Physicochemical Properties
| Molecular Formula | C45H53NO14 |
| Molecular Weight | 831.912 |
| Exact Mass | 831.346 |
| CAS # | 71610-00-9 |
| PubChem CID | 6436208 |
| Appearance | White to off-white solid powder |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 929.5±65.0 °C at 760 mmHg |
| Melting Point | 139-141ºC |
| Flash Point | 516.0±34.3 °C |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.615 |
| LogP | 6.59 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 14 |
| Rotatable Bond Count | 14 |
| Heavy Atom Count | 60 |
| Complexity | 1790 |
| Defined Atom Stereocenter Count | 11 |
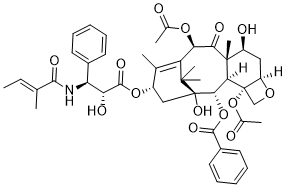
| SMILES | C/C=C(\C)/C(=O)N[C@@H](C1=CC=CC=C1)[C@H](C(=O)O[C@H]2C[C@]3([C@H]([C@H]4[C@@]([C@H](C[C@@H]5[C@]4(CO5)OC(=O)C)O)(C(=O)[C@@H](C(=C2C)C3(C)C)OC(=O)C)C)OC(=O)C6=CC=CC=C6)O)O |
| InChi Key | DBXFAPJCZABTDR-WBYYIXQISA-N |
| InChi Code | InChI=1S/C45H53NO14/c1-9-23(2)39(52)46-33(27-16-12-10-13-17-27)34(50)41(54)58-29-21-45(55)38(59-40(53)28-18-14-11-15-19-28)36-43(8,30(49)20-31-44(36,22-56-31)60-26(5)48)37(51)35(57-25(4)47)32(24(29)3)42(45,6)7/h9-19,29-31,33-36,38,49-50,55H,20-22H2,1-8H3,(H,46,52)/b23-9+/t29-,30-,31+,33-,34+,35+,36-,38-,43+,44-,45+/m0/s1 |
| Chemical Name | [(1S,2S,3R,4S,7R,9S,10S,12R,15S)-4,12-diacetyloxy-1,9-dihydroxy-15-[(2R,3S)-2-hydroxy-3-[[(E)-2-methylbut-2-enoyl]amino]-3-phenylpropanoyl]oxy-10,14,17,17-tetramethyl-11-oxo-6-oxatetracyclo[11.3.1.03,10.04,7]heptadec-13-en-2-yl] benzoate |
| Synonyms | NSC318735 NSC 318735 NSC 318735 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| References |
[1]. Synthesis, isolation, stereostructure and cytotoxicity of paclitaxel analogs from cephalomannine.Fitoterapia. 2013 Oct;90:79-84. [2]. Microbial transformation of cephalomannine by Luteibacter sp. J Nat Prod. 2007 Dec;70(12):1846-9. [3]. Cephalomannine inhibits hypoxia-induced cellular function via the suppression of APEX1/HIF-1α interaction in lung cancer. Cell death & disease vol. 2021;12(5):490. [4]. UBE2S as a novel ubiquitinated regulator of p16 and β-catenin to promote bone metastasis of prostate cancer. Int J Biol Sci. 2022 May 16;18(8):3528-3543. |
| Additional Infomation |
Taxol B has been reported in Taxus sumatrana, Taxus cuspidata, and other organisms with data available. Cephalomannine is a diterpene taxane obtained from the bark and leaves of the yew tree (Taxus brevifolia) and can convert to taxol. (NCI) |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 100 mg/mL (~120.21 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (3.01 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (3.01 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.2021 mL | 6.0103 mL | 12.0205 mL | |
| 5 mM | 0.2404 mL | 1.2021 mL | 2.4041 mL | |
| 10 mM | 0.1202 mL | 0.6010 mL | 1.2021 mL |