Triheptanoin ( IND-106011; UX-007; Dojolvi) is a novel and potent fatty acid metabolic modulator and a triglyceride used for the treatment of inherited metabolic diseases, epilepsy, ventricular hypertrophy and myocardial glucose oxidation. As of 2020, it has been approved by FDA for the treatment of children and adults with molecularly confirmed long-chain fatty acid oxidation.disorders.
Physicochemical Properties
| Molecular Formula | C24H44O6 |
| Molecular Weight | 428.61 |
| Exact Mass | 428.313 |
| CAS # | 620-67-7 |
| PubChem CID | 69286 |
| Appearance | Colorless to light yellow liquid(Density:0.904 g/cm3) |
| Density | 1.0±0.1 g/cm3 |
| Boiling Point | 470.0±12.0 °C at 760 mmHg |
| Flash Point | 195.5±19.6 °C |
| Vapour Pressure | 0.0±1.2 mmHg at 25°C |
| Index of Refraction | 1.457 |
| LogP | 7.73 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 23 |
| Heavy Atom Count | 30 |
| Complexity | 421 |
| Defined Atom Stereocenter Count | 0 |
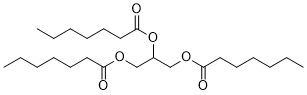
| SMILES | CCCCCCC(=O)OCC(COC(=O)CCCCCC)OC(=O)CCCCCC |
| InChi Key | PJHKBYALYHRYSK-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C24H44O6/c1-4-7-10-13-16-22(25)28-19-21(30-24(27)18-15-12-9-6-3)20-29-23(26)17-14-11-8-5-2/h21H,4-20H2,1-3H3 |
| Chemical Name | 2,3-di(heptanoyloxy)propyl heptanoate |
| Synonyms | TriheptanoinIND106011 UX-007 IND-106011IND 106011 UX007 UX 007 Glycerol trienanthate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vivo | Triheptanoin is an anaplerotic chemical that can be used to bypass enzymatic flaws that obstruct long-chain amplification oxidation (LC-FAOD) by providing latent and amplifying enzymes [1]. When using calcium supplements synergistically, triheptanoin may be utilized to treat telangiectasia. By adding extra coenzyme A and propionyl coenzyme A to their TCA, along with the trianatomy (TCA) cycle intermediate enanthate (C7), triheptanoin functions as an anaplerotic agent. Triheptanoate supports the energetic hypothesis of Alzheimer's disease by increasing brain ATP consumption and mitochondrial dysfunction, including respiration and redox balance in models of the disease [2]. In the hippocampus formation of pilot pilocarpine-induced galvanic states, rats treated with triheptanoic acid (35E%) for 10 days before to pilocarpine administration maintained mitochondrial function [3]. |
| Animal Protocol |
Animal/Disease Models: 7-8 week old male CD1 (35-40 g)[3] Doses: 35E% oral triheptanoic acid mixed into mouse feed Route of Administration: 35E% triheptanoic acid treatment was given 10 days before SE induction Experimental Results: Mitochondrial function was preserved in SE mice. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion A single 0.3 g/kg dose of triheptanoin reaches a Cmax of 178.9 µmol/L, with a Tmax 0.5 h, and an AUC of 336.5 µmol\*h/L. A single 0.4 g/kg dose of triheptanoin reaches a Cmax of 259.1 µmol/L, with a Tmax 0.8 h, and an AUC of 569.1 µmol\*h/L. Triheptanoin is minimally eliminated in the urine. A single dose of 0.3 g/kg results in a mean apparent clearance of 6.05 L/h/kg for heptanoate. A single dose of 0.4 g/kg results in a mean apparent clearance of 4.31 L/h/kg for heptanoate. Metabolism / Metabolites Triheptanoin is hydrolysed to heptanoate, which can be further metabolized to β-hydroxypentanoate or β-hydroxybutyrate. Biological Half-Life Due to multiple peak concentrations of the heptanoate metabolite, the half life of triheptanoin could not be determined. |
| Toxicity/Toxicokinetics |
Hepatotoxicity Serum enzyme elevations during triheptanoin therapy are frequent, mostly reflecting the underlying condition of muscle and hepatic accumulation of toxic fatty acid products and lack of adequate energy production. Elevations in CPK were present in 81%, ALT in 72% and AST in 68% of subjects treated in preregistration studies. The elevations were greater than 5 times the upper limit of normal (ULN) of CPK in 47%, ALT in 24% and AST in 15%. Nevertheless, the changes were not considered due to the triheptanoin or MCT therapy but rather to the underlying metabolic disorder and mitochondrial dysfunction. In small trials of triheptanoin in other genetic metabolic diseases, laboratory values were reported to be unchanged during supplementation. Since its approval and more widescale use, there have been no published cases of clinically apparent liver injury attributed to triheptanoin therapy, although its total clinical experience with its use has been limited. Likelihood score: E (unlikely cause of clinically apparent liver injury). Protein Binding Triheptanoin is approximately 80% protein bound in plasma, likely serum albumin. |
| References |
[1]. Matt Shirley. Triheptanoin: First Approval. Drugs. 2020 Oct;80(15):1595-1600. [2]. A J Yeo, et al. An anaplerotic approach to correct the mitochondrial dysfunction in ataxia-telangiectasia (A-T). Mol Metab. 2021 Dec;54:101354. [3]. Kah Ni Tan, et al. Triheptanoin protects against status epilepticus-induced hippocampal mitochondrial dysfunctions, oxidative stress and neuronal degeneration. J Neurochem. 2018 Feb;144(4):431-442. |
| Additional Infomation |
Triheptanoin is a source of heptanoate fatty acids, which can be metabolized without the enzymes of long chain fatty acid oxidation. In clinical trials, patients with long chain fatty acid oxidation disorders (lc-FAODs) treated with triheptanoin are less likely to develop hypoglycemia, cardiomyopathy, rhabdomyolysis, and hepatomegaly. Complications in lc-FAOD patients are reduced from approximately 60% to approximately 10% with the addition of triheptanoin. Triheptanoin was granted FDA approval on 30 June 2020. Triheptanoin is a Medium-chain Triglyceride. Triheptanoin is synthetic, medium-chain triglyceride that was developed for nutritional support of patients with long-chain fatty acid oxidation disorders. Triheptanoin is given orally by feeding tube titrated to provide approximately 30% of calories. Triheptanoin therapy has not been associated with elevations in serum aminotransferase or bilirubin levels or to instances of clinically apparent liver injury. Drug Indication Triheptanoin is a medium chain triglyceride indicated to provide calories and fatty acids to treat long chain fatty acid oxidation disorders (lc-FAODs). FDA Label Mechanism of Action Triheptanoin is a source of heptanoate fatty acids, which can be metabolized without the enzymes of long chain fatty acid oxidation. In clinical trials, patients with lc-FAODs treated with triheptanoin experienced improvements in hypoglycemia, cardiomyopathy, and rhabdomyolysis. Pharmacodynamics Triheptanoin is a source of medium chain fatty acids for patients with lc-FAODs. It has a moderate duration of action and a wide therapeutic window. Patients should be counselled regarding the risk of feeding tube dysfunction and intestinal malabsorption due to pancreatic insufficiency. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~116.66 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.83 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.83 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.83 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3331 mL | 11.6656 mL | 23.3312 mL | |
| 5 mM | 0.4666 mL | 2.3331 mL | 4.6662 mL | |
| 10 mM | 0.2333 mL | 1.1666 mL | 2.3331 mL |