Physicochemical Properties
| Molecular Formula | C26H38CLN3O3 |
| Molecular Weight | 476.0512 |
| Exact Mass | 421.272 |
| CAS # | 167626-17-7 |
| Related CAS # | 478946-89-3 (OPC-28326 free); |
| PubChem CID | 219020 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 624.5±55.0 °C at 760 mmHg |
| Flash Point | 331.5±31.5 °C |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.590 |
| LogP | 3.59 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 31 |
| Complexity | 569 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | Cl[H].O=C(C1C([H])=C(C([H])([H])[H])C(=C(C([H])([H])[H])C=1[H])N([H])C(C([H])([H])C([H])([H])[H])=O)N1C([H])([H])C([H])([H])C([H])(C([H])([H])C1([H])[H])N(C([H])([H])[H])C([H])([H])C([H])([H])C1C([H])=C([H])C([H])=C([H])C=1[H].O([H])[H] |
| InChi Key | BESKMDLUOAVUJF-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C26H35N3O2/c1-5-24(30)27-25-19(2)17-22(18-20(25)3)26(31)29-15-12-23(13-16-29)28(4)14-11-21-9-7-6-8-10-21/h6-10,17-18,23H,5,11-16H2,1-4H3,(H,27,30) |
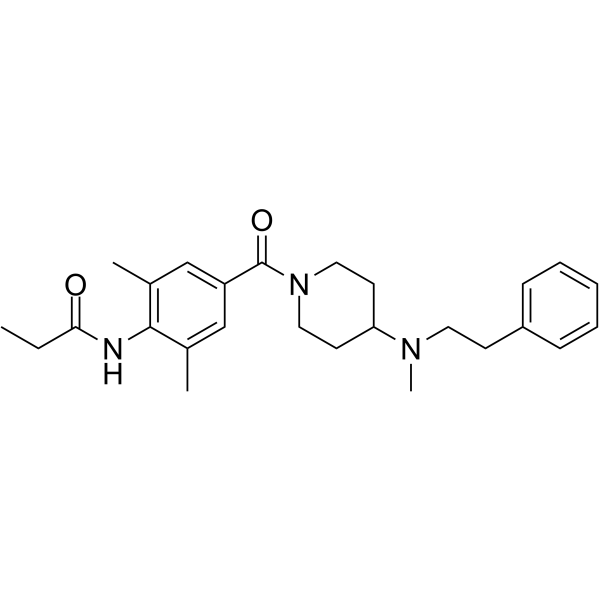
| Chemical Name | N-[2,6-dimethyl-4-[4-[methyl(2-phenylethyl)amino]piperidine-1-carbonyl]phenyl]propanamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | OPC-28326 stimulates microvessel development and improves endothelial tube formation. OPC-28326 does not alter the overall levels of eNOS or Akt, but it does cause the phosphorylation of these molecules in HAEC in a dose-dependent manner [1]. Yohimbine (10-1000 nM) and OPC-28326 (1-100 μM) elicit a rightward shift in the clonidine concentration-response curve [2]. |
| ln Vivo | OPC-28326 improves the recovery of blood flow in mice suffering from hindlimb ischemia. The ischemic leg's histologically detectable capillary count is markedly increased by OPC-28326. In eNOS−/− mice, OPC-28326 had no therapeutic effects on blood flow restoration [1]. Yohimbine and OPC-28326 (3–30 mg/kg, i.v.) generate a dose-dependent rightward change in the pressor dose–response curve induced by B-HT 920. OPC-28326 has no effect on rats' mydriasis [2]. |
| References |
[1]. Sumi M, et al. OPC-28326, a selective femoral arterial vasodilator, augments ischemia induced angiogenesis. Biomed Pharmacother. 2007 May;61(4):209-15. Epub 2006 Dec 28. [2]. Orito K, et al. alpha(2)-adrenoceptor antagonist properties of OPC-28326, a novel selective peripheral vasodilator. Br J Pharmacol. 2001 Oct;134(4):763-70 |
| Additional Infomation |
At low doses, OPC 28326 selectively vasodilates the femoral arterial bed due to its inhibitory action at alpha-2-adrenoceptors while having minimal action on systemic blood pressure, heart rate and coronary, carotid, vertebral, renal, and mesenteric blood flows. It is the only clinical compound with this profile. It is currently being investigated in the treatment of peripheral vascular diseases and Raynaud's syndrome. Drug Indication Investigated for use/treatment in peripheral vascular disease and raynaud's disease. Mechanism of Action At low doses, OPC 28326 selectively vasodilates the femoral arterial bed due to its inhibitory action at alpha-2C-adrenoceptors. Other studies have also reported selectivity for a-2B-adrenoceptors. Pharmacodynamics At low doses, OPC 28326 selectively vasodilates the femoral arterial bed due to its inhibitory action at alpha-2-adrenoceptors while having minimal action on systemic blood pressure, heart rate and coronary, carotid, vertebral, renal, and mesenteric blood flows. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1006 mL | 10.5031 mL | 21.0062 mL | |
| 5 mM | 0.4201 mL | 2.1006 mL | 4.2012 mL | |
| 10 mM | 0.2101 mL | 1.0503 mL | 2.1006 mL |