Physicochemical Properties
| Molecular Formula | C20H19N3O4S |
| Molecular Weight | 397.447563409805 |
| Exact Mass | 397.109 |
| CAS # | 858979-50-7 |
| PubChem CID | 23144148 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 2.5 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 28 |
| Complexity | 572 |
| Defined Atom Stereocenter Count | 0 |
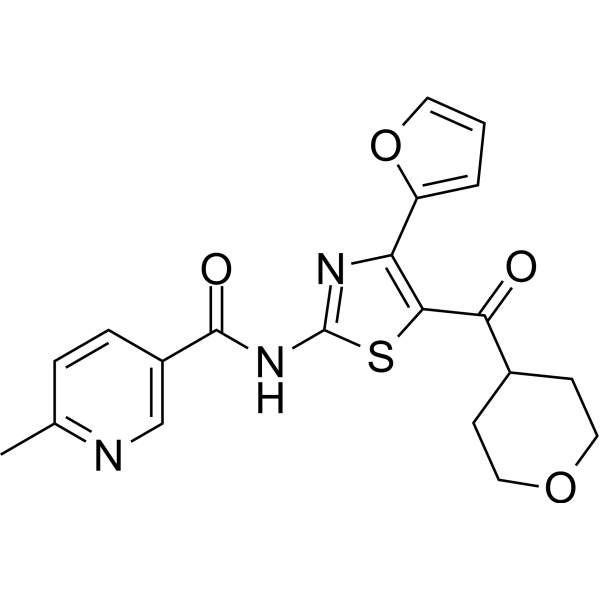
| SMILES | S1C(NC(C2C=NC(C)=CC=2)=O)=NC(C2=CC=CO2)=C1C(C1CCOCC1)=O |
| InChi Key | KMFLQPJJHQNKKF-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C20H19N3O4S/c1-12-4-5-14(11-21-12)19(25)23-20-22-16(15-3-2-8-27-15)18(28-20)17(24)13-6-9-26-10-7-13/h2-5,8,11,13H,6-7,9-10H2,1H3,(H,22,23,25) |
| Chemical Name | N-[4-(furan-2-yl)-5-(oxane-4-carbonyl)-1,3-thiazol-2-yl]-6-methylpyridine-3-carboxamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vivo | Sipagladenant (oral; 0.3 mg/kg; once) therapy improves cognitive impairment caused by decreased dopamine function in the medial prefrontal cortex [2]. Treatment with cisparadenam (oral; 0.1 mg/kg; once) improves alternating behavior [2]. Treatment with cisparadenam (oral; 0.1 mg/kg; once) improves gait metrics [2]. |
| Animal Protocol |
Animal/Disease Models: Medial prefrontal lobe dopaminergic terminal damage CD (SD) IGS male rat [2] Doses: 0.3 mg/kg Route of Administration: Oral; Route of Administration: Oral. 0.3 mg/kg; primary Experimental Results: demonstrated longer exploration time for novel objects (65.03%) than for familiar objects (34.97%) (p<0.001). Animal/Disease Models: Cognitive impairment and/or movement disorder ICR mice [2] Doses: 0.1 mg/kg Route of Administration: Oral; 0.1 mg/kg; Experimental Results: Compared with the excipient administration group (59.6%), demonstrated Dramatically higher alternation behavior (69.5%) (p<0.01). Animal/Disease Models: Cognitive impairment and/or movement impairment ICR mice [2] Doses: 0.1 mg/kg Route of Administration: Oral; 0.1 mg/kg; Experimental Results: Compared with the vehicle administration group, the maximum left hind paw The contact area and gait area increased Dramatically (p<0.05), and the maximum contact area and gait area of the right front paw tended to become larger (p<0.1). |
| References |
[1]. https://cdn.who.int/media/docs/default-source/international-nonproprietary-names-(inn)/pl127.pdf?sfvrsn=8544ca1e_3&download=true [2]. Horita, Takako. THERAPEUTIC AGENT FOR FRONTAL LOBE DYSFUNCTION, WO2016148308A1. |
| Additional Infomation | KW-6356 is a selective antagonist of adenosine A2A receptors developed by Kyowa Kirin. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~125.80 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.29 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5160 mL | 12.5802 mL | 25.1604 mL | |
| 5 mM | 0.5032 mL | 2.5160 mL | 5.0321 mL | |
| 10 mM | 0.2516 mL | 1.2580 mL | 2.5160 mL |