Physicochemical Properties
| Molecular Formula | C19H23CLFN3O |
| Molecular Weight | 363.856827020645 |
| Exact Mass | 363.151 |
| CAS # | 1639220-17-9 |
| PubChem CID | 118723363 |
| Appearance | White to off-white solid powder |
| LogP | 4.5 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 25 |
| Complexity | 389 |
| Defined Atom Stereocenter Count | 0 |
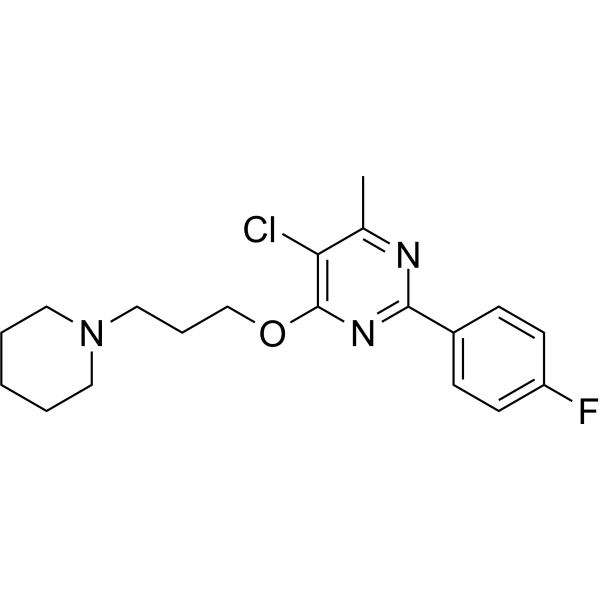
| SMILES | ClC1=C(C)N=C(C2C=CC(=CC=2)F)N=C1OCCCN1CCCCC1 |
| InChi Key | OAIHSWLLDNASTO-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C19H23ClFN3O/c1-14-17(20)19(25-13-5-12-24-10-3-2-4-11-24)23-18(22-14)15-6-8-16(21)9-7-15/h6-9H,2-5,10-13H2,1H3 |
| Chemical Name | 5-chloro-2-(4-fluorophenyl)-4-methyl-6-(3-piperidin-1-ylpropoxy)pyrimidine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
137 exhibited high binding affinity for the sigma-1 receptor (Ki σ₁ = 1.06 nM) and excellent selectivity over the sigma-2 receptor (Ki σ₂ = 1425 nM, selectivity σ₂/σ₁ = 1344-fold). [1] In a functional phenytoin-shift assay, 137 showed a Ki ratio (without/with phenytoin) of 0.87, confirming its antagonist profile at the sigma-1 receptor. [1] 137 showed weak affinity for the hERG potassium channel (IC₅₀ = 6.98 µM), indicating a low potential for cardiac toxicity. [1] In a broad selectivity panel against receptors and ion channels implicated in pain (e.g., μ-opioid, serotonin, NMDA, Nav1.7, TRPV1), 137 showed no significant affinity (% inhibition < 50% at 1 µM). [1] |
| ln Vivo |
In the mouse formalin test, pretreatment with 137 (80 mg/kg, ip) significantly reduced pain responses in both Phase I (licking time: 12.65 ± 5.07 s) and Phase II (licking time: 37.41 ± 8.89 s), with effects comparable to the reference sigma-1 antagonist S1RA. The antinociceptive effect was dose-dependent (ED₅₀ values: 48.36 ± 5.11 mg/kg for Phase I and 42.15 ± 3.96 mg/kg for Phase II). [1] In the rat chronic constriction injury (CCI) model of neuropathic pain, single and repeated oral administration of 137 (20, 40, 80 mg/kg) dose-dependently inhibited both mechanical allodynia (von Frey test) and thermal hyperalgesia (plantar test). The ED₅₀ values for single-dose treatment were 58.25 ± 6.04 mg/kg (mechanical) and 47.23 ± 3.87 mg/kg (thermal). [1] In the rotarod test, 137 at analgesic doses did not impair motor coordination in mice, unlike the positive control pregabalin. [1] |
| Enzyme Assay |
The sigma-1 receptor binding affinity of 137 was determined using guinea pig brain membranes. Membranes were incubated with the radioligand [³H]-(+)-pentazocine and various concentrations of the test compound (10⁻⁵ to 10⁻¹⁰ M) in Tris-HCl buffer (pH 8.0) at 25°C for 180 minutes. Nonspecific binding was defined using haloperidol (10 µM). The incubation was terminated by rapid vacuum filtration through GF/B filters, followed by washing and scintillation counting. Ki values were calculated using the Cheng-Prusoff equation. [1] The sigma-2 receptor binding assay was conducted similarly using guinea pig brain membranes incubated with [³H]-DTG in the presence of (+)-SKF-10047 (400 nM) to block sigma-1 sites. Incubation proceeded at 25°C for 120 minutes. Nonspecific binding was defined with DTG (10 µM). [1] |
| Cell Assay | The potential of 137 to block the hERG potassium channel was assessed using the whole-cell patch-clamp technique on HEK293 cells stably expressing hERG channels. Cells were voltage-clamped, and tail currents were measured at -50 mV following a depolarizing pulse to +50 mV. The effect of 137 was tested at concentrations of 0.3, 1, 3, and 10 µM. The IC₅₀ value was determined from the concentration-response curve of the tail current inhibition. [1] |
| Animal Protocol |
Formalin Test: Mice were administered 137 (dissolved in 30% PEG 400) or vehicle intraperitoneally (ip) 15 minutes before an intraplantar injection of 2.5% formalin solution (20 µL) into the hind paw. The time spent licking or biting the injected paw was recorded during Phase I (0-5 min) and Phase II (15-45 min) post-injection. [1] Chronic Constriction Injury (CCI) Model: Under anesthesia, the right sciatic nerve of rats was exposed and loosely ligated with four silk sutures. Sham-operated rats underwent exposure without ligation. Starting on day 14 post-surgery, rats were orally administered 137 (suspended in 0.5% methylcellulose) or vehicle twice daily for 4 consecutive days. Mechanical allodynia was assessed using von Frey filaments (up-down method), and thermal hyperalgesia was assessed using a plantar test apparatus (radiant heat) on day 15 (single dose) and day 18 (repeated dose). [1] Rotarod Test: Mice were trained to stay on a rotating rod (10 rpm). After ip administration of 137 or vehicle, the latency to fall from the rod was recorded at 30, 60, 90, and 120 minutes. [1] Acute Toxicity Study: Mice were orally administered a single dose of 137 (suspended in 0.5% methylcellulose) at doses of 200, 500, 1000, 1500, and 2000 mg/kg. Mortality was recorded over 24 hours, and the LD₅₀ was calculated. [1] |
| ADME/Pharmacokinetics |
In rats following a single intravenous (iv) dose (16 mg/kg), 137 had a half-life (t₁/₂) of 10.57 hours and an AUC₀‑∞ of 3686.17 ng·h/mL. [1] Following a single oral (po) dose (320 mg/kg), 137 had a t₁/₂ of 3.81 hours, a Tmax of 0.5 hours, a Cmax of 17394.90 ng/mL, and an AUC₀‑∞ of 55393.45 ng·h/mL. [1] The oral bioavailability (F) of 137 in rats was calculated to be 75.1%. [1] |
| Toxicity/Toxicokinetics |
The median lethal dose (LD₅₀) of 137 in mice following oral administration was greater than 2000 mg/kg. [1] No treatment-related adverse effects or motor impairments were observed in behavioral tests at efficacious doses. [1] |
| References |
[1]. Synthesis and biological evaluation of novel sigma-1 receptor antagonists based on pyrimidine scaffold as agents for treating neuropathic pain. J Med Chem. 2014 Dec 26;57(24):10404-23. |
| Additional Infomation |
137 (5-Chloro-2-(4-chlorophenyl)-4-methyl-6-(3-(piperidin-1-yl)propoxy)pyrimidine) is a novel, potent, and selective sigma-1 receptor antagonist identified from a pyrimidine-based scaffold. [1] It is proposed as a potential therapeutic agent for the treatment of neuropathic pain, based on its efficacy in preclinical pain models, favorable pharmacokinetic profile, and good safety margin. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~12.5 mg/mL (~34.35 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.25 mg/mL (3.44 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7483 mL | 13.7415 mL | 27.4831 mL | |
| 5 mM | 0.5497 mL | 2.7483 mL | 5.4966 mL | |
| 10 mM | 0.2748 mL | 1.3742 mL | 2.7483 mL |