six days in the presence of pibrentasvir or zidovudine (AZT; positive control). The virus titers are predetermined so that the control wells infected with the virus showed between 85% and 95% of the cells' viability being lost as a result of virus replication. Consequently, the observation of an antiviral effect or cytoprotection occurs when a compound hinders the replication of viruses. Twenty to twenty-five microliters of MTS reagent are added to each well six days after infection, and the microtiter plates are then incubated for four to six hours in order to measure the viability of the cells. Plates are read using a Molecular Devices Vmax or SpectraMax Plus plate reader spectrophotometrically at 490/650 nm. Antiviral Activity in Stable HCV Replicon Cells: Stable HCV subgenomic replicon cell lines (e.g., containing NS5A from genotypes 1a, 1b, 2a, or chimeric replicons) were cultured. The cells were incubated with serial dilutions of pibrentasvir for 3 days in medium containing 5% fetal bovine serum, with or without 40% human plasma. HCV replication inhibition was determined by measuring luciferase reporter activity from cell lysates using a luminometer. The EC50 was calculated using nonlinear regression curve fitting. Cytotoxicity Assay (MTT): Cytotoxicity was assessed in Huh-7 cells (with genotype 1a replicon), HepG2 cells, and MT4 cells. Cells were plated in 96-well plates and incubated with pibrentasvir. After an incubation period, cell viability was measured using an MTT colorimetric assay. The 50% cytotoxic concentration (CC50) was calculated. The CC50 for pibrentasvir was >32,000,000 pM in Huh-7 cells and >10,000,000 pM in HepG2 and MT4 cells, indicating a high therapeutic index (>10⁷-fold). Antiviral Activity Against HIV-1 and HBV: For HIV-1, an antiviral cytoprotection assay was used. CEM-SS cells were infected with HIV-1 (IIIB strain) in the presence of pibrentasvir and incubated for 6 days. Cell viability was assessed using an MTS reagent, with protection indicating antiviral effect. For HBV, HepG2 2.2.15 cells were treated with pibrentasvir for 6 days. Extracellular HBV DNA in the culture supernatant was quantified by real-time quantitative PCR (TaqMan) after protease treatment. The EC50 was calculated from the reduction in HBV DNA levels. Resistance Selection Assay: HCV stable replicon cells containing NS5A from specific genotypes were plated and grown in the presence of G418 and pibrentasvir at concentrations 10- or 100-fold above the respective EC50. The medium was refreshed every 3-4 days for approximately 3 weeks. Surviving colonies were picked, expanded, and their NS5A coding region was amplified by RT-PCR and sequenced to identify resistance-associated amino acid substitutions. The colony survival rate was calculated. Transient Replicon Assay for Mutant Susceptibility: HCV replicons engineered with specific amino acid substitutions in NS5A, NS3, or NS5B were constructed. Plasmids were linearized, transcribed into RNA, and transfected into Huh-7 cells. Inhibition of replicon replication by pibrentasvir was measured using a luciferase assay. Replication efficiency was calculated as a percentage of wild-type replication. Combination Study (Checkerboard Assay): In genotype 1b-Con1 replicon cells, pibrentasvir was combined with another HCV inhibitor (e.g., IFN-α, ribavirin, glecaprevir) in a checkerboard format using serial 2-fold dilutions. HCV replication was determined by a luciferase reporter assay. The data were analyzed using the MacSynergy II program to calculate synergy/antagonism volumes and determine the interaction type (e.g., minor synergy, moderate synergy). [1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion In healthy subjects, the time it takes to reach the peak plasma concentration (Tmax) is approximately 5 hours. The mean peak plasma concentration (Cmax) is 110ng/mL in non-cirrhotic HCV-infected subjects. Relative to fasting conditions, the consumption of meals increases the absorption of pibrentasvir by 40-53%. The predominant route of elimination of the drug is biliary-fecal, where 96.6% of administered drug is excreted in feces and 0% of the drug is excreted in the urine. Metabolism / Metabolites Pibrentasvir is not metabolized. Biological Half-Life The elimination half life (t1/2) is approximately 13 hours. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Pibrentasvir has not been studied in nursing mothers being treated for hepatitis C infection. Because it is >99.9% bound to maternal plasma proteins, amounts in breastmilk are likely to be very low. Hepatitis C is not transmitted through breastmilk and breastmilk has been shown to inactivate hepatitis C virus (HCV). However, the Centers for Disease Control recommends that mothers with HCV infection should consider abstaining from breastfeeding if their nipples are cracked or bleeding. It is not clear if this warning would apply to mothers who are being treated for hepatitis C. Infants born to mothers with HCV infection should be tested for HCV infection; because maternal antibody is present for the first 18 months of life and before the infant mounts an immunologic response, nucleic acid testing is recommended. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Protein Binding Pibrentasvir is >99.9% bound to human plasma proteins. The Blood-to-plasma ratio is approximately 0.62. The study provides in vitro cytotoxicity data (CC50), as described in the Cell Assay section, indicating low cellular toxicity. [1] |
| References |
[1]. In Vitro Antiviral Activity and Resistance Profile of the Next-Generation Hepatitis C Virus NS5A Inhibitor Pibrentasvir. Antimicrob Agents Chemother. 2017 Apr 24;61(5). pii: e02558-16. |
| Additional Infomation |
Pibrentasvir is a direct acting antiviral agent and Hepatitis C virus (HCV) NS5A inhibitor that targets the the viral RNA replication and viron assembly. In combination with [DB13879], pibrentastiv is a useful therapy for patients who experienced therapeutic failure from other NS5A inhibitors. In cell cultures, the emergence of amino acid substitutions at known NS5A inhibitor resistance-associated positions in HCV genotype 1a, 2a or 3a replicons led to reduced susceptibility and resistance to pibrentasvir. These resistance-associated amino acid substitutions included Q30D/deletion, Y93D/H/N or H58D +Y93H in genotype 1a replicons, F28S + M31I or P29S + K30G in genotype 2a replicons, and Y93H in genotype 3a replicons. Individual NS5A amino acid substitutions that reduced susceptibility to pibrentasvir include M28G or Q30D in a genotype 1a replicon and P32-deletion in a genotype 1b replicon. Pibrentasvir is available as an oral combination therapy with [DB13879] under the brand name Mavyret. This fixed-dose combination therapy was FDA-approved in August 2017 to treat adults with chronic hepatitis C virus (HCV) genotypes 1-6 without cirrhosis (liver disease) or with mild cirrhosis, including patients with moderate to severe kidney disease and those who are on dialysis. Mavyret is also indicated for HCV genotype 1-infected patients who have been previously treated with regimens either containing an NS5A inhibitor or an NS3/4A protease inhibitor, but not both. Hepatitis C viral infection often leads to decreased liver function and subsequent liver failure, causing a significantly negative impact on the patients' quality of life. The ultimate goal of the combination treatment is to achieve sustained virologic response (SVR) and cure the patients from the infection. In clinical trials, this combination therapy achieved SVR12 rate, or undetectable Hepatitis C for twelve or more weeks after the end of treatment, of ≥93% across genotypes 1a, 2a, 3a, 4, 5 and 6. Pibrentasvir is a Hepatitis C Virus NS5A Inhibitor. The mechanism of action of pibrentasvir is as a P-Glycoprotein Inhibitor, and Breast Cancer Resistance Protein Inhibitor, and Organic Anion Transporting Polypeptide 1B1 Inhibitor, and Organic Anion Transporting Polypeptide 1B3 Inhibitor, and Cytochrome P450 3A Inhibitor, and Cytochrome P450 1A2 Inhibitor, and UGT1A1 Inhibitor. Drug Indication Indicated for the treatment of adult patients with chronic hepatitis C virus (HCV) genotype 1, 2, 3, 4, 5 or 6 infection without cirrhosis or with compensated cirrhosis (Child-Pugh A). MAVYRET is also indicated for the treatment of adult patients with HCV genotype 1 infection, who previously have been treated with a regimen containing an HCV NS5A inhibitor or an NS3/4A protease inhibitor (PI), but not both. FDA Label Mechanism of Action NS5A is a phosphoprotein that plays an essential role in replication, assembly and maturation of infectious viral proteins. The basal phosphorylated form of NS5A, which is maintained by C-terminal serine cluster, is key in ensuring its interaction with the viral capsid protein, or the core protein. By blocking this interaction, pibrentasvir inhibits the assembly of proteins and production of mature HCV particles. NS5A also interacts with viral and cellular proteins to form the HCV replicase complex, and supports the RNA replication of HCV. Pharmacodynamics Pibrentasvir is a pan-genotypic . According to HCV replicon assays, pibrentasvir has EC50 values ranging from 0.08-4.6 nM agaisnt laboratory and clinical isolates from subtypes 1a, 1b, 2a, 2b, 3a, 4a, 4d, 5a, and 6a, or EC50 values of 0.5-4.3 pM against laboratory and clinical isolates from subtypes 1a, 1b, 2a, 2b, 3a, 4a, 4b, 4d, 5a, 6a, 6e and 6p. It is active against common resistance-conferring substitutions in HCV genotypes 1 to 6 that confers resistance and decreased therapeutic response from other NS5A inhibitors, inluding positions 24, 28, 30, 31, 58, 92, or 93 in NS5A. In a QT study, pibrentasvir is not shown to prolong the QTc interval. Pibrentasvir is a novel, next-generation HCV NS5A inhibitor with a chemical name provided in the Materials and Methods. Its mechanism of action is inhibition of the HCV NS5A protein, which plays multiple critical roles in viral RNA replication and virion assembly. The study highlights pibrentasvir's high genetic barrier to resistance, as evidenced by the low frequency of colony selection in vitro and the low rate of emergent resistance in a short-term monotherapy clinical trial. Pibrentasvir has been co-administered with glecaprevir (an HCV NS3/4A protease inhibitor) in clinical studies. This combination achieved high sustained virologic response rates in treatment-naive non-cirrhotic patients infected with HCV genotypes 1-6, and in genotype 1-infected patients who had failed a prior direct-acting antiviral regimen. [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.8983 mL | 4.4916 mL | 8.9831 mL | |
| 5 mM | 0.1797 mL | 0.8983 mL | 1.7966 mL | |
| 10 mM | 0.0898 mL | 0.4492 mL | 0.8983 mL |