|
Telotristat besilate, the besilate salt of telotristat, is an analog of Telotristat ethyl (formerly LX 1032; LX 1606; trade name: Xermelo), which is an orally bioavailable tryptophan hydroxylase (TPH) inhibitor with potential antiserotonergic activity. Telotristat ethyl was approved in 2017 to treat carcinoid syndrome diarrhea.
|
Physicochemical Properties
| Molecular Formula |
C31H28CLF3N6O6S
|
| Molecular Weight |
705.1
|
| Exact Mass |
704.143
|
| Elemental Analysis |
C, 52.81; H, 4.00; Cl, 5.03; F, 8.08; N, 11.92; O, 13.61; S, 4.55
|
| CAS # |
1374745-52-4
|
| Related CAS # |
1137608-69-5 (etiprate);1033805-22-9 (ethyl);1033805-28-5 (acid);1374745-52-4 (besilate);
|
| PubChem CID |
56962367
|
| Appearance |
Typically exists as solid at room temperature
|
| Hydrogen Bond Donor Count |
4
|
| Hydrogen Bond Acceptor Count |
14
|
| Rotatable Bond Count |
9
|
| Heavy Atom Count |
48
|
| Complexity |
974
|
| Defined Atom Stereocenter Count |
2
|
| SMILES |
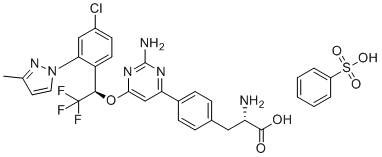
S(C1C=CC=CC=1)(O)(=O)=O.[C@@H](C1C=CC(Cl)=CC=1N1N=C(C)C=C1)(C(F)(F)F)OC1=NC(N)=NC(C2C=CC(C[C@H](N)C(=O)O)=CC=2)=C1
|
| InChi Key |
WYZQOTLDKIRRDC-VNJAQMQMSA-N
|
| InChi Code |
InChI=1S/C25H22ClF3N6O3.C6H6O3S/c1-13-8-9-35(34-13)20-11-16(26)6-7-17(20)22(25(27,28)29)38-21-12-19(32-24(31)33-21)15-4-2-14(3-5-15)10-18(30)23(36)37;7-10(8,9)6-4-2-1-3-5-6/h2-9,11-12,18,22H,10,30H2,1H3,(H,36,37)(H2,31,32,33);1-5H,(H,7,8,9)/t18-,22+;/m0./s1
|
| Chemical Name |
(2S)-2-amino-3-[4-[2-amino-6-[(1R)-1-[4-chloro-2-(3-methylpyrazol-1-yl)phenyl]-2,2,2-trifluoroethoxy]pyrimidin-4-yl]phenyl]propanoic acid;benzenesulfonic acid
|
| Synonyms |
| (S)-2-amino-3-(4-(2-amino-6-((R)-1-(4-chloro-2-(3-methyl-1H-pyrazol-1-yl)phenyl)-2,2,2-trifluoroethoxy)pyrimidin-4-yl)phenyl)propanoic acid compound with benzenesulfonic acid (1:1); 4NUK2IW1VH; 1374745-52-4 (besilate); (S)-2-Amino-3-(4-(2-amino-6-((R)-1-(4-chloro-2-(3-methyl-1H-pyrazol-1-yl)phenyl)-2,2,2-trifluoroethoxy)pyrimidin-4-yl)phenyl)propanoic acid compound with benzenesulfonic acid (1:1); (2S)-2-amino-3-[4-[2-amino-6-[(1R)-1-[4-chloro-2-(3-methylpyrazol-1-yl)phenyl]-2,2,2-trifluoroethoxy]pyrimidin-4-yl]phenyl]propanoic acid;benzenesulfonic acid; (S)-2-amino-3-(4-(2-amino-6-((r)-1-(4-chloro-2-(3-methyl-1h-pyrazol-1-yl)phenyl)-2,2,2-trifluoroethoxy)pyrimidin-4-yl)phenyl)propanoic acid benzenesulfonic acid; L-Phenylalanine, 4-(2-amino-6-((1R)-1-(4-chloro-2-(3-methyl-1H-pyrazol-1-yl)phenyl)-2,2,2-trifluoroethoxy)-4-pyrimidinyl)-, benzenesulfonate (1:1); |
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder-20°C 3 years
4°C 2 years
In solvent -80°C 6 months
-20°C 1 month
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
|
Biological Activity
| Targets |
Tryptophan hydroxylase
|
| ln Vitro |
In vitro activity: Telotristat (formerly known as LP-778902) is the active metabolite of LX1606 (Telotristat etiprate) which is an orally bioavailable, small-molecule, tryptophan hydroxylase (TPH) inhibitor with potential an in vivo IC50 of 0.028 μM and with antiserotonergic activity. Telotristat has activity in controlling diarrhea associated with carcinoid syndrome. Telotristat acts by inhibiting the enzyme tryptophan hydoxylase (TPH) and reduces serotonin production both inside and outside the GI tract without affecting brain serotonin levels. Blocking peripheral serotonin synthesis by telotristat reduces severity of both chemical- and infection-induced intestinal inflammation.
Kinase Assay: Telotristat (formerly known as LP-778902) is the active metabolite of LX1606 (Telotristat etiprate) which is an orally bioavailable, small-molecule, tryptophan hydroxylase (TPH) inhibitor with potential an in vivo IC50 of 0.028 μM and with antiserotonergic activity.
Cell Assay: BON CBA cells are grown in equal volume of DMEM and F12K with 5% bovine serum for 3-4 hours (20 K cell/well) and telotristat is added at a concentration range of 0.07 to 50 μM. The cells are incubated at 37°C overnight. 50 μM of the culture supernatant is then taken for 5HTP measurement. The supernatant is mixed with equal volume of 1M TCA, then filtered through glass fiber. The filtrate is loaded on reverse phase HPLC for 5HTP concentration measurement. The cell viability is measured by treating the remaining cells with Celltiter-Glo Luminescent Cell Viability Assay. |
|
| ln Vivo |
| LX1606 (LX 1606, LX-1606) is useful for Neurological Disease. LX1606 were given orally to mice. LX1606 reduced 5-HT significantly in the gut and blood but not in the brain. oral LX1032 reduced the severity of TNBS-induced colitis; the expression of 24% of 84 genes encoding inflammation-related cytokines and chemokines was lowered at least fourfold and the reduced expression of 17% was statistically significant [1]. Treatment with LX1606 showed a strong positive effect in ameliorating TNBS-induced IBD in mice as assessed by various parameters of disease development. These preclinical data demonstrate that inhibition of TPH activity by LX1606 may provide a new approach for the treatment of IBD and its serotonin-mediated symptoms. |
|
| Animal Protocol |
| Dissolved in 15% cyclodextrin or 0.25% methylcellulose; 300 mg/kg; p.o. | | Male C57BL/6 mice and male C57 albino mice. | |
|
| References |
Gut.2014 Jun;63(6):928-37.
|
|
Solubility Data
| Solubility (In Vitro) |
| DMSO: ≥ 100 mg/mL | | Water:N/A | | Ethanol:N/A |
|
| Solubility (In Vivo) |
| N[C@@H](CC1=CC=C(C2=NC(N)=NC(O[C@H](C3=CC=C(Cl)C=C3N4N=C(C)C=C4)C(F)(F)F)=C2)C=C1)C(O)=O.O=S(C5=CC=CC=C5)(O)=O |
(Please use freshly prepared in vivo formulations for optimal results.)
|
| Preparing Stock Solutions |
|
1 mg |
5 mg |
10 mg |
| 1 mM |
1.4182 mL |
7.0912 mL |
14.1824 mL |
| 5 mM |
0.2836 mL |
1.4182 mL |
2.8365 mL |
| 10 mM |
0.1418 mL |
0.7091 mL |
1.4182 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles. |