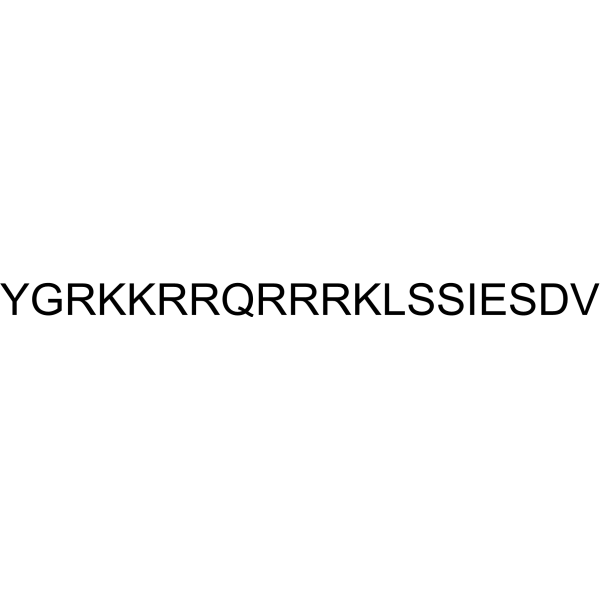Physicochemical Properties
| Molecular Formula | C105H188N42O30 |
| Molecular Weight | 2518.8796 |
| Exact Mass | 2517.447 |
| CAS # | 500992-11-0 |
| Related CAS # | Tat-NR2B9c TFA;1834571-04-8 |
| PubChem CID | 44568939 |
| Appearance | White to off-white solid powder |
| Density | 1.5±0.1 g/cm3 |
| Index of Refraction | 1.670 |
| LogP | -11.95 |
| Hydrogen Bond Donor Count | 49 |
| Hydrogen Bond Acceptor Count | 40 |
| Rotatable Bond Count | 99 |
| Heavy Atom Count | 177 |
| Complexity | 5380 |
| Defined Atom Stereocenter Count | 20 |
| SMILES | CC[C@H](C)[C@@H](C(=O)N[C@@H](CCC(=O)O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](C(C)C)C(=O)O)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCCNC(=N)N)NC(=O)[C@H](CCCNC(=N)N)NC(=O)[C@H](CCCNC(=N)N)NC(=O)[C@H](CCC(=O)N)NC(=O)[C@H](CCCNC(=N)N)NC(=O)[C@H](CCCNC(=N)N)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCCNC(=N)N)NC(=O)CNC(=O)[C@H](CC1=CC=C(C=C1)O)N |
| InChi Key | XWQVQFBTSBCKLI-FKXNDIMNSA-N |
| InChi Code | InChI=1S/C105H188N42O30/c1-7-55(6)80(98(175)140-69(34-36-77(154)155)92(169)143-72(50-148)95(172)142-71(48-78(156)157)94(171)146-79(54(4)5)99(176)177)147-97(174)74(52-150)145-96(173)73(51-149)144-93(170)70(46-53(2)3)141-90(167)62(22-10-13-39-108)133-85(162)63(24-15-41-124-101(113)114)135-87(164)65(26-17-43-126-103(117)118)136-88(165)66(27-18-44-127-104(119)120)138-91(168)68(33-35-75(110)152)139-89(166)67(28-19-45-128-105(121)122)137-86(163)64(25-16-42-125-102(115)116)134-84(161)61(21-9-12-38-107)132-83(160)60(20-8-11-37-106)131-82(159)59(23-14-40-123-100(111)112)130-76(153)49-129-81(158)58(109)47-56-29-31-57(151)32-30-56/h29-32,53-55,58-74,79-80,148-151H,7-28,33-52,106-109H2,1-6H3,(H2,110,152)(H,129,158)(H,130,153)(H,131,159)(H,132,160)(H,133,162)(H,134,161)(H,135,164)(H,136,165)(H,137,163)(H,138,168)(H,139,166)(H,140,175)(H,141,167)(H,142,172)(H,143,169)(H,144,170)(H,145,173)(H,146,171)(H,147,174)(H,154,155)(H,156,157)(H,176,177)(H4,111,112,123)(H4,113,114,124)(H4,115,116,125)(H4,117,118,126)(H4,119,120,127)(H4,121,122,128)/t55-,58-,59-,60-,61-,62-,63-,64-,65-,66-,67-,68-,69-,70-,71-,72-,73-,74-,79-,80-/m0/s1 |
| Chemical Name | (4S)-4-[[(2S,3S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-6-amino-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-5-amino-2-[[(2S)-2-[[(2S)-2-[[(2S)-6-amino-2-[[(2S)-6-amino-2-[[(2S)-2-[[2-[[(2S)-2-amino-3-(4-hydroxyphenyl)propanoyl]amino]acetyl]amino]-5-carbamimidamidopentanoyl]amino]hexanoyl]amino]hexanoyl]amino]-5-carbamimidamidopentanoyl]amino]-5-carbamimidamidopentanoyl]amino]-5-oxopentanoyl]amino]-5-carbamimidamidopentanoyl]amino]-5-carbamimidamidopentanoyl]amino]-5-carbamimidamidopentanoyl]amino]hexanoyl]amino]-4-methylpentanoyl]amino]-3-hydroxypropanoyl]amino]-3-hydroxypropanoyl]amino]-3-methylpentanoyl]amino]-5-[[(2S)-1-[[(2S)-3-carboxy-1-[[(1S)-1-carboxy-2-methylpropyl]amino]-1-oxopropan-2-yl]amino]-3-hydroxy-1-oxopropan-2-yl]amino]-5-oxopentanoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- The primary target of Tat-NR2B9c is the PDZ domain interaction between postsynaptic density protein 95 (PSD-95) and the NR2B subunit of N-methyl-D-aspartate (NMDA) receptors.[3] |
| ln Vitro | Tat-NR2B9c is an inhibitor of PSD-95 that has an EC50 of 6.7 nM for PSD-95d2 and an affinity for this domain that is almost 100 times higher than PSD-95d1 (EC50, 0.67 μM). The NMDAR2A, NMDAR2B, and NMDAR2C binding to PSD-95 is inhibited by Tat-NR2B9c, with an IC50 of 0.5 μM, ~8 μM, and 0.75 μM, respectively. With an IC50 of around 0.2 μM, Tat-NR2B9c also inhibits the interaction between PSD-95 and nNOS [1]. Tat-NR2B9c showed no effect on NMDA-induced JNK activation, but it decreased p38 activation and PSD-95 interaction with GluN2B in YAC128 striatum tissue by around 50% and 50%, respectively [2]. |
| ln Vivo |
In male C57BL/6 mice, Tat-NR2B9c (10 nmol/g, i.v.) decreases infarct volume, but has little impact at 3 nM/g [3]. - Tat-NR2B9c exerts neuroprotective effects in a mouse model of transient middle cerebral artery occlusion (tMCAO, a model of ischemic stroke). Intravenous administration of Tat-NR2B9c at doses of 0.3, 1, and 3 mg/kg (given 1 hour after tMCAO onset) reduces cerebral infarct volume in a dose-dependent manner: the 3 mg/kg group shows a 40% reduction in infarct volume compared to the vehicle control group (saline). Additionally, the 3 mg/kg group exhibits significant improvement in neurobehavioral function (assessed by the Bederson score and rotarod test): Bederson scores decrease from 2.5 (control) to 1.2, and rotarod latency increases from 45 seconds (control) to 80 seconds at 24 hours post-tMCAO [3] - Dose translation between species is critical for Tat-NR2B9c efficacy. In mice, doses below 0.3 mg/kg show no significant neuroprotection (infarct volume reduction <10%), while doses above 3 mg/kg do not further enhance efficacy (infarct volume reduction plateaus at ~40%). Pharmacokinetic analysis shows that the mouse effective plasma concentration (Cₑff) of Tat-NR2B9c is 50–100 nM, which guides dose conversion for potential use in larger species [3] |
| Enzyme Assay |
1. Prepare brain homogenates from tMCAO mice (24 hours post-tMCAO) treated with Tat-NR2B9c (3 mg/kg) or vehicle. Lyse brain tissue in RIPA buffer containing protease inhibitors, and centrifuge at 12,000 × g for 15 minutes at 4°C to collect supernatants. 2. Incubate 500 μg of total protein with anti-PSD-95 antibody (2 μg) at 4°C overnight, then add protein A/G agarose beads and incubate for another 4 hours with gentle shaking. 3. Centrifuge the mixture at 2,000 × g for 5 minutes, discard the supernatant, and wash the beads 3 times with cold RIPA buffer to remove non-specific binding. 4. Resuspend the beads in SDS sample buffer, boil for 5 minutes, and perform Western blot analysis using anti-NR2B antibody to detect the amount of NR2B co-immunoprecipitated with PSD-95. 5. Quantify the band intensity using densitometry software. The Tat-NR2B9c-treated group shows a 60% reduction in PSD-95-NR2B complex formation compared to the vehicle group [3] |
| Cell Assay |
1. Isolate cortical neurons from E18 rat embryos, culture in neurobasal medium supplemented with B27 and glutamine for 7–10 days (until mature). 2. Pre-treat neurons with Tat-NR2B9c (10, 30, 100 nM) for 1 hour, then induce excitotoxicity by adding NMDA (100 μM) and glycine (10 μM) to the medium (without Mg²⁺) for 20 minutes. 3. Remove the NMDA-containing medium, replace with fresh neurobasal medium, and incubate for another 24 hours. 4. Assess neuron viability using the MTT assay: add MTT reagent (5 mg/mL) to each well (10 μL/well), incubate for 4 hours, dissolve formazan crystals with DMSO, and measure absorbance at 570 nm. 5. The 100 nM Tat-NR2B9c group shows a 55% increase in neuron viability compared to the NMDA-only group; the EC₅₀ for neuroprotection is ~35 nM [3] |
| Animal Protocol |
1. Use 8–10 week-old male C57BL/6 mice (n=8 per group). Anesthetize mice with isoflurane (2% for induction, 1.5% for maintenance), and maintain body temperature at 37°C with a heating pad. 2. Establish the tMCAO model: Expose the right middle cerebral artery (MCA) via a cervical incision, occlude the MCA with a silicone-coated nylon suture (diameter 0.18 mm) for 60 minutes, then withdraw the suture to allow reperfusion. 3. Prepare Tat-NR2B9c by dissolving it in sterile physiological saline (0.9% NaCl) to concentrations of 0.03, 0.1, 0.3 mg/mL (for doses of 0.3, 1, 3 mg/kg body weight, respectively). 4. Administer Tat-NR2B9c via tail vein injection (10 mL/kg body weight) at 1 hour after reperfusion. The vehicle control group receives an equal volume of sterile saline. 5. At 24 hours post-reperfusion: - Assess neurobehavioral function using the Bederson score (0 = no deficit, 4 = severe deficit) and rotarod test (latency to fall at 10 rpm). - Sacrifice mice, remove brains, and cut into 2 mm coronal slices. Stain slices with 2% 2,3,5-triphenyltetrazolium chloride (TTC) at 37°C for 20 minutes. Measure infarct volume using image analysis software (infarct volume = contralateral hemisphere volume - non-infarcted ipsilateral hemisphere volume). 6. The 3 mg/kg Tat-NR2B9c group shows the most significant neuroprotection: infarct volume reduces from 35% (control) to 21% of the ipsilateral hemisphere, and Bederson scores improve from 2.5 to 1.2 [3] |
| Toxicity/Toxicokinetics |
- In vitro, Tat-NR2B9c shows low toxicity to primary cortical neurons. At concentrations up to 300 nM (10× EC₅₀ for neuroprotection), neuron viability remains >90% (MTT assay), with no significant apoptosis (Annexin V-FITC staining, apoptotic rate <5%) [3] - In vivo, Tat-NR2B9c (0.3–3 mg/kg, single tail vein injection) is well-tolerated in mice: - Body weight: No significant change (<3% reduction) at 24 and 48 hours post-administration. - Major organs: Histopathological analysis of brain, liver, kidney, and heart shows no obvious inflammation or necrosis. - No adverse effects (e.g., seizures, lethargy) are observed [3] |
| References |
[1]. PDZ protein interactions underlying NMDA receptor-mediated excitotoxicity and neuroprotection by PSD-95 inhibitors. J Neurosci. 2007 Sep 12;27(37):9901-15. [2]. P38 MAPK is involved in enhanced NMDA receptor-dependent excitotoxicity in YAC transgenic mouse model of Huntington disease. Neurobiol Dis. 2012 Mar;45(3):999-1009. [3]. Efficacy of the PSD95 inhibitor Tat-NR2B9c in mice requires dose translation between species. J Cereb Blood Flow Metab. 2016 Mar;36(3):555-61. [4]. N-methyl-D-aspartate Receptor Subunit- And Neuronal-Type Dependence of Excitotoxic Signaling Through Post-Synaptic Density 9. J Neurochem. 2010 Nov;115(4):1045-56. |
| Additional Infomation |
Na 1 is under investigation in clinical trial NCT00728182 (Evaluating Neuroprotection in Aneurysm Coiling Therapy). - Tat-NR2B9c is a cell-penetrating peptide-based PSD-95 inhibitor, composed of the Tat cell-penetrating sequence (to enhance brain penetration) and the NR2B PDZ-binding motif (to compete with PSD-95 for binding to NR2B) [3] - Its neuroprotective mechanism involves blocking the PSD-95-NR2B interaction: This prevents PSD-95 from scaffolding NMDA receptors to downstream pro-apoptotic signaling molecules (e.g., nNOS), thereby reducing NMDA receptor-mediated excitotoxicity in ischemic stroke or neurodegenerative diseases [3] - Literature [3] emphasizes the importance of dose translation for Tat-NR2B9c: The mouse effective dose (3 mg/kg) corresponds to a human equivalent dose of ~0.25 mg/kg (based on body surface area conversion), providing a basis for clinical trial dose design [3] |
Solubility Data
| Solubility (In Vitro) | H2O : ~100 mg/mL (~39.70 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.3970 mL | 1.9850 mL | 3.9700 mL | |
| 5 mM | 0.0794 mL | 0.3970 mL | 0.7940 mL | |
| 10 mM | 0.0397 mL | 0.1985 mL | 0.3970 mL |