Physicochemical Properties
| Molecular Formula | C15H15N3O6 |
| Molecular Weight | 333.296103715897 |
| Exact Mass | 333.096 |
| Elemental Analysis | C, 54.05; H, 4.54; N, 12.61; O, 28.80 |
| CAS # | 745055-86-1 |
| PubChem CID | 11674376 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 603.0±65.0 °C at 760 mmHg |
| Flash Point | 318.5±34.3 °C |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.659 |
| LogP | 1.58 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 24 |
| Complexity | 576 |
| Defined Atom Stereocenter Count | 0 |
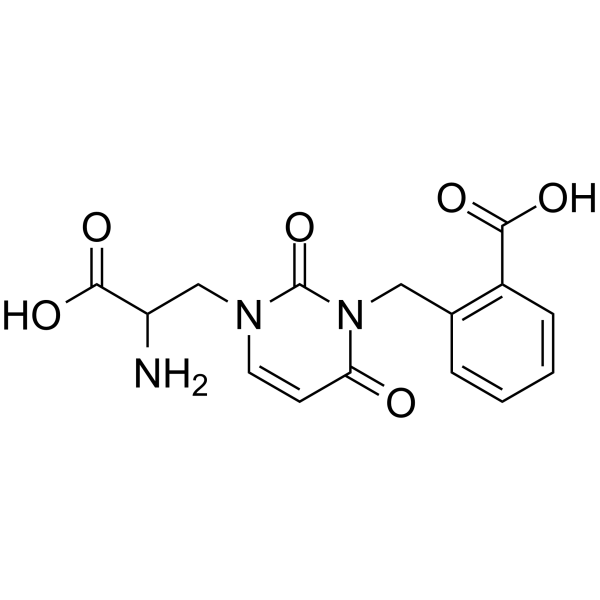
| SMILES | O=C1N(C(C=CN1CC(C(=O)O)N)=O)CC1C=CC=CC=1C(=O)O |
| InChi Key | UUIYULWYHDSXHL-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C15H15N3O6/c16-11(14(22)23)8-17-6-5-12(19)18(15(17)24)7-9-3-1-2-4-10(9)13(20)21/h1-6,11H,7-8,16H2,(H,20,21)(H,22,23) |
| Chemical Name | 2-[[3-(2-amino-2-carboxyethyl)-2,6-dioxopyrimidin-1-yl]methyl]benzoic acid |
| Synonyms | UBP 296; 745055-86-1; (RS)-1-(2-amino-2-carboxyethyl)-3-(2-carboxybenzyl)pyrimidine-2,4-dione; CHEMBL372631; UBP296; 2-[[3-(2-amino-2-carboxyethyl)-2,6-dioxopyrimidin-1-yl]methyl]benzoic acid; 2-[[3-(2-amino-2-carboxyethyl)-2,6-dioxo-1-pyrimidinyl]methyl]benzoic acid; 2-((3-(2-Amino-2-carboxyethyl)-2,6-dioxo-2,3-dihydropyrimidin-1(6H)-yl)methyl)benzoic acid; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | GLUK5-containing kainate receptor |
| ln Vitro |
Willardiine derivatives with an N3-benzyl substituent bearing an acidic group have been synthesized with the aim of producing selective antagonists for GLUK5-containing kainate receptors. UBP296 was found to be a potent and selective antagonist of native GLUK5-containing kainate receptors in the spinal cord, with activity residing in the S enantiomer (UBP302). In cells expressing human kainate receptor subunits, UBP296 selectively depressed glutamate-induced calcium influx in cells containing GLUK5 in homomeric or heteromeric forms. In radioligand displacement binding studies, the willardiine analogues displaced [3H]kainate binding with IC50 values >100 microM at rat GLUK6, GLUK2 or GLUK6/GLUK2. An explanation of the GLUK5 selectivity of UBP296 was obtained using homology models of the antagonist bound forms of GLUK5 and GLUK6 [1].
Characterisation of UBP296 on human kainate receptors using calcium influx studies [1] UBP296 depressed glutamate-induced calcium influx in HEK293 cells stably expressing recombinant human GLUK5-containing kainate receptors. Glutamate induced calcium influx in cells expressing GLUK5, GLUK5/GLUK6 and GLUK5/GLUK2 with EC50 values of 22±5, 27±9 and 35±41 μM, respectively, and this was inhibited by UBP296 with IC50 values of 3.5±1.5, 4.0±0.7 and 7.0±5.1 μM, respectively (n=3, mean±s.e.m.). Using the Cheng–Prusoff equation Kb values of 0.6±0.1, 0.8±0.1 and 1.0±0.4 μM (n=3, mean±s.e.m.) were calculated for the antagonism by UBP296 of glutamate-induced calcium influx in cells expressing GLUK5, GLUK5/GLUK6 and GLUK5/GLUK2, respectively (Fig. 4A). UBP296, up to a concentration of 300 μM, had little effect on calcium influx in cells expressing GLUK6 and GLUK6/GLUK2 (Fig. 4A). Electrophysiological characterisation of UBP296 on human kainate receptors [1] In HEK293 cells expressing human GLUK5, UBP296 depressed steady-state ion currents induced by 100 μM glutamate. Concentrations of 1, 3 and 10 μM UBP296 inhibited the glutamate currents by 41±5, 67±3 and 83±3%, respectively (Fig. 4B; n=3, mean±s.e.m.). |
| ln Vivo |
In rat hippocampal slices, UBP296 reversibly blocked ATPA-induced depressions of synaptic transmission at concentrations subthreshold for affecting AMPA receptor-mediated synaptic transmission directly. UBP296 also completely blocked the induction of mossy fibre LTP, in medium containing 2 mM (but not 4 mM) Ca2+. These data provide further evidence for a role for GLUK5-containing kainate receptors in mossy fibre LTP. In conclusion, UBP296 is the most potent and selective antagonist of GLUK5-containing kainate receptors so far described.
UBP296 is a selective antagonist of kainate receptors in the spinal cord [1] To investigate the selectivity of the most potent and selective kainate receptor antagonist, the ability of UBP296 to block equi-effective responses to AMPA, NMDA, DHPG (mGlu1 and mGlu5 receptor agonist) and kainate on motoneurones was measured. In the presence of UBP296 (50 μM), responses to kainate, AMPA, DHPG and NMDA were 60.3±5.2, 82.2±6.7, 110±5 and 116±12%, respectively, of those in the absence of antagonist (n=3; mean±s.e.m.). A sample trace is shown in Fig. 3. A paired Student’s t-test showed that the NMDA and DHPG values in the presence of UBP296 were not significantly different from control responses (P>0.10). It was therefore clear that UBP296 did not act as an antagonist at the NMDA or group 1 metabotropic glutamate receptors present on spinal motoneurones. |
| Cell Assay |
UBP296 was added in the absence of agonist during the first addition, and in the presence of 100 μM glutamate during the second addition. The presence of concanavalin A may modify the estimated IC50 values (Paternain et al., 1998) so that they are not definitive for physiological experiments, however, the rank order of antagonist potency would not be expected to change. In electrophysiological assays whole-cell voltage clamp recordings (Vh=−70 mV) were made from single cells using the tight seal whole-cell configuration of the patch-clamp technique (Hamill et al., 1981). Glass fragments of coverslips with adherent cells were pre-incubated for at least 10 min in media containing 250 μg/ml concanavalin A in order to block desensitization, then placed in a perfusion chamber and rinsed with buffer of composition: 138 mM NaCl, 5 mM CaCl2, 5 mM KCl, 1 mM MgCl2, 10 mM HEPES, and 10 mM glucose, pH 7.5 with NaOH (osmolality 295 mOsm/kg). Pipette solutions contained: 140 mM CsCl, 1 mM MgCl2, 14 mM diTris creatine phosphate, 50 U/ml creatine phosphokinase, 14 mM MgATP, 10 mM HEPES, and 15 mM BAPTA, pH 7.2 with CsOH (osmolality 295 mOsm/kg). Experiments were performed at room temperature (20–22 °C) and recorded on an Axopatch 200 A amplifier using pClamp 8 software. Pipette resistance was typically 1.5–2.5 MΩ. Concentration–response curves for UBP296 were analysed using GraphPad Prism 3.02 software, with slope factor fixed at 1, and top and bottom fixed at 100 and 0% inhibition, respectively. The dissociation constant (Kb) was calculated from the IC50 value for inhibiting 100 μM glutamate-induced calcium influx according to the Cheng–Prusoff equation (Cheng and Prusoff, 1973): where [Glu] is the concentration of glutamate (100 μM) and EC50 Glu is the EC50 value of glutamate for evoking calcium influx in the given cell line, determined from glutamate concentration–response curves run in the same plate as UBP296 concentration–response curves [1]. |
| Animal Protocol |
Characterisation of UBP296 across a range of glutamate receptors expressed on neonatal rat motoneurones [1] Experiments performed to investigate the effect of UBP296 on receptors expressed on motoneurones were carried out in the presence of tetrodotoxin (TTX, 10 μM for 2 min, then 0.1 μM continuously) to block action potential-dependent release. Using this protocol, no stimulation was applied and the peak amplitude of agonist-induced depolarisation of motoneurones was measured from d.c. shifts in ventral root polarity after agonist application (More et al., 2002). In experiments to determine the antagonist selectivity of UBP296, medium containing approximately equi-effective concentrations of either (S)-AMPA (0.7 μM), kainate (2 μM), NMDA (10 μM), or (S)-3,5-dihydroxyphenylglycine (DHPG) (20 μM) was applied for 1 min, in the absence and presence of UBP296 (50 μM; 30 min pre-incubation). A paired Student’s t-test was used to test for statistical significance. |
| References | [1]. Characterisation of UBP296: a novel, potent and selective kainate receptor antagonist. Neuropharmacology. 2004 Jul;47(1):46-64. |
| Additional Infomation |
2-[[3-(2-amino-2-carboxyethyl)-2,6-dioxo-1-pyrimidinyl]methyl]benzoic acid is an alpha-amino acid. A range of novel willardiine derivatives acting as selective kainate receptor antagonists has been identified. UBP296 and its purified S enantiomer UBP302 proved to be potent and highly selective GLUK5 antagonists. These compounds have advantages over previously reported antagonists such as LY377770 and LY382884, such as their greater selectivity for GLUK5 vs. AMPA receptors and improved water solubility, thus making them useful pharmacological tools for studying the physiological roles of GLUK5-containing kainate receptors. UBP296 has been used to provide further evidence that GLUK5-containing kainate receptors play a role in controlling synaptic transmission at the mossy fibre to CA3 synapse in the hippocampus and in the induction of mossy fibre LTP. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0003 mL | 15.0015 mL | 30.0030 mL | |
| 5 mM | 0.6001 mL | 3.0003 mL | 6.0006 mL | |
| 10 mM | 0.3000 mL | 1.5002 mL | 3.0003 mL |