Syk inhibitor II, a pyrimidine-carboxamide analog, is a novel potent, cell-permeable, ATP-competitive inhibitor of Syk (Spleen tyrosine kinase) tyrosine kinase that selectively and reversibly inhibits Syk with IC50 of 41 nM. It is much less potent against PKCε, PKCβII, ZAP-70, Btk, and Itk with IC50s of 5.1, 11, 11.2, 15.5, and 22.6 µM, respectively. It has been demonstrated that Syk inhibitor II inhibits passive cutaneous anaphylaxis reactions in mice with an ID50 of 13.2 mg/kg and prevents FcεRI-mediated 5-HT release in RBL-2H3 cells in vitro with an IC50 of 460 nM. Hematopoietic cells are the primary source of expression for Spleen Tyrosine Kinase (SYK), a non-receptor cytoplasmic tyrosine kinase. Being a major mediator for several inflammatory cells, such as mast cells, B cells, neutrophils, and macrophages, SYK is a promising treatment option for inflammatory conditions as well as cancer-related indications.
Physicochemical Properties
| Molecular Formula | C14H15F3N6O | |
| Molecular Weight | 340.30 | |
| Exact Mass | 340.125 | |
| CAS # | 726695-51-8 | |
| Related CAS # | Syk Inhibitor II dihydrochloride;227449-73-2;Syk Inhibitor II dihydrochloride dihydrate;1965323-05-0 | |
| PubChem CID | 9549296 | |
| Appearance | White to off-white solid | |
| Density | 1.5±0.1 g/cm3 | |
| Boiling Point | 541.6±60.0 °C at 760 mmHg | |
| Flash Point | 281.4±32.9 °C | |
| Vapour Pressure | 0.0±1.4 mmHg at 25°C | |
| Index of Refraction | 1.633 | |
| LogP | 1.25 | |
| Hydrogen Bond Donor Count | 4 | |
| Hydrogen Bond Acceptor Count | 9 | |
| Rotatable Bond Count | 6 | |
| Heavy Atom Count | 24 | |
| Complexity | 422 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | C1(NCCN)=NC=C(C(N)=O)C(NC2=CC=CC(C(F)(F)F)=C2)=N1 |
|
| InChi Key | FQNFLNSVHWCZML-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C14H15F3N6O/c15-14(16,17)8-2-1-3-9(6-8)22-12-10(11(19)24)7-21-13(23-12)20-5-4-18/h1-3,6-7H,4-5,18H2,(H2,19,24)(H2,20,21,22,23) | |
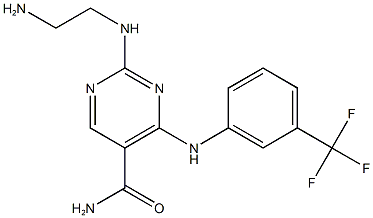
| Chemical Name | 2-(2-aminoethylamino)-4-[3-(trifluoromethyl)anilino]pyrimidine-5-carboxamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Syk (IC50 = 41 nM); serotonin (IC50 = 460 nM); PKCε (IC50 = 5.1 μM); PKCβ2 (IC50 = 11 μM); ZAP-70 (IC50 = 11.2 μM); Btk (IC50 = 15.5 μM); Itk (IC50 = 22.6 μM) Syk Inhibitor II (Compound 9a) exhibits a negligible impact on PKCε, PKCβ2, ZAP-70, Btk, and Itk, as seen by their respective IC50 values of 5.1 μM, 11 μM, 11.2 μM, 15.5 μM, and 22.6 μM [1]. |
| ln Vitro |
Syk Inhibitor II (Compound 9a) exhibits a negligible impact on PKCε, PKCβ2, ZAP-70, Btk, and Itk, as seen by their respective IC50 values of 5.1 μM, 11 μM, 11.2 μM, 15.5 μM, and 22.6 μM [1]. A series of 4-anilinopyrimidine-5-carboxamide derivatives were synthesized and evaluated as Syk inhibitors. Compound 9a potently inhibited Syk enzymatic activity with an IC₅₀ of 0.041 µM and showed good selectivity against other kinases including ZAP-70, c-Src, Itk, Btk, and PKC isoforms. In RBL-2H3 cell-based assays, 9a inhibited antigen-induced serotonin (5-HT) release with an IC₅₀ of 0.46 µM. Structure-activity relationship (SAR) studies revealed that a 2-(2-aminoethylamino) moiety on the pyrimidine ring and a primary 5-carboxamide group were crucial for Syk inhibitory activity. Modification of the 4-anilino group indicated that meta-substitution, particularly with small alkyl (e.g., 9j, IC₅₀ = 0.03 µM) or bromo (9r, IC₅₀ = 0.023 µM) groups, enhanced potency. The 3’-bromo derivative 9r was the most potent in the series in both enzyme and cellular (5-HT release, IC₅₀ = 0.27 µM) assays. [1] |
| ln Vivo |
Mice were given Syk Inhibitor II (Compound 9a; 10-100 mg/kg) subcutaneously half an hour prior to the appropriate challenge. Syk Inhibitor II has an ID50 value of 13.2 mg/kg and inhibits the reaction in a dose-dependent manner[1]. Compound 9a was evaluated in a mouse model of passive cutaneous anaphylaxis (PCA). Subcutaneous administration of 9a (30 min before antigen challenge) dose-dependently inhibited the PCA reaction with an ID₅₀ of 13.2 mg/kg. [1] |
| Enzyme Assay |
Kinase activity assays for Syk, ZAP-70, Itk, and Btk were performed using a scintillation proximity assay (SPA) system. Recombinant kinases were expressed in insect cells and partially purified. Biotinylated substrate peptides specific for each kinase were used. The reaction mixture containing kinase, substrate peptide, MgCl₂ or MnCl₂, DTT, and [γ-³²P]ATP was incubated with test compounds at room temperature for 1 hour. The reaction was terminated by adding PBS containing SPA beads, EDTA, ATP, and Triton X-100. After precipitation of beads, radioactivity was measured to calculate kinase inhibition. [1] For PKCβ2 and PKCε assays, recombinant enzymes and a specific pseudosubstrate peptide were used under similar SPA conditions. [1] |
| Cell Assay |
Inhibition of serotonin (5-HT) release from rat basophilic leukemia (RBL-2H3) cells was measured. Cells were pre-loaded with tritium-labeled 5-HT overnight and primed with dinitrophenyl (DNP)-specific IgE antibody. Cells were then incubated with or without test compounds, followed by stimulation with DNP-conjugated bovine serum albumin. Aliquots of the culture supernatant were collected, and the released radioactivity was quantified to determine the inhibitory effect of compounds on degranulation. [1] |
| Animal Protocol |
The anti-allergic activity was evaluated in a mouse passive cutaneous anaphylaxis (PCA) model. ICR mice were passively sensitized by subcutaneous injection of anti-DNP IgE under the right ear pinna. After 24 hours, mice were challenged intravenously with a mixture of DNP-conjugated bovine serum albumin and Evans blue dye to induce the PCA reaction. Test compound 9a or vehicle (control) was administered subcutaneously 30 minutes before the antigen challenge. Thirty minutes after challenge, mice were sacrificed, and both ears were collected. The dye extracted from the tissues with formamide was colorimetrically measured at 620 nm. The inhibition ratio was calculated based on the difference in dye leakage between the sensitized and unsensitized ears in treated versus control groups. [1] |
| References |
[1]. Synthetic studies on novel Syk inhibitors. Part 1: Synthesis and structure-activity relationships of pyrimidine-5-carboxamide derivatives. Bioorg Med Chem. 2005 Aug 15;13(16):4936-51. |
| Additional Infomation |
Syk is a cytoplasmic non-receptor tyrosine kinase expressed in hematopoietic cells and is involved in signal transduction pathways related to allergic and immunological responses, making it a therapeutic target for allergic disorders. The binding mode of compounds like 9a was proposed via molecular modeling using the Lck kinase domain as a template. The model suggested that the 4-anilino NH and 5-carboxamide groups form hydrogen bonds with residues in the Syk ATP-binding site, while the 2-ethylenediamino moiety may mimic the triphosphate group of ATP. X-ray crystallographic analysis of compound 9q (an isopropoxy analogue) supported the planar conformation of the inhibitor core stabilized by an intramolecular hydrogen bond between the 5-carboxamide and the 4-anilino NH group. [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9386 mL | 14.6929 mL | 29.3858 mL | |
| 5 mM | 0.5877 mL | 2.9386 mL | 5.8772 mL | |
| 10 mM | 0.2939 mL | 1.4693 mL | 2.9386 mL |