SCH 412348 (SCH-412348) is a novel and potent competitive antagonists of the human A(2A) receptor (K(i) = 1.1 and 0.6 nM, respectively) with potential anti-PD (Parkinson disease). It has >1000-fold selectivity over all other adenosine receptors, making it the most selective A(2A) receptor antagonist reported to date. SCH 412348 attenuates hypolocomotion induced by the A(2A) receptor agonist CGS-21680 [2-[p-(2-carboxyethyl)phenethylamino]-5'-N-ethylcarboxamidoadenosine], suggesting that it inhibits A(2A) receptor activity in vivo. Its high degree of selectivity and robust in vivo activity make SCH 412348 useful tools to investigate the role of the A(2A) receptor system in animal models of PD and depression. Oral administration of preladenant and SCH 412348 (0.1-1 mg/kg) to rats potentiated 3,4-dihydroxy-L-phenylalanine (L-Dopa)-induced contralateral rotations after 6-hydroxydopamine lesions in the medial forebrain bundle and potently attenuated the cataleptic effects of haloperidol. Preladenant (1 mg/kg) inhibited L-Dopa-induced behavioral sensitization after repeated daily administration, which suggests a reduced risk of the development of dyskinesias. Finally, preladenant and SCH 412348 exhibited antidepressant-like profiles in models of behavioral despair, namely the mouse tail suspension test and the mouse and rat forced swim test. These studies demonstrate that preladenant and SCH 412348 are potent and selective A(2A) receptor antagonists and provide further evidence of the potential therapeutic benefits of A(2A) receptor inhibition in PD (with reduced risk of dyskinesias) and depression (one of the primary nonmotor symptoms of PD).
Physicochemical Properties
| Molecular Formula | C22H21F2N9O |
| Molecular Weight | 465.458649396896 |
| Exact Mass | 465.183 |
| CAS # | 377727-26-9 |
| PubChem CID | 9912318 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 2.3 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 34 |
| Complexity | 701 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | JZAMQDDHRXHDFR-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C22H21F2N9O/c23-14-3-4-17(16(24)12-14)31-8-5-30(6-9-31)7-10-32-20-15(13-26-32)21-27-19(18-2-1-11-34-18)29-33(21)22(25)28-20/h1-4,11-13H,5-10H2,(H2,25,28) |
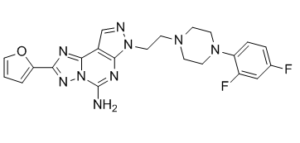
| Chemical Name | 10-[2-[4-(2,4-difluorophenyl)piperazin-1-yl]ethyl]-4-(furan-2-yl)-3,5,6,8,10,11-hexazatricyclo[7.3.0.02,6]dodeca-1(9),2,4,7,11-pentaen-7-amine |
| Synonyms | SCH 412348; SCH412348; SCH-412348 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Additionally, in cells expressing recombinant human A2A receptors, Sch412348 (SCH 412348) totally antagonizes cAMP. The A2A receptor's KB value for Sch412348 was found to be 0.3 nM; this value is in good accord with the Ki value found in the radioligand binding experiment. To show selectivity for the A2B receptor, analogous functional tests were carried out with cells that expressed the A2B receptor. Sch412348 is 910 times more selective for the A2A receptor than the A2B receptor in this experiment, with a KB value of 273 nM [1]. |
| ln Vivo | Oral treatment of Sch412348 (0.1-1 mg/kg) to rats increased 3,4-dihydroxy-L-phenylalanine (L-Dopa)-induced contralateral rotation following 6-hydroxydopamine injury to the medial forebrain bundle and Effectively attenuates haloperidol. Sch412348 (1 and 3 mg/kg) dose-dependently reduced haloperidol-induced catalepsy at 1 hour [F(3,20)=3.9, p<0.05] and 4 hours [F(3,20)=7.5 , p<0.01] after delivery. Sch412348 [F(2,51) =10.6, p<0.01] (0.1-1 mg/kg) lowers immobility time in the tail suspension test (TST) in mice at a dosage of 1 mg/kg. Sch412348 (SCH 412348) significantly enhanced activity levels in mice [F(4,27)=2.9, p<0.05]. Both the 0.3 mg/kg and 3 mg/kg treatment groups were substantially more active than the vehicle-treated mice. The 1 mg/kg group neared significance (p=0.052)[1]. |
| References | [1]. Hodgson RA, et al. Characterization of the potent and highly selective A2A receptor antagonists preladenant and SCH 412348 [7-[2-[4-2,4-difluorophenyl]-1-piperazinyl]ethyl]-2-(2-furanyl)-7H-pyrazolo[4,3-e][1,2,4]triazolo[1,5-c]pyrimidin-5-amine] in rodent models of movement disorders and depression. J Pharmacol Exp Ther. 2009 Jul;330(1):294-303 |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1484 mL | 10.7421 mL | 21.4841 mL | |
| 5 mM | 0.4297 mL | 2.1484 mL | 4.2968 mL | |
| 10 mM | 0.2148 mL | 1.0742 mL | 2.1484 mL |