Physicochemical Properties
| Molecular Formula | C32H30N2O4 |
| Molecular Weight | 506.5916 |
| Exact Mass | 506.221 |
| CAS # | 63631-36-7 |
| PubChem CID | 173952 |
| Appearance | White to off-white solid powder |
| Density | 1.198g/cm3 |
| Boiling Point | 774.3ºC at 760 mmHg |
| Flash Point | 422.1ºC |
| Index of Refraction | 1.611 |
| LogP | 5.394 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 12 |
| Heavy Atom Count | 38 |
| Complexity | 732 |
| Defined Atom Stereocenter Count | 2 |
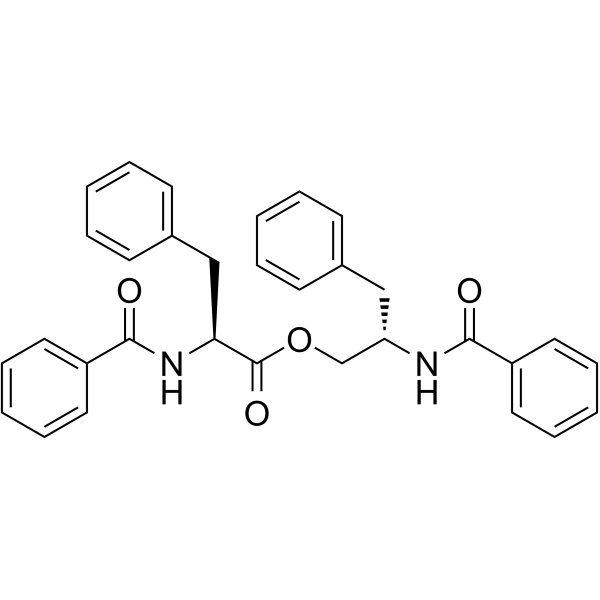
| SMILES | C1=CC=C(C=C1)C[C@@H](COC(=O)[C@H](CC2=CC=CC=C2)NC(=O)C3=CC=CC=C3)NC(=O)C4=CC=CC=C4 |
| InChi Key | CVULDJMCSSACEO-VMPREFPWSA-N |
| InChi Code | InChI=1S/C32H30N2O4/c35-30(26-17-9-3-10-18-26)33-28(21-24-13-5-1-6-14-24)23-38-32(37)29(22-25-15-7-2-8-16-25)34-31(36)27-19-11-4-12-20-27/h1-20,28-29H,21-23H2,(H,33,35)(H,34,36)/t28-,29-/m0/s1 |
| Chemical Name | [(2S)-2-benzamido-3-phenylpropyl] (2S)-2-benzamido-3-phenylpropanoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Asperphenamate completely induces autophagy, which suppresses the growth of cancer cells. In addition to having a modest inhibitory impact on cathepsin S, asperphenamate also inhibits cathepsin L. |
| References |
[1]. Synthesis of asperphenamate, a novel fungal metabolite. Phytochemistry. [2]. Total synthesis and anticancer activity studies of the stereoisomers of asperphenamate and patriscabratine. Chinese Chemical Letters Volume 21, Issue 2, February 2010, Pages 155-158. [3]. Discovery of novel cathepsin inhibitors with potent anti-metastatic effects in breast cancer cells. Bioorg Chem. 2018 Dec;81:672-680. |
| Additional Infomation |
Asperphenamate is a carboxylic ester resulting from the formal condensation of the carboxy group of N-benzoyl-L-phenylalanine with the hydroxy group of N-benzoyl-L-phenylalaninol. A metabolite found in several Pencillium and Aspergillus species, as well as in plants as a product of endophytic fungi. It has a role as an antineoplastic agent. It is a L-phenylalanine derivative, a member of benzamides and a carboxylic ester. It is functionally related to a N-benzoyl-L-phenylalanine and a N-benzoyl-L-phenylalaninol. Asperphenamate has been reported in Begonia nantoensis, Artemisia anomala, and other organisms with data available. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~197.40 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.93 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9740 mL | 9.8699 mL | 19.7398 mL | |
| 5 mM | 0.3948 mL | 1.9740 mL | 3.9480 mL | |
| 10 mM | 0.1974 mL | 0.9870 mL | 1.9740 mL |